Abstract
Study design
Pilot study (case series).
Objective
The objective of this study was to establish spinal neurophysiological changes following high-frequency transspinal stimulation during robot-assisted step training in individuals with chronic motor complete spinal cord injury (SCI).
Setting
University research laboratory (Klab4Recovery).
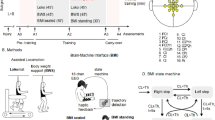
Methods
Four individuals with motor complete SCI received an average of 18 sessions of transspinal stimulation over the thoracolumbar region with a pulse train at 333 Hz during robotic-assisted step training. Each session lasted ~1 h, with an average of 240 stimulations delivered during each training session. Before and after the combined intervention, we evaluated the amplitude modulation of the long-latency tibialis anterior (TA) flexion reflex and transspinal evoked potentials (TEP) recorded from flexors and extensors during assisted stepping, and the TEP recruitment curves at rest.
Results
The long-latency TA flexion reflex was depressed in all phases of the step cycle and the phase-dependent amplitude modulation of TEPs was altered during assisted stepping, while spinal motor output based on TEP recruitment curves was increased after the combined intervention.
Conclusion
This is the first study documenting noninvasive transspinal stimulation coupled with locomotor training depresses flexion reflex excitability and concomitantly increases motoneuron output over multiple spinal segments for both flexors and extensors in people with motor complete SCI. While both transspinal stimulation and locomotor training may act via similar activity-dependent neuroplasticity mechanisms, combined interventions for rehabilitation of neurological disorders has not been systematically assessed. Our current findings support locomotor training induced neuroplasticity may be augmented with transspinal stimulation.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Data availability
The original contributions presented in the study are included in the article, further inquiries can be directed to the corresponding author.
References
Knikou M. Neural control of locomotion and training-induced plasticity after spinal and cerebral lesions. Clin Neurophysiol. 2010;121:1655–68.
Côté M-P, Murray LM, Knikou M. Spinal control of locomotion: individual neurons, their circuits and functions. Front Physiol. 2018;9:784.
Smith AC, Knikou M. A Review on locomotor training after spinal cord injury: reorganization of spinal neuronal circuits and recovery of motor function. Neural Plast 2016. 2016;12162:58
Dietz V, Wirz M, Curt A, Colombo G. Locomotor pattern in paraplegic patients: training effects and recovery of spinal cord function. Spinal Cord. 1998;36:380–90.
Nam KY, Kim HJ, Kwon BS, Park J-W, Lee HJ, Yoo A. Robot-assisted gait training (Lokomat) improves walking function and activity in people with spinal cord injury: a systematic review. J Neuroeng Rehabil. 2017;14:24.
Hofstoetter US, Freundl B, Danner SM, Krenn MJ, Mayr W, Binder H, et al. Transcutaneous spinal cord stimulation induces temporary attenuation of spasticity in individuals with spinal cord injury. J Neurotrauma. 2020;37:481–93.
Gad PN, Kreydin E, Zhong H, Latack K, Reggie Edgerton V. Non-invasive neuromodulation of spinal cord restores lower urinary tract function after paralysis. Front Neurosci. 2018;1:432.
Kreydin E, Zhong H, Latack K, Ye S, Edgerton VR, Gad P. Transcutaneous electrical spinal cord neuromodulator (TESCoN) improves symptoms of overactive bladder. Front Syst Neurosci. 2020;14:1.
Phillips AA, Squair JW, Sayenko DG, Edgerton VR, Gerasimenko Y, Krassioukov AV. An autonomic neuroprosthesis: noninvasive electrical spinal cord stimulation restores autonomic cardiovascular function in individuals with spinal cord injury. J Neurotrauma. 2018;35:446–51.
Harkema SJ, Wang S, Angeli CA, Chen Y, Boakye M, Ugiliweneza B. et al. Normalization of blood pressure with spinal cord epidural stimulation after severe spinal cord injury. Front Hum Neurosci. 2018;12:83.
Hofstoetter US, Krenn M, Danner SM, Hofer C, Kern H, McKay WB, et al. Augmentation of voluntary locomotor activity by transcutaneous spinal cord stimulation in motor-incomplete spinal cord-injured individuals. Artif Organs. 2015;39:E176–86.
Gerasimenko YP, Lu DC, Modaber M, Zdunowski S, Gad P, Sayenko DG, et al. Noninvasive reactivation of motor descending control after paralysis. J Neurotrauma. 2015;32:1968–80.
Gerasimenko Y, Sayenko D, Gad P, Kozesnik J, Moshonkina T, Grishin A, et al. Electrical spinal stimulation, and imagining of lower limb movements to modulate brain-spinal connectomes that control locomotor-like behavior. Front Physiol. 2018;9:1196.
Gerasimenko Y, Gorodnichev R, Puhov A, Moshonkina T, Savochin A, Selionov V, et al. Initiation and modulation of locomotor circuitry output with multisite transcutaneous electrical stimulation of the spinal cord in noninjured humans. J Neurophysiol. 2015;113:834–42.
Formento E, Minassian K, Wagner F, Mignardot JB, Le Goff-Mignardot CG, Rowald A, et al. Electrical spinal cord stimulation must preserve proprioception to enable locomotion in humans with spinal cord injury. Nat Neurosci. 2018;21:1728–41.
Kumru H, Benito-Penalva J, Valls-Sole J, Murillo N, Tormos JM, Flores C, et al. Placebo-controlled study of rTMS combined with Lokomat® gait training for treatment in subjects with motor incomplete spinal cord injury. Exp Brain Res. 2016;234:3447–55.
Raithatha R, Carrico C, Powell ES, Westgate PM, Chelette KC, Lee K, et al. Non-invasive brain stimulation and robot-assisted gait training after incomplete spinal cord injury: a randomized pilot study. NeuroRehabilitation. 2016;38:15–25.
Estes SP, Iddings JA, Field-Fote EC. Priming neural circuits to modulate spinal reflex excitability. Front Neurol. 2017;8:17.
Knikou M, Conway BA. Effects of electrically induced muscle contraction on flexion reflex in human spinal cord injury. Spinal Cord. 2005;43:640–8.
Knikou M. Plantar cutaneous input modulates differently spinal reflexes in subjects with intact and injured spinal cord. Spinal Cord. 2007;45:69–77.
Gorassini MA, Knash ME, Harvey PJ, Bennett DJ, Yang JF. Role of motoneurons in the generation of muscle spasms after spinal cord injury. Brain. 2004;127:2247–58.
Knikou M, Murray LM. Repeated transspinal stimulation decreases soleus H-reflex excitability and restores spinal inhibition in human spinal cord injury. PLoS ONE. 2019;14:e0223135.
Knikou M, Angeli CA, Ferreira CK, Harkema SJ. Flexion reflex modulation during stepping in human spinal cord injury. Exp Brain Res. 2009;196:341–51.
Zaaya M, Pulverenti TS, Islam MA, Knikou M. Transspinal stimulation downregulates activity of flexor locomotor networks during walking in humans. J Electromyogr Kinesiol. 2020;52:102420.
Pulverenti TS, Islam MA, Alsalman O, Murray LM, Harel NY, Knikou M. Transspinal stimulation decreases corticospinal excitability and alters the function of spinal locomotor networks. J Neurophysiol. 2019;122:2331–43.
Zehr EP, Komiyama T, Stein RB. Cutaneous reflexes during human gait: electromyographic and kinematic responses to electrical stimulation. J Neurophysiol. 1997;77:3311–25.
Zehr EP, Fujita K, Stein RB. Reflexes from the superficial peroneal nerve during walking in stroke subjects. J Neurophysiol. 1998;79:848–58.
Murray LM, Islam MA, Knikou M. Cortical and subcortical contributions to neuroplasticity after repetitive transspinal stimulation in humans. Neural Plast 2019. 2019. 4750768.
Rossignol S, Martinez M, Escalona M, Kundu A, Delivet-Mongrain H, Alluin O. et al. The ‘beneficial' effects of locomotor training after various types of spinal lesions in cats and rats. Prog Brain Res. 2015;218:173–98.
Côté M-P, Gossard J-P. Step training-dependent plasticity in spinal cutaneous pathways. J Neurosci. 2004;24:11317–27.
Martinez M, Delivet-Mongrain H, Leblond H, Rossignol S. Incomplete spinal cord injury promotes durable functional changes within the spinal locomotor circuitry. J Neurophysiol. 2012;108:124–34.
Goldshmit Y, Lythgo N, Galea MP, Turnley AM. Treadmill training after spinal cord hemisection in mice promotes axonal sprouting and synapse formation and improves motor recovery. J Neurotrauma. 2008;25:449–65.
Côté M-P, Ménard A, Gossard J-P. Spinal cats on the treadmill: changes in load pathways. J Neurosci. 2003;23:2789–96.
Knikou M, Mummidisetty CK. Locomotor training improves premotoneuronal control after chronic spinal cord injury. J Neurophysiol. 2014;111:2264–75.
Knikou M, Dixon L, Santora D, Ibrahim MM. Transspinal constant-current long-lasting stimulation: a new method to induce cortical and corticospinal plasticity. J Neurophysiol. 2015;114:1486–99.
Andén N‐E, Jukes MGM, Lundberg A, Vyklicky L. The effect of DOPA on the spinal cord. 1. Influence on transmission from primary aflerents. Acta Physiol Scand. 1966;67:373–86.
Lundberg A. Multisensory control of spinal reflex pathways. Prog Brain Res. 1979;50:11–28.
Andén NE, Jukes MGM, Lundberg A, Vyklický L. A new spinal flexor reflex. Nature. 1964;202:1344–5.
Jankowska E, Jukes MGM, Lund S, Lundberg A. The Effect of DOPA on the spinal cord 6. Half‐centre organization of interneurones transmitting effects from the flexor reflex afferents. Acta Physiol Scand. 1967;70:389–402.
Jankowska E, Jukes MGM, Lund S, Lundberg A. The Effect of DOPA on the Spinal Cord 5. Reciprocal organization of pathways transmitting excitatory action to alpha motoneurones of flexors and extensors. Acta Physiol Scand. 1967;70:369–88.
Roby-Brami A, Bussel B. Effects of flexor reflex afferent stimulation on the soleus H reflex in patients with a complete spinal cord lesion: evidence for presynaptic inhibition of Ia transmission. Exp Brain Res. 1990;81:593–601.
Roby-Brami A, Bussel B. Inhibitory effects on flexor reflexes in patients with a complete spinal cord lesion. Exp Brain Res. 1992;90:201–8.
Knikou M. Plantar cutaneous afferents normalize the reflex modulation patterns during stepping in chronic human spinal cord injury. J Neurophysiol. 2010;103:1304–14.
Knikou M, Murray LM. Neural interactions between transspinal evoked potentials and muscle spindle afferents in humans. J Electromyogr Kinesiol. 2018;43:174–83.
Smith AC, Mummidisetty CK, Rymer WZ, Knikou M. Locomotor training alters the behavior of flexor reflexes during walking in human spinal cord injury. J Neurophysiol. 2014;112:2164–75.
Knikou M. Functional reorganization of soleus H-reflex modulation during stepping after robotic-assisted step training in people with complete and incomplete spinal cord injury. Exp Brain Res. 2013;228:279–96.
Lünenburger L, Bolliger M, Czell D, Müller R, Dietz V. Modulation of locomotor activity in complete spinal cord injury. Exp Brain Res. 2006;174:638–46.
Meyns P, Van De Crommert HWAA, Rijken H, Van Kuppevelt DHJM, Duysens J. Locomotor training with body weight support in SCI: EMG improvement is more optimally expressed at a low testing speed. Spinal Cord. 2014;52:887–93.
Denny-Brown DE, Sherrington CS. Subliminal fringe in spinal flexion. J Physiol. 1928;66:175–80.
Knikou M. Neurophysiological characterization of transpinal evoked potentials in human leg muscles. Bioelectromagnetics. 2013;34:630–40.
Funding
Research reported in this publication was supported by the Spinal Cord Injury Research Board (SCIRB) of the New York State Department of Health (NYSDOH) via C32248GG and C32095GG contracts, and in part by the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD), National Institutes of Health (NIH) under award number R01HD100544 awarded to MK. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the paper.
Author information
Authors and Affiliations
Contributions
MZ: EMG data analysis, developed figures, and wrote the first draft of the paper. TSP performed experiments, contributed to paper writing and reviewing. MK formulated the hypotheses, designed the experiments, performed experiments and analysis, and contributed to paper writing and reviewing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zaaya, M., Pulverenti, T.S. & Knikou, M. Transspinal stimulation and step training alter function of spinal networks in complete spinal cord injury. Spinal Cord Ser Cases 7, 55 (2021). https://doi.org/10.1038/s41394-021-00421-6
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41394-021-00421-6
This article is cited by
-
Transspinal direct current stimulation alters neuronal excitability but not homosynaptic inhibition in human spinal cord injury: a pilot clinical trial
Experimental Brain Research (2025)
-
Soleus H-reflex amplitude modulation during walking remains physiological during transspinal stimulation in humans
Experimental Brain Research (2024)
-
Priming locomotor training with transspinal stimulation in people with spinal cord injury: study protocol of a randomized clinical trial
Trials (2023)
-
Neuromodulation with transcutaneous spinal stimulation reveals different groups of motor profiles during robot-guided stepping in humans with incomplete spinal cord injury
Experimental Brain Research (2023)
-
Brain and spinal cord paired stimulation coupled with locomotor training affects polysynaptic flexion reflex circuits in human spinal cord injury
Experimental Brain Research (2022)