Abstract
Background
Analyse the effects of Bifidobacterium BB-12 on intestinal metabolites and serum inflammatory factors in premature infants.
Methods
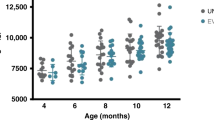
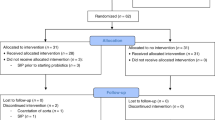
71 premature infants at gestational age of ≤32 weeks were randomly divided into the probiotic (n = 36) and control (n = 35) groups. Faecal and blood samples were collected from the two groups of premature infants at the 2nd and 4th week of life for intestinal metabolite detection and assessment of the level of the serum inflammatory markers TLR4, NF- κ B, IL-1β, and TNF- α.
Results
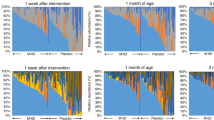
Compared to the control group, the probiotic group contained more amino acids, these elements were enriched on multiple amino acid metabolic pathways, and the probiotic group showed significantly lower levels of the serum inflammatory markers TLR4, NF-κB, IL-1β, and TNF-α. Finally, the probiotic group showed a lower incidence of feeding intolerance.
Conclusions
The administration of Bifidobacterium BB-12 is associated with increasing the levels of glutamine, glutamic acid, and kynurenine in the gut of premature infants, and associated with reducing the levels of TLR4 and NF-κB in the serum, further decreasing the secretion of the pro-inflammatory factors IL-1β and TNF-α, and alleviating systemic inflammatory reactions, thereby reducing the incidence of feeding intolerance.
Impact
-
1.
The use of Bifidobacterium BB-12 in premature infants can increase the levels of amino acids in the intestine.
-
2.
Increases in Bifidobacterium BB-12 may decrease the serum levels of TLR4, NF-κB, IL-1β, and TNF-α.
-
3.
Kynurenine may improve the prognosis of preterm infants by reducing inflammation.
-
4.
Bifidobacterium BB-12 may improve the feeding tolerance of premature infants, thus reducing the incidence of feeding intolerance.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 14 print issues and online access
$259.00 per year
only $18.50 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout










Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Kassebaum, N. J. et al. Global, regional, and national prevalence, incidence, and disability-adjusted life years for oral conditions for 195 countries, 1990-2015: a systematic analysis for the global burden of diseases, injuries, and risk factors. J. Dent. Res. 96, 380–387 (2017).
Stewart, C. J. et al. Development of the preterm gut microbiome in twins at risk of necrotising enterocolitis and sepsis. PLoS One 8, e73465 (2013).
Hackam, D. J., Afrazi, A., Good, M. & Sodhi, C. P. Innate immune signaling in the pathogenesis of necrotizing enterocolitis. Clin. Dev. Immunol. 2013, 2013475415 (2013)
Stewart, C. J. et al. Longitudinal development of the gut microbiome and metabolome in preterm neonates with late onset sepsis and healthy controls. Microbiome 5, 75 (2017).
Costello, E. K., Carlisle, E. M., Bik, E. M., Morowitz, M. J. & Relman, D. A. Microbiome assembly across multiple body sites in low-birthweight infants. mBio 4, e00782–00713 (2013).
Suez, J., Zmora, N., Segal, E. & Elinav, E. The pros, cons, and many unknowns of probiotics. Nat. Med. 25, 716–729 (2019).
Morgan, R. L. et al. Probiotics reduce mortality and morbidity in preterm, low-birth-weight infants: a systematic review and network meta-analysis of randomized trials. Gastroenterology 159, 467–480 (2020).
Chernikova, D. A. et al. The premature infant gut microbiome during the first 6 weeks of life differs based on gestational maturity at birth. Pediatr. Res. 84, 71–79 (2018).
Mazziotta, C., Tognon, M., Martini, F., Torreggiani, E. & Rotondo, J. C. Probiotics mechanism of action on immune cells and beneficial effects on human health. Cells 12, 184 (2023).
Hui, Y. et al. The effect of early probiotic exposure on the preterm infant gut microbiome development. Gut Microbes 13, 1951113 (2021).
Lundelin, K., Poussa, T., Salminen, S. & Isolauri, E. Long-term safety and efficacy of perinatal probiotic intervention: evidence from a follow-up study of four randomized, double-blind, placebo-controlled trials. Pediatr. Allergy Immunol. 28, 170–175 (2017).
Tan, T. P. et al. Safety of Bifidobacterium animalis Subsp. Lactis (B. lactis) strain BB-12-supplemented yogurt in healthy children. J. Pediatr. Gastroenterol. Nutr. 64, 302–309 (2017).
Mohan, R. et al. Effects of Bifidobacterium lactis Bb12 supplementation on intestinal microbiota of preterm infants: a double-blind, placebo-controlled, randomized study. J. Clin. Microbiol. 44, 4025–4031 (2006).
Gong, Chen, Yang, Li. Qi & Cheng, Yan Effects of probiotics on intestinal flora metabolism and clinical diseases in preterm infants in NICU. J. Anhui Med. Univ. 56, 1132–1136 (2021).
Butel, M. J. et al. Conditions of bifidobacterial colonization in preterm infants: a prospective analysis. J. Pediatr. Gastroenterol. Nutr. 44, 577–582 (2007).
Penders, J. et al. Factors influencing the composition of the intestinal microbiota in early infancy. Pediatrics 118, 511–521 (2006).
Samara, J. et al. Supplementation with a probiotic mixture accelerates gut microbiome maturation and reduces intestinal inflammation in extremely preterm infants. Cell Host Microbe 30, 696–711.e695 (2022).
Ying, Sui Efficacy of Bifidobacterium Bb-12 in the treatment of neonatal feeding intolerance. Chin. Mod. Dr. 56, 43–46 (2018).
Jie, Zhang et al. Efficacy analysis of Bifidobacterium triple viable powder in the treatment of neonatal necrotizing enterocolitis. Modern practical. Medicine 29, 347–349 (2017).
Si, Zhou, Chen, Qiuyang & Lin, ZhuangCheng Prevention of neonatal antibiotic-related diarrhea by Bifidobacterium. Heilongjiang Med. 31, 303–304 (2018).
Ursell, L. K. et al. The intestinal metabolome: an intersection between microbiota and host. Gastroenterology 146, 1470–1476 (2014).
Bistoletti, M., Bosi, A., Banfi, D., Giaroni, C. & Baj, A. The microbiota-gut-brain axis: focus on the fundamental communication pathways. Prog. Mol. Biol. Transl. Sci. 176, 43–110 (2020).
Fabiano, V. et al. Term infant formulas influencing gut microbiota: an overview. Nutrients 13, 4200 (2021).
Rooks, M. G. & Garrett, W. S. Gut microbiota, metabolites and host immunity. Nat. Rev. Immunol. 16, 341–352 (2016).
Baier, J. et al. Arginase impedes the resolution of colitis by altering the microbiome and metabolome. J. Clin. Investig. 130, 5703–5720 (2020).
Andrade, M. E. et al. Pretreatment and treatment with l-arginine attenuate weight loss and bacterial translocation in dextran sulfate sodium colitis. J. Parenter. Enter. Nutr. 40, 1131–1139 (2016).
Thomas, K. et al. Glutamine prevents acute kidney injury by modulating oxidative stress and apoptosis in tubular epithelial cells. JCI Insight 7, e163161 (2022).
Albrecht, J., Sidoryk-Węgrzynowicz, M., Zielińska, M. & Aschner, M. Roles of glutamine in neurotransmission. Neuron Glia Biol. 6, 263–276 (2010).
Soeters, P. B. & Grecu, I. Have we enough glutamine and how does it work? A clinician’s view. Ann. Nutr. Metab. 60, 17–26 (2012).
Ko, T. C., Beauchamp, R. D., Townsend, C. M. Jr. & Thompson, J. C. Glutamine is essential for epidermal growth factor-stimulated intestinal cell proliferation. Surgery 114, 147–153 (1993).
Louis, P. & Flint, H. J. Diversity, metabolism and microbial ecology of butyrate-producing bacteria from the human large intestine. FEMS Microbiol. Lett. 294, 1–8 (2009).
O’Neill, L. A. & Kaltschmidt, C. NF-kappa B: a crucial transcription factor for glial and neuronal cell function. Trends Neurosci. 20, 252–258 (1997).
Zasada M et al. Analysis of selected aspects of inflammasome function in the monocytes from neonates born extremely and very prematurely. Immunobiology 223, 18–24 (2018).
Yang, L., Ruan, X. & Wang, L. A correlation study on the effects of probiotics combined with early micro feeding on the gut microbiota and immune function of premature infants. Chin. J. Microecol. 31, 694–698 (2019).
Chi, C. et al. Effects of probiotics in preterm infants: a network meta-analysis. Pediatrics 147, e20200706 (2021).
Gao, K. et al. Increasing carbohydrate availability in the hindgut promotes hypothalamic neurotransmitter synthesis: aromatic amino acids linking the microbiota-brain axis. J. Neurochem. 149, 641–659 (2019).
Almulla, A. F. et al. The tryptophan catabolite or kynurenine pathway in schizophrenia: meta-analysis reveals dissociations between central, serum, and plasma compartments. Mol. Psychiatry 27, 3679–3691 (2022).
Gargaro, M. et al. Engagement of nuclear coactivator 7 by 3-hydroxyanthranilic acid enhances activation of aryl hydrocarbon receptor in immunoregulatory dendritic cells. Front. Immunol. 10, 1973 (2019).
Belladonna, M. L. et al. Immunosuppression via tryptophan catabolism: the role of kynurenine pathway enzymes. Transplantation 84, S17–S20 (2007).
Pedraz-Petrozzi, B. et al. Effects of inflammation on the kynurenine pathway in schizophrenia—a systematic review. J. Neuroinflammation 17, 56 (2020).
Dermyshi, E. et al. The “Golden Age” of probiotics: a systematic review and meta-analysis of randomized and observational studies in preterm infants. Neonatology 112, 9–23 (2017).
AlFaleh, K. & Anabrees, J. Probiotics for prevention of necrotizing enterocolitis in preterm infants. Evid. Based Child Health 9, 584–671 (2014).
Costeloe, K., Hardy, P., Juszczak, E., Wilks, M. & Millar, M. R. Bifidobacterium breve BBG-001 in very preterm infants: a randomised controlled phase 3 trial. Lancet 387, 649–660 (2016).
Athalye-Jape, G., Minaee, N., Nathan, E., Simmer, K. & Patole, S. Outcomes in preterm small versus appropriate for gestation infants after Bifidobacterium breve M-16V supplementation. J. Matern Fetal Neonatal. Med. 33, 2209–2215 (2020).
Sowden, M. et al. Effect of a multi-strain probiotic on the incidence and severity of necrotizing enterocolitis and feeding intolerances in preterm neonates. Nutrients 14, 3305 (2022).
Rojas, M. A. et al. Prophylactic probiotics to prevent death and nosocomial infection in preterm infants. Pediatrics 130, e1113–e1120 (2012).
Funding
This work was supported by the 2022 Quanzhou “Guiding Science and Technology programme in the field of Health Care”. (2022NS047).
Author information
Authors and Affiliations
Contributions
D.M., J.X., H.W. conceived and designed research, interpreted results of experiments, edited and revised manuscript. H.W., H.L., C.F., L.Y., and L.W. performed experiments, analyzed data, and drafted manuscript. J.X., H.W., and D.M. approved final version of manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval and consent to participate
This study has been approved by the Ethics Committee of Quanzhou Children’s Hospital (Ethics No. 8, 2022), and all premature infants participating in the study have signed informed consent forms by their guardians.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, H., Chen, D., Li, H. et al. Bifidobacterium regulates premature infant gut metabolites, reducing serum inflammatory factors: a randomised controlled trial. Pediatr Res 97, 1171–1182 (2025). https://doi.org/10.1038/s41390-024-03552-2
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41390-024-03552-2
This article is cited by
-
Changes in intestinal bacterial characteristics during hospitalization in the NICU in very low birth weight infants
Italian Journal of Pediatrics (2025)