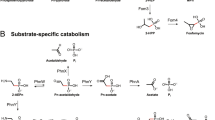
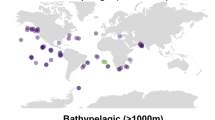
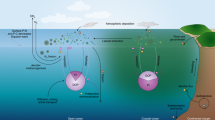
Abstract
Many areas of the ocean are nutrient-poor yet support large microbial populations, leading to intense competition for and recycling of nutrients. Organic phosphonates are frequently found in marine waters, but require specialist enzymes for catabolism. Previous studies have shown that the genes that encode these enzymes in marine systems are under Pho regulon control and so are repressed by inorganic phosphate. This has led to the conclusion that phosphonates are recalcitrant in much of the ocean, where phosphorus is not limiting despite the degradative genes being common throughout the marine environment. Here we challenge this paradigm and show, for the first time, that bacteria isolated from marine samples have the ability to mineralise 2-aminoethylphosphonate, the most common biogenic marine aminophosphonate, via substrate-inducible gene regulation rather than via Pho-regulated metabolism. Substrate-inducible, Pho-independent 2-aminoethylphosphonate catabolism therefore represents a previously unrecognised component of the oceanic carbon, nitrogen and phosphorus cycles.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Moore CM, Mills MM, Arrigo KR, Berman-Frank I, Bopp L, Boyd PW, et al. Processes and patterns of oceanic nutrient limitation. Nat Geosci. 2013;6:701–10.
Kolowith LC, Ingall ED, Benner R. Composition and cycling of marine organic phosphorus. Limnol Oceanogr. 2001;46:309–20.
Clark LL, Ingall E, Benner R. Marine organic phosphorus cycling; novel insights from nuclear magnetic resonance. Am J Sci. 1999;299:724–37.
Van Mooy BAS, Krupke A, Dyhrman ST, Fredricks HF, Frischkorn KR, Ossolinski JE, et al. Major role of planktonic phosphate reduction in the marine phosphorus redox cycle. Science. 2015;348:783–5.
Mukhamedova KS, Glushenkova AI. Natural phosphonolipids. Chem Nat Compd. 2000;36:329–41.
Steiner S, Conti SF, Lester RL. Occurrence of phosphonosphingolipids in Bdellovibrio bacteriovorus strain UKi2. J Bacteriol. 1973;116:1199–211.
White AK, Metcalf WW. Microbial metabolism of reduced phosphorus compounds. Annu Rev Microbiol. 2007;61:379–400.
Martínez A, Ventouras L-A, Wilson ST, Karl DM, DeLong EF. Metatranscriptomic and functional metagenomic analysis of methylphosphonate utilization by marine bacteria. Front Microbiol. 2013;4:340.
Dyhrman ST, Chappell PD, Haley ST, Moffett JW, Orchard ED, Waterbury JB, et al. Phosphonate utilization by the globally important marine diazotroph Trichodesmium. Nature. 2006;439:68–71.
Heath RT. Microbial turnover of organic phosphorus in aquatic systems. In: Turner BL, Frossard E, Baldwin D, editors. Organic phosphorus in the environment. Cambridge MA: CABI Publishing; 2005. p. 185–204.
Martinez A, Tyson GW, DeLong EF. Widespread known and novel phosphonate utilization pathways in marine bacteria revealed by functional screening and metagenomic analyses. Environ Microbiol. 2010;12:222–38.
Villarreal-Chiu JF, Quinn JP, McGrath JW. The genes and enzymes of phosphonate metabolism by bacteria, and their distribution in the marine environment. Front Microbiol. 2012;3:19.
McGrath JW, Chin JP, Quinn JP. Organophosphonates revealed: new insights into the microbial metabolism of ancient molecules. Nat Rev Microbiol. 2013;11:412–9.
Ternan NG, Quinn JP. Phosphate starvation-independent 2-aminoethylphosphonic acid biodegradation in a newly isolated strain of Pseudomonas putida, NG2. Syst Appl Microbiol. 1998;21:346–52.
Benitez-Nelson CR, O’Neill L, Kolowith LC, Pellechia P, Thunell R. Phosphonates and particulate organic phosphorus cycling in an anoxic marine basin. Limnol Oceanogr. 2004;49:1593–604.
Wanner BL. Signal transduction in the control of phosphate-regulated genes of Escherichia coli. Kidney Int. 1996;49:964–7.
Tyrrell T. The relative influences of nitrogen and phosphorus on oceanic primary production. Nature. 1999;400:525–31.
Karl DM, Björkman KM, Dore JE, Fujieki L, Hebel DV, Houlihan T, et al. Ecological nitrogen-to-phosphorus stoichiometry at station ALOHA. Deep Sea Res Part II Top Stud Oceanogr. 2001;48:1529–66.
Krieg NR, Holt JG. Enrichment and isolation. In: Gerhardt P editor. Manual of methods for general bacteriology. Washington, D.C: American Society for Microbiology; 1981. p. 112–42. .
Difco Laboratories. Difco manual of dehydrated culture media and reagents for microbiological and clinical laboratory procedures. 9th edn, Detroit: Difco Laboratories; 1953.
McSorley FR, Wyatt PB, Martinez A, DeLong EF, Hove-Jensen B, Zechel DL. PhnY and PhnZ comprise a new oxidative pathway for enzymatic cleavage of a carbon–phosphorus bond. J Am Chem Soc. 2012;134:8364–7.
Marchesi JR, Sato T, Weightman AJ, Martin TA, Fry JC, Hiom SJ, et al. Design and evaluation of useful bacterium-specific PCR primers that amplify genes coding for bacterial 16S rRNA. Appl Environ Microbiol. 1998;64:795–9.
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ. Basic local alignment search tool. J Mol Biol. 1990;215:403–10.
Young CL, Ingall ED. Marine dissolved organic phosphorus composition: insights from samples recovered using combined electrodialysis/reverse osmosis. Aquat Geochem. 2010;16:563–74.
Hori T, Arakawa I, Sugita M. Distribution of ceramide 2-aminoethylphosphonate and ceramide aminoethylphosphate (sphingoethanolamine) in some aquatic animals. J Biochem. 1967;62:67–70.
Kittredge JS, Hughes RR. The Occurrence of α-Amino-β-phosphonopropionic Acid in the Zoanthid, Zoanthus sociatus, and the Ciliate, Tetrahymena pyriformis. Biochemistry. 1964;3:991–6.
Kittredge JS, Roberts E, Simonsen DG. The occurrence of free 2-aminoethylphosphonic acid in the Sea Anemone, Anthopleura elegantissima. Biochemistry. 1962;1:624–8.
Quin LD. The presence of compounds with a carbon-phosphorus bond in some marine invertebrates. Biochemistry. 1965;4:324–30.
Quin LD, Quin GS. Screening for carbon-bound phosphorus in marine animals by high-resolution 31P-NMR spectroscopy: coastal and hydrothermal vent invertebrates. Comp Biochem Physiol B Biochem Mol Biol. 2001;128:173–85.
Shilova IN, Robidart JC, James Tripp H, Turk-Kubo K, Wawrik B, Post AF, et al. A microarray for assessing transcription from pelagic marine microbial taxa. ISME J. 2014;8:1476–91.
Karl DM, Björkman KM. Dynamics of DOP. In: Hansell DA, Carlson CA, editors. Biogeochemistry of marine dissolved organic matter. Academic Press Inc, London, UK; 2002. p. 249–366.
Paytan A, McLaughlin K. The oceanic phosphorus cycle. Chem Rev. 2007;107:563–76.
Mackey KRM, Labiosa RG, Calhoun M, Street JH, Post AF, Paytan A. Phosphorus availability, phytoplankton community dynamics, and taxon-specific phosphorus status in the Gulf of Aqaba, Red Sea. Limnol Oceanogr. 2007;52:873–85.
Cooley NA, Kulakova AN, Villarreal-Chiu JF, Gilbert JA, McGrath JW, Quinn JP. Phosphonoacetate biosynthesis: in vitro detection of a novel NADP(+)-dependent phosphonoacetaldehyde-oxidizing activity in cell-extracts of a Marine Roseobacter. Microbiology. 2011;80:335–40.
Borisova SA, Christman HD, Metcalf MEM, Zulkepli NA, Zhang JK, van der Donk WA, et al. Genetic and biochemical characterization of a pathway for the degradation of 2-aminoethylphosphonate in Sinorhizobium meliloti 1021. J Biol Chem. 2011;286:22283–90.
Metcalf WW, Wanner BL. Mutational analysis of an Escherichia coli fourteen-gene operon for phosphonate degradation, using TnphoA’ elements. J Bacteriol. 1993;175:3430–42.
McGrath JW, Quinn JP. A plate assay for the detection of organophosphonate mineralization by environmental bacteria, and its modification as an activity stain for identification of the carbon-phosphorus bond cleavage enzyme phosphonoacetate hydrolase. Biotechnol Tech. 1995;9:497–502.
Karl DM. Microbially mediated transformations of phosphorus in the sea: new views of an old cycle. Annu Rev Mar Sci. 2014;6:279–337.
Hoppe H, Ullrich S. Profiles of ectoenzymes in the Indian Ocean: phenomena of phosphatase activity in the mesopelagic zone. Aquat Microb Ecol. 1999;19:139–48.
Koike I, Nagata T. High potential activity of extracellular alkaline phosphatase in deep waters of the central Pacific. Deep Sea Res Part II Top Stud Oceanogr. 1997;44:2283–94.
Baltar F, Arístegui J, Sintes E, van Aken HM, Gasol JM, Herndl GJ. Prokaryotic extracellular enzymatic activity in relation to biomass production and respiration in the meso- and bathypelagic waters of the (sub)tropical Atlantic. Environ Microbiol. 2009;11:1998–2014.
Acknowledgements
We thank F. Hammerschmidt and K. Pallitsch, University of Vienna, Austria, and the late H.B.F. Dixon, Cambridge University, UK, for the provision of several phosphonate substrates, and J. Megaw, Queen’s University Belfast, UK, for the provision of a marine water sample. JPC was funded by the Department for Employment and Learning, Northern Ireland, UK.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Chin, J.P., Quinn, J.P. & McGrath, J.W. Phosphate insensitive aminophosphonate mineralisation within oceanic nutrient cycles. ISME J 12, 973–980 (2018). https://doi.org/10.1038/s41396-017-0031-7
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41396-017-0031-7
This article is cited by
-
The microbial phosphorus cycle in aquatic ecosystems
Nature Reviews Microbiology (2025)
-
The facilitating role of phycospheric heterotrophic bacteria in cyanobacterial phosphonate availability and Microcystis bloom maintenance
Microbiome (2023)
-
PCycDB: a comprehensive and accurate database for fast analysis of phosphorus cycling genes
Microbiome (2022)
-
Global and seasonal variation of marine phosphonate metabolism
The ISME Journal (2022)
-
Transporter characterisation reveals aminoethylphosphonate mineralisation as a key step in the marine phosphorus redox cycle
Nature Communications (2021)