Abstract
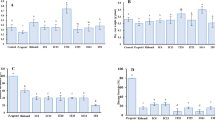
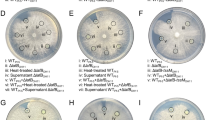
Soil and rhizosphere microbiomes play important roles in suppression of plant pathogens through production of antagonistic secondary metabolites, yet mechanisms that determine the strength of pathogen control are not well understood. Many Pseudomonas species are associated with soil and rhizosphere microbiomes, and their ability to suppress pathogens is well documented. Here, we investigate how interactions within the Pseudomonas genus affect their production of antimicrobial metabolites. From a biosensor-based screen, we identify P. capeferrum species as capable of modulating secondary metabolite production in P. protegens. We show that P. capeferrum alters production of pyoluteorin and 2,4-diacetylphloroglucinol (DAPG) in P. protegens via two distinct and sequential mechanisms that depends on spatial proximity of the two species. Specifically, P. capeferrum secretes a diffusible signal that induce pyoluteorin production up to 100-fold in neighboring P. protegens colonies. In contrast, the interaction results in reduced DAPG production, but only within mixed-species colonies. Additionally, we found that increased pyoluteorin production and cell lysis of P. capeferrum is required for inhibition of DAPG production, suggesting that pyoluteorin-facilitated antibiosis of P. protegens on P. capeferrum leads to release of cell-associated metabolites and subsequent inhibition of DAPG production in P. protegens. As the interaction modulates in vitro bioactivity of the species, genus-specific interactions may assist in improving efficacy of biocontrol strains and consortia.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Data availability
All data are included in the article and the supplemental material will be made available upon request to the corresponding author. Additionally, the de novo assembled genome sequence of P. capeferrum F8 generated in this study is available in the NCBI GenBank under the accession number CP099575.
References
Berendsen RL, Pieterse CMJ, Bakker PAHM. The rhizosphere microbiome and plant health. Trends Plant Sci. 2012;17:478–86.
Haas D, Défago G. Biological control of soil-borne pathogens by fluorescent pseudomonads. Nat Rev Microbiol. 2005;3:307–19.
Whipps JM. Microbial interactions and biocontrol in the rhizosphere. J Exp Bot. 2001;52:487–511.
Mendes R, Kruijt M, De Bruijn I, Dekkers E, Van Der Voort M, Schneider J, et al. Deciphering the rhizosphere microbiome for disease-suppressive bacteria. Science 2011;332:1097–100.
Jousset A, Becker J, Chatterjee S, Karlovsky P, Scheu S, Eisenhauer N. Biodiversity and species identity shape the antifungal activity of bacterial communities. Ecology 2014;95:1184–90.
Becker J, Eisenhauer N, Scheu S, Jousset A. Increasing antagonistic interactions cause bacterial communities to collapse at high diversity. Ecol Lett. 2012;15:468–74.
Hu J, Wei Z, Friman VP, Gu SH, Wang XF, Eisenhauer N, et al. Probiotic diversity enhances rhizosphere microbiome function and plant disease suppression. mBio. 2016;7:e01790–16.
Mehrabi Z, McMillan VE, Clark IM, Canning G, Hammond-Kosack KE, Preston G, et al. Pseudomonas spp. diversity is negatively associated with suppression of the wheat take-all pathogen. Sci Rep. 2016;6:1–10.
Ma Z, Geudens N, Kieu NP, Sinnaeve D, Ongena M, Martins JC, et al. Biosynthesis, chemical structure, and structure-activity relationship of orfamide lipopeptides produced by Pseudomonas protegens and related species. Front Microbiol. 2016;7:1–16.
Yan Q, Philmus B, Chang JH, Loper JE. Novel mechanism of metabolic co-regulation coordinates the biosynthesis of secondary metabolites in Pseudomonas protegens. Elife 2017;6:e22835.
Ramette A, Moënne-Loccoz Y, Défago G. Prevalence of fluorescent pseudomonads producing antifungal phloroglucinols and/or hydrogen cyanide in soils naturally suppressive or conducive to tobacco black root rot. FEMS Microbiol Ecol. 2003;44:35–43.
Raaijmakers JM, Weller DM. Natural Plant Protection by 2,4-Diacetylphloroglucinol-Producing Pseudomonas spp. in Take-All Decline Soils. Mol Plant-Microbe Interact. 1998;11:144–52.
Murata K, Suenaga M, Kai K. Genome Mining Discovery of Protegenins A–D, Bacterial Polyynes Involved in the Antioomycete and Biocontrol Activities of Pseudomonas protegens. ACS Chem Biol. 2021. https://pubs.acs.org/doi/10.1021/acschembio.1c00276. Online ahead of print.
Achkar J, Xian M, Zhao H, Frost JW. Biosynthesis of Phloroglucinol. J Am Chem Soc. 2005;127:5332–3.
Bangera MG, Thomashow LS. Identification and Characterization of a Gene Cluster for Synthesis of the Polyketide Antibiotic 2,4-Diacetylphloroglucinol from Pseudomonas fluorescens Q2-87. J Bacteriol. 1999;181:3155–63.
Bottiglieri M, Keel C. Characterization of PhlG, a hydrolase that specifically degrades the antifungal compound 2,4-diacetylphloroglucinol in the biocontrol agent Pseudomonas fluorescens CHA0. Appl Environ Microbiol. 2006;72:418–27.
Yan X, Yang R, Zhao R-X, Han J-T, Jia W-J, Li D-Y, et al. Transcriptional Regulator PhlH Modulates 2,4-Diacetylphloroglucinol Biosynthesis in Response to the Biosynthetic Intermediate and End Product. Appl Environ Microbiol. 2017;83:e01419–17.
Dorrestein PC, Yeh E, Garneau-Tsodikova S, Kelleher NL, Walsh CT. Dichlorination of a pyrrolyl-S-carrier protein by FADH2- dependent halogenase PltA during pyoluteorin biosynthesis. Proc Natl Acad Sci USA. 2005;102:13843–8.
Thomas MG, Burkart MD, Walsh CT. Conversion of L-proline to pyrrolyl-2-carboxyl-S-PCP during undecylprodigiosin and pyoluteorin biosynthesis. Chem Biol. 2002;9:171–84.
Schnider-Keel U, Seematter A, Maurhofer M, Blumer C, Duffy B, Gigot-Bonnefoy C, et al. Autoinduction of 2,4-diacetylphloroglucinol biosynthesis in the biocontrol agent Pseudomonas fluorescens CHA0 and repression by the bacterial metabolites salicylate and pyoluteorin. J Bacteriol. 2000;182:1215–25.
Brodhagen M, Henkels MD, Loper JE. Positive autoregulation and signaling properties of pyoluteorin, an antibiotic produced by the biological control organism Pseudomonas fluorescens Pf-5. Appl Environ Microbiol. 2004;70:1758–66.
Maurhofer M, Baehler E, Notz R, Martinez V, Keel C. Cross Talk between 2,4-Diacetylphloroglucinol-Producing Biocontrol Pseudomonads on Wheat Roots. Appl Environ Microbiol. 2004;70:1990–8.
Clifford JC, Buchanan A, Vining O, Kidarsa TA, Chang JH, McPhail KL, et al. Phloroglucinol functions as an intracellular and intercellular chemical messenger influencing gene expression in Pseudomonas protegens. Environ Microbiol. 2016;18:3296–308.
Kidarsa TA, Goebel NC, Zabriskie TM, Loper JE. Phloroglucinol mediates cross-talk between the pyoluteorin and 2,4-diacetylphloroglucinol biosynthetic pathways in Pseudomonas fluorescens Pf-5. Mol Microbiol. 2011;81:395–414.
Hassan KA, Johnson A, Shaffer BT, Ren Q, Kidarsa TA, Elbourne LDH, et al. Inactivation of the GacA response regulator in Pseudomonas fluorescens Pf-5 has far-reaching transcriptomic consequences. Environ Microbiol. 2010;12:899–915.
Dubuis C, Haas D. Cross-species GacA-controlled induction of antibiosis in pseudomonads. Appl Environ Microbiol. 2007;73:650–4.
Hansen ML, He Z, Wibowo M, Jelsbak L. A Whole-Cell Biosensor for Detection of 2,4- Diacetylphloroglucinol (DAPG)-Producing Bacteria from Grassland Soil. Appl Environ Microbiol. 2021;87:e01400–e01420.
Hesse C, Schulz F, Bull CT, Shaffer BT, Yan Q, Shapiro N, et al. Genome‐based evolutionary history of Pseudomonas spp. Environ Microbiol. 2018;20:2142–59.
Lozano-Andrade CN, Strube ML, Kovács ÁT. Complete genome sequences of four soil-derived isolates for studying synthetic bacterial community assembly. Microbiol Resour Announc. 2021;10:e00848–21.
Le Roux M, Kirkpatrick RL, Montauti EI, Tran BQ, Brook Peterson S, Harding BN, et al. Kin cell lysis is a danger signal that activates antibacterial pathways of Pseudomonas aeruginosa. Elife. 2015;2015:1–65.
Tyc O, van den Berg M, Gerards S, van Veen JA, Raaijmakers JM, de Boer W, et al. Impact of interspecific interactions on antimicrobial activity among soil bacteria. Front Microbiol. 2014;5:1–10.
Qi SS, Bogdanov A, Cnockaert M, Acar T, Ranty-Roby S, Coenye T, et al. Induction of antibiotic specialized metabolism by co-culturing in a collection of phyllosphere bacteria. Environ Microbiol. 2021;23:2132–51.
Cornforth DM, Foster KR. Competition sensing: The social side of bacterial stress responses. Nat Rev Microbiol. 2013;11:285–93.
LeRoux M, Peterson SB, Mougous JD. Bacterial danger sensing. J Mol Biol. 2015;427:3744–53.
Westhoff S, van Wezel GP, Rozen DE. Distance-dependent danger responses in bacteria. Curr Opin Microbiol. 2017;36:95–101.
Davies J, Spiegelman GB, Yim G. The world of subinhibitory antibiotic concentrations. Curr Opin Microbiol. 2006;9:445–53.
Garbeva P, Silby MW, Raaijmakers JM, Levy SB, Boer WDE. Transcriptional and antagonistic responses of Pseudomonas fluorescens Pf0-1 to phylogenetically different bacterial competitors. ISME J. 2011;5:973–85.
Abrudan MI, Smakman F, Grimbergen AJ, Westhoff S, Miller EL, Van Wezel GP, et al. Socially mediated induction and suppression of antibiosis during bacterial coexistence. Proc Natl Acad Sci USA. 2015;112:11054–9.
Kehe J, Ortiz A, Kulesa A, Gore J, Blainey PC, Friedman J. Positive interactions are common among culturable bacteria. Sci Adv. 2021;7:1–10.
Yang KM, Kim JS, Kim HS, Kim YY, Oh JK, Jung HW, et al. Lactobacillus reuteri AN417 cell-free culture supernatant as a novel antibacterial agent targeting oral pathogenic bacteria. Sci Rep. 2021;11:1–16.
Dubern JF, Lugtenberg BJJ, Bloemberg GV. The ppuI-rsaL-ppuR quorum-sensing system regulates biofilm formation of Pseudomonas putida PCL1445 by controlling biosynthesis of the cyclic lipopeptides putisolvins I and II. J Bacteriol. 2006;188:2898–906.
Wellington S, Peter Greenberg E. Quorum sensing signal selectivity and the potential for interspecies cross talk. mBio. 2019;10:e00146–19.
Duffy BK, Défago G. Zinc Improves Biocontrol of Fusarium Crown and Root Rot of Tomato by Pseudomonas fluorescens and Represses the Production of Pathogen Metabolites Inhibitory to Bacterial Antibiotic Biosynthesis. Phytopathology. 1997;87:1250–7.
Li W, Estrada-de los Santos P, Matthijs S, Xie G-L, Busson R, Cornelis P, et al. Promysalin, a Salicylate-Containing Pseudomonas putida Antibiotic, Promotes Surface Colonization and Selectively Targets Other Pseudomonas. Chem Biol. 2011;18:1320–30.
Parnell JJ, Berka R, Young HA, Sturino JM, Kang Y, Barnhart DM, et al. From the lab to the farm: An industrial perspective of plant beneficial microorganisms. Front Plant Sci. 2016;7:1–12.
Berendsen RL, van Verk MC, Stringlis IA, Zamioudis C, Tommassen J, Pieterse CMJ, et al. Unearthing the genomes of plant-beneficial Pseudomonas model strains WCS358, WCS374 and WCS417. BMC Genomics. 2015;16:1–23.
Niu B, Paulson JN, Zheng X, Kolter R. Simplified and representative bacterial community of maize roots. Proc Natl Acad Sci USA. 2017;114:E2450–E2459.
Zhuang L, Li Y, Wang Z, Yu Y, Zhang N, Yang C, et al. Synthetic community with six Pseudomonas strains screened from garlic rhizosphere microbiome promotes plant growth. Micro Biotechnol. 2021;14:488–502.
Zobel S, Benedetti I, Eisenbach L, De Lorenzo V, Wierckx N, Blank LM. Tn7-Based Device for Calibrated Heterologous Gene Expression in Pseudomonas putida. ACS Synth Biol. 2015;4:1341–51.
Van Gestel J, Weissing FJ, Kuipers OP, Kovács ÁT. Density of founder cells affects spatial pattern formation and cooperation in Bacillus subtilis biofilms. ISME J. 2014;8:2069–79.
Schneider CA, Rasband WS, Eliceiri KW. NIH Image to ImageJ: 25 years of image analysis. Nat Methods. 2012;9:671–5.
Hmelo LR, Borlee BR, Almblad H, Love ME, Randall TE, Tseng BS, et al. Precision-engineering the Pseudomonas aeruginosa genome with two-step allelic exchange. Nat Protoc. 2015;10:1820–41.
Yang L, Hengzhuang W, Wu H, Damkiær S, Jochumsen N, Song Z. et al. Polysaccharides serve as scaffold of biofilms formed by mucoid Pseudomonas aeruginosa. FEMS Immunol Med Microbiol. 2012;65:366–76.
Acknowledgements
We thank professor Victor de Lorenzo for providing the SEVA plasmids used in this study. Additionally, we thank Mikael Lenz Strube for aiding with statistical data analysis, as well as Mikkel Anbo and members of the Centre for Microbial Secondary Metabolites (CeMiSt) for general scientific discussions.
Funding
This study was carried out as part of the Center of Excellence for Microbial Secondary Metabolites funded by The Danish National Research Foundation (DNRF137).
Author information
Authors and Affiliations
Contributions
MLH and LJ conceived and designed the study. MLH carried out microbiological, molecular and genetic assays. MW and SAJ carried out metabolite extractions and chemical analyses. MW, SAJ and TOL supervised interpretation of LCMS data. MLH and LJ wrote the manuscript, with contributions and approval of all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hansen, M.L., Wibowo, M., Jarmusch, S.A. et al. Sequential interspecies interactions affect production of antimicrobial secondary metabolites in Pseudomonas protegens DTU9.1. ISME J 16, 2680–2690 (2022). https://doi.org/10.1038/s41396-022-01322-8
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/s41396-022-01322-8
This article is cited by
-
Recent advances in experimental design of synthetic microbial communities for biocontrol application
BioControl (2025)
-
Enrichment of novel entomopathogenic Pseudomonas species enhances willow resistance to leaf beetles
Microbiome (2024)
-
Pyoluteorin-deficient Pseudomonas protegens improves cooperation with Bacillus velezensis, biofilm formation, co-colonizing, and reshapes rhizosphere microbiome
npj Biofilms and Microbiomes (2024)