Abstract
Study design
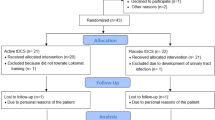
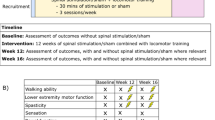
Feasibility study, consisting of random-order, cross-over study of a single intervention session, followed by a parallel-arm study of 16 sessions.
Objectives
To investigate the feasibility of a novel combinatorial approach with simultaneous delivery of transcutaneous spinal direct current stimulation (tsDCS) and locomotor training (tsDCS + LT) after spinal cord injury, compared to sham stimulation and locomotor training (sham + LT), and examine preliminary effects on walking function.
Setting
Clinical research center in the southeastern United States.
Methods
Eight individuals with chronic incomplete spinal cord injury (ISCI) completed the two-part protocol. Feasibility was assessed based on safety (adverse responses), tolerability (pain, spasticity, skin integrity), and protocol achievement (session duration, intensity). Walking function was assessed with the 10 m and 6 min walk tests.
Results
There were no major adverse responses. Minimal reports of skin irritation and musculoskeletal pain were consistent between groups. Average training peak heart rate as percent of maximum (mean(SD); tsDCS + LT: 66 (4)%, sham + LT: 69 (10)%) and Borg ratings of perceived exertion (tsDCS + LT: 17.5 (1.2), sham + LT: 14.4 (1.8)) indicate both groups trained at high intensities. Walking speed gains exceeded the minimal clinically important difference (MCID) in three of four who received tsDCS + LT (0.18 (0.29) m/s) and one of four in sham + LT (−0.05 (0.23) m/s). Gains in walking endurance exceeded the MCID in one of four in each group (tsDCS + LT: 36.4 (69.0) m, sham + LT: 4.9 (56.9) m).
Conclusions
Combinatorial tsDCS and locomotor training is safe and feasible for individuals with chronic ISCI, even those with considerable walking impairment. Study outcomes support the need to investigate the efficacy of this approach.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Behrman AL, Harkema SJ. Physical rehabilitation as an agent for recovery after spinal cord injury. Phys Med Rehabil Clin N Am. 2007;18:183–202.
Ferris DP, Gordon KE, Beres-Jones JA, Harkema SJ. Muscle activation during unilateral stepping occurs in the nonstepping limb of humans with clinically complete spinal cord injury. Spinal Cord. 2004;42:14–23.
McCrea DA, Rybak IA. Organization of mammalian locomotor rhythm and pattern generation. Brain Res Rev. 2008;57:134–46.
Beres-Jones JA, Harkema SJ. The human spinal cord interprets velocity-dependent afferent input during stepping. Brain J Neurol. 2004;127:2232–46.
Harkema SJ, Hurley SL, Patel UK, Requejo PS, Dobkin BH, Edgerton VR, et al. Human lumbosacral spinal cord interprets loading during stepping. J Neurophysiol. 1997;77:797–811.
O’Shea TM, Burda JE, Sofroniew MV. Cell biology of spinal cord injury and repair. J Clin Investig. 2017;127:3259–70.
Morawietz C, Moffat F. Effects of locomotor training after incomplete spinal cord injury: a systematic review. Arch Phys Med Rehabil. 2013;94:2297–308.
Harkema SJ. Plasticity of interneuronal networks of the functionally isolated human spinal cord. Brain Res Rev. 2008;57:255–64.
Morse LR, Field-Fote EC, Contreras-Vidal J, Noble-Haeusslein LJ, Rodreick M, Shields RK, et al. Meeting proceedings for SCI 2020: launching a decade of disruption in spinal cord injury research. J Neurotrauma. 2021;38:1251–66.
Gomes ED, Silva NA, Salgado AJ. Combinatorial therapies for spinal cord injury: strategies to induce regeneration. Neural Regen Res. 2019;14:69–71.
Griffin JM, Bradke F. Therapeutic repair for spinal cord injury: combinatory approaches to address a multifaceted problem. EMBO Mol Med. 2020;12:e11505.
Estes SP, Iddings JA, Field-Fote EC. Priming neural circuits to modulate spinal reflex excitability. Front Neurol. 2017;8:17.
Cogiamanian F, Vergari M, Schiaffi E, Marceglia S, Ardolino G, Barbieri S, et al. Transcutaneous spinal cord direct current stimulation inhibits the lower limb nociceptive flexion reflex in human beings. Pain. 2011;152:370–5.
Lamy JC, Ho C, Badel A, Arrigo RT, Boakye M. Modulation of soleus H reflex by spinal DC stimulation in humans. J Neurophysiol. 2012;108:906–14.
Murray LM, Tahayori B, Knikou M. Transspinal direct current stimulation produces persistent plasticity in human motor pathways. Sci Rep. 2018;8:717.
Winkler T, Hering P, Straube A. Spinal DC stimulation in humans modulates post-activation depression of the H-reflex depending on current polarity. Clin Neurophysiol. 2010;121:957–61.
Hubli M, Dietz V, Schrafl-Altermatt M, Bolliger M. Modulation of spinal neuronal excitability by spinal direct currents and locomotion after spinal cord injury. Clin Neurophysiol. 2013;124:1187–95.
Harkema SJ, Hillyer J, Schmidt-Read M, Ardolino E, Sisto SA, Behrman AL, et al. Locomotor training: as a treatment of spinal cord injury and in the progression of neurologic rehabilitation. Arch Phys Med Rehabil. 2012;93:1588–97.
Roy RR, Harkema SJ, Edgerton VR. Basic concepts of activity-based interventions for improved recovery of motor function after spinal cord injury. Arch Phys Med Rehabil. 2012;93:1487–97.
Park JW, Seo CH, Han SH, Lee YG. Sympathetic influence on biomechanical skin properties after spinal cord injury. Spinal Cord. 2011;49:236–43.
Gray M, Black JM, Baharestani MM, Bliss DZ, Colwell JC, Goldberg M, et al. Moisture-associated skin damage: overview and pathophysiology. J Wound Ostomy Cont Nurs. 2011;38:233–41.
National Institute for Health and Care Research. Feasibility study. In: Glossary. 2021. https://www.nihr.ac.uk/glossary/?letter=F&postcategory=-1. Accessed 10 Oct 2021.
Estes S, Zarkou A, Hope JM, Suri C, Field-Fote EC. Combined transcutaneous spinal stimulation and locomotor training to improve walking function and reduce spasticity in subacute spinal cord injury: a randomized study of clinical feasibility and efficacy. J Clin Med. 2021;10:1167.
McHugh LV, Miller AA, Leech KA, Salorio C, Martin RH. Feasibility and utility of transcutaneous spinal cord stimulation combined with walking-based therapy for people with motor incomplete spinal cord injury. Spinal Cord Ser Cases. 2020;6:104.
Megía García A, Serrano-Muñoz D, Taylor J, Avendaño-Coy J, Gómez-Soriano J. Transcutaneous spinal cord stimulation and motor rehabilitation in spinal cord injury: a systematic review. Neurorehabil Neural Repair. 2020;34:3–12.
Manson GA, Calvert JS, Ling J, Tychhon B, Ali A, Sayenko DG. The relationship between maximum tolerance and motor activation during transcutaneous spinal stimulation is unaffected by the carrier frequency or vibration. Physiol Rep. 2020;8:e14397.
Sayenko DG, Rath M, Ferguson AR, Burdick JW, Havton LA, Edgerton VR, et al. Self-assisted standing enabled by non-invasive spinal stimulation after spinal cord injury. J Neurotrauma. 2019;36:1435–50.
Awosika OO, Matthews S, Staggs EJ, Boyne P, Song X, Rizik BA, et al. Backward locomotor treadmill training combined with transcutaneous spinal direct current stimulation in stroke: a randomized pilot feasibility and safety study. Brain Commun. 2020;2:fcaa045.
Parazzini M, Fiocchi S, Liorni I, Rossi E, Cogiamanian F, Vergari M, et al. Modeling the current density generated by transcutaneous spinal direct current stimulation (tsDCS). Clin Neurophysiol. 2014;125:2260–70.
Brazg G, Fahey M, Holleran CL, Connolly M, Woodward J, Hennessy PW, et al. Effects of training intensity on locomotor performance in individuals with chronic spinal cord injury: a randomized crossover study. Neurorehabil Neural Repair. 2017;31:944–54.
Hornby TG, Reisman DS, Ward IG, Scheets PL, Miller A, Haddad D, et al. Clinical practice guideline to improve locomotor function following chronic stroke, incomplete spinal cord injury, and brain injury. J Neurol Phys Ther JNPT. 2020;44:49–100.
Behrman AL, Lawless-Dixon AR, Davis SB, Bowden MG, Nair P, Phadke C, et al. Locomotor training progression and outcomes after incomplete spinal cord injury. Phys Ther. 2005;85:1356–71.
Scivoletto G, Tamburella F, Laurenza L, Foti C, Ditunno JF, Molinari M. Validity and reliability of the 10-m walk test and the 6-min walk test in spinal cord injury patients. Spinal Cord. 2011;49:736–40.
Button KS, Ioannidis JPA, Mokrysz C, Nosek BA, Flint J, Robinson ESJ, et al. Power failure: why small sample size undermines the reliability of neuroscience. Nat Rev Neurosci. 2013;14:365–76.
Musselman KE. Clinical significance testing in rehabilitation research: what, why, and how? Phys Ther Rev. 2007;12:287–96.
Forrest GF, Hutchinson K, Lorenz DJ, Buehner JJ, Vanhiel LR, Sisto SA, et al. Are the 10 meter and 6min walk tests redundant in patients with spinal cord injury? PloS ONE. 2014;9:e94108.
Cha J, Heng C, Reinkensmeyer DJ, Roy RR, Edgerton VR, De Leon RD. Locomotor ability in spinal rats is dependent on the amount of activity imposed on the hindlimbs during treadmill training. J Neurotrauma. 2007;24:1000–12.
Field-Fote EC, Roach KE. Influence of a locomotor training approach on walking speed and distance in people with chronic spinal cord injury: a randomized clinical trial. Phys Ther. 2011;91:48–60.
Thorfinn J, Sjöberg F, Sjöstrand L, Lidman D. Perfusion of the skin of the buttocks in paraplegic and tetraplegic patients, and in healthy subjects after a short and long load. Scand J Plast Reconstr Surg Hand Surg. 2006;40:153–60.
Smith AC, Knikou M. A review on locomotor training after spinal cord injury: reorganization of spinal neuronal circuits and recovery of motor function. Neural Plast. 2016;2016:1216258.
Acknowledgements
The authors would like to acknowledge the participants and their families for their time and dedication to SCI rehabilitation research.
Funding
This work was supported by the REACT Center of NIH/NICHD [RFA-HD-15-010], Brooks-PHHP Research Collaboration, the NIH National Center for Medical Rehabilitation Research T32 Neuromuscular Plasticity Training Program [grant number T32HDO43730] and the NIH/NICHD K12 Rehabilitation Research Career Development Program [grant number HDO55929].
Author information
Authors and Affiliations
Contributions
DDF, DJC, and EJF conceived and designed the study. KAH, LAD, AV, HJS, CC, CW, GT, and EJF collected the data. KAH, LAD, HJS, and EJF performed the analysis and interpretation. KAH and EJF drafted the article. KAH, LAD, AV, HJS, CC, CW, GT, DDF, DJC, and EJF critically reviewed the draft and provided revisions. KAH, LAD, AV, HJS, CC, CW, GT, DDF, DJC, and EJF approved the final version to be published.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
The Institutional Review Board at the University of Florida approved the study (201801582) and all research was performed in accordance with the Declaration of Helsinki. We certify that all applicable institutional and governmental regulations concerning the ethical use of human volunteers were followed during the course of this research.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hawkins, K.A., DeMark, L.A., Vistamehr, A. et al. Feasibility of transcutaneous spinal direct current stimulation combined with locomotor training after spinal cord injury. Spinal Cord 60, 971–977 (2022). https://doi.org/10.1038/s41393-022-00801-1
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41393-022-00801-1
This article is cited by
-
Spinal Cord Stimulation for Functional Restoration in Patients with Chronic Spinal Cord Injury: A Review
Current Physical Medicine and Rehabilitation Reports (2025)