Abstract
Study design
Experimental Study.
Objectives
To characterize the specific hindlimb electromyographic (EMG) patterns in response to muscle stretch and to measure the applied forces during stretching in the rat model of moderate SCI.
Setting
Kentucky Spinal Cord Injury Research Center, Louisville, KY, USA.
Methods
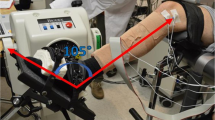
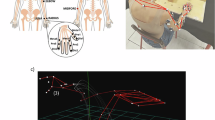
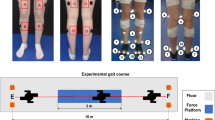
Female Sprague Dawley rats (n = 4) were instrumented for telemetry-based EMG recording (right rectus femoris and biceps femoris) and received a moderate T10 spinal cord injury (SCI). The major hindlimb muscle groups were stretched using our clinically modeled protocol. The EMG responses were recorded biweekly for 8 weeks. The forces applied during stretching were measured using a custom-designed glove. Locomotor function was assessed using the BBB Open Field Locomotor Scale, 3D kinematics and gait analysis.
Results
Three main EMG patterns in response to stretch were identified: clonic-like, air-stepping, and spasms. Torques applied during stretching ranged from 0.4-8 N•cm, and with the exception of the quadriceps, did not change significantly over the weeks of stretching. Two stretching sessions a week did not result in a significant disruption to locomotor function.
Conclusions
Stretching evokes EMG patterns in rats similar to those reported in humans including clonus and spasms. The torques used during stretching are comparable, based on the ratio of torque to body weight, to the few previously published studies that measured the forces and/or torques applied by physical therapists when stretching patients. Future studies are warranted to fully explore the impact of muscle stretch on spinal cord function after injury.
Sponsorship
DoD, KSCHIRT, NIH.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Nas K, Yazmalar L, Sah V, Aydin A, Ones K. Rehabilitation of spinal cord injuries. World J Orthop. 2015;6:8–16.
Strommen JA. Management of spasticity from spinal cord dysfunction. Neurol Clin. 2013;31:269–86.
Harvey LA, Herbert RD. Muscle stretching for treatment and prevention of contracture in people with spinal cord injury. Spinal Cord. 2002;40:1–9.
Williams PE. Use of intermittent stretch in the prevention of serial sarcomere loss in immobilised muscle. Ann Rheum Dis. 1990;49:316–7.
Harvey LA, Katalinic OM, Herbert RD, Moseley AM, Lannin NA, Schurr K. Stretch for the treatment and prevention of contracture: an abridged republication of a Cochrane Systematic Review. J Physiother. 2017;63:67–75.
Keller AV, Wainwright GN, Shum-Siu A, Prince D, Hoeper A, Martin E, et al. Disruption of locomotion in response to hindlimb muscle stretch at acute and chronic time points after a spinal cord injury in rats. J Neurotrauma. 2016;34:661–70.
Keller AV, Rees KM, Prince DC, Morehouse JR, Shum-Siu A, Magnuson DS. Dynamic “range of motion” hindlimb stretching disrupts locomotor function in rats with moderate subacute spinal cord injuries. J Neurotrauma. 2017;34:2086–91.
Young W, Elliott S. Acute effects of static stretching, proprioceptive neuromuscular facilitation stretching, and maximum voluntary contractions on explosive force production and jumping performance. Res Q Exerc Sport. 2001;72:273–9.
Nelson AG, Kokkonen J, Arnall DA. Acute muscle stretching inhibits muscle strength endurance performance. J Strength Cond Res. 2005;19:338–43.
Nelson AG, Guillory IK, Cornwell C, Kokkonen J. Inhibition of maximal voluntary isokinetic torque production following stretching is velocity-specific. J Strength Cond Res. 2001;15:241–6.
Nelson AG, Allen JD, Cornwell A, Kokkonen J. Inhibition of maximal voluntary isometric torque production by acute stretching is joint-angle specific. Res Q Exerc Sport. 2001;72:68–70.
Avela J, Kyrolainen H, Komi PV. Altered reflex sensitivity after repeated and prolonged passive muscle stretching. J Appl Physiol. 1999;86:1283–91.
Cramer JT, Housh TJ, Coburn JW, Beck TW, Johnson GO. Acute effects of static stretching on maximal eccentric torque production in women. J Strength Cond Res. 2006;20:354–8.
Harvey LA, Glinsky JA, Katalinic OM, Ben M. Contracture management for people with spinal cord injuries. NeuroRehabilitation. 2011;28:17–20.
Magnuson DS, Smith RR, Brown EH, Enzmann G, Angeli C, Quesada PM, et al. Swimming as a model of task-specific locomotor retraining after spinal cord injury in the rat. Neurorehabil Neural Repair. 2009;23:535–45.
Caudle KL, Atkinson DA, Brown EH, Donaldson K, Seibt E, Chea T, et al. Hindlimb stretching alters locomotor function after spinal cord injury in the adult rat. Neurorehabil Neural Repair. 2015;29:268–77.
Bandy WD, Irion JM. The effect of time on static stretch on the flexibility of the hamstring muscles. Phys Ther. 1994;74:845–50. discussion 50-2
Kuerzi J, Brown EH, Shum-Siu A, Siu A, Burke D, Morehouse J, et al. Task-specificity vs. ceiling effect: step-training in shallow water after spinal cord injury. Exp Neurol. 2010;224:178–87.
Basso DM, Beattie MS, Bresnahan JC. A sensitive and reliable locomotor rating scale for open field testing in rats. J Neurotrauma. 1995;12:1–21.
Jonkers BW, Sterk JC, Wouterlood FG. Transcardial perfusion fixation of the CNS by means of a compressed-air-driven device. J Neurosci Methods. 1984;12:141–9.
Magnuson DS, Trinder TC, Zhang YP, Burke D, Morassutti DJ, Shields CB. Comparing deficits following excitotoxic and contusion injuries in the thoracic and lumbar spinal cord of the adult rat. Exp Neurol. 1999;156:191–204.
Agarwal GC, Gottlieb GL. Oscillation of the human ankle joint in response to applied sinusoidal torque on the foot. J Physiol. 1977;268:151–76.
Wallace DM, Ross BH, Thomas CK. Characteristics of lower extremity clonus after human cervical spinal cord injury. J Neurotrauma. 2012;29:915–24.
Adams MM, Hicks AL. Spasticity after spinal cord injury. Spinal Cord. 2005;43:577–86.
Hultborn H, Malmsten J. Changes in segmental reflexes following chronic spinal cord hemisection in the cat. I. Increased monosynaptic and polysynaptic ventral root discharges. Acta Physiol Scand. 1983;119:405–22.
Bose P, Parmer R, Thompson FJ. Velocity-dependent ankle torque in rats after contusion injury of the midthoracic spinal cord: time course. J Neurotrauma. 2002;19:1231–49.
Bennett DJ, Gorassini M, Fouad K, Sanelli L, Han Y, Cheng J. Spasticity in rats with sacral spinal cord injury. J Neurotrauma. 1999;16:69–84.
van Gorp S, Deumens R, Leerink M, Nguyen S, Joosten EA, Marsala M. Translation of the rat thoracic contusion model; part 1-supraspinally versus spinally mediated pain-like responses and spasticity. Spinal Cord. 2014;52:524–8.
Hagbarth KE, Wallin G, Lofstedt L, Aquilonius SM. Muscle spindle activity in alternating tremor of Parkinsonism and in clonus. J Neurol Neurosurg Psychiatry. 1975;38:636–41.
Calancie B. Spinal myoclonus after spinal cord injury. J Spinal Cord Med. 2006;29:413–24.
Harvey LA, McQuade L, Hawthorne S, Byak A. Quantifying the magnitude of torque physiotherapists apply when stretching the hamstring muscles of people with spinal cord injury. Arch Phys Med Rehabil. 2003;84:1072–5.
Harvey LA, Batty J, Crosbie J, Poulter S, Herbert RD. A randomized trial assessing the effects of 4 weeks of daily stretching on ankle mobility in patients with spinal cord injuries. Arch Phys Med Rehabil. 2000;81:1340–7.
Caudle KL, Brown EH, Shum-Siu A, Burke DA, Magnuson TS, Voor MJ, et al. Hindlimb immobilization in a wheelchair alters functional recovery following contusive spinal cord injury in the adult rat. Neurorehabil Neural Repair. 2011;25:729–39.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Keller, A.V., Rees, K.M., Seibt, E.J. et al. Electromyographic patterns of the rat hindlimb in response to muscle stretch after spinal cord injury. Spinal Cord 56, 560–568 (2018). https://doi.org/10.1038/s41393-018-0069-z
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41393-018-0069-z
This article is cited by
-
Human Placental Mesenchymal Stem Cell-derived Exosomes in Combination with Hyperbaric Oxygen Synergistically Promote Recovery after Spinal Cord Injury in Rats
Neurotoxicity Research (2023)
-
Broad opioid antagonism amplifies disruption of locomotor function following therapy-like hindlimb stretching in spinal cord injured rats
Spinal Cord (2022)
-
Adjusting vascular permeability, leukocyte infiltration, and microglial cell activation to rescue dopaminergic neurons in rodent models of Parkinson’s disease
npj Parkinson's Disease (2021)