Abstract
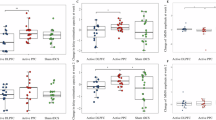
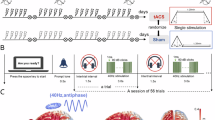
Gamma-band neural oscillations are critically involved in working memory and are disrupted in schizophrenia. Transcranial alternating current stimulation (tACS) at gamma frequency is a promising noninvasive approach to restore oscillatory synchrony and enhance cognition. This randomized, double-blind trial tested whether 40 Hz tACS targeting frontoparietal networks modulates gamma-band activity and connectivity during working memory, and whether these electrophysiological changes relate to cognition in schizophrenia. Patients with schizophrenia (n = 33) were randomized to 10 sessions of active or sham tACS over the left dorsolateral prefrontal cortex (F3) and right parietal cortex (P4), with cognition assessed using standardized neurocognitive measures (MATRICS Consensus Cognitive Battery, MCCB) and an n-back working-memory task. EEG during an n-back task was recorded pre- and post-intervention to assess gamma power, phase-locking value (PLV), and phase-amplitude coupling (PAC). A significant Group × Time interaction indicated that 1-back minus 0-back PLV increased in the active group but not in sham (P = 0.048, Cohen’s d = 1.08). For PAC, a significant interaction showed that delta-high gamma coupling at F3 remained stable in the active group but declined in sham (P = 0.036, Cohen’s d = 1.00). There was no significant correlation with n-back measures of working memory, but an exploratory significant finding linking this modulation to visual learning at 4-week follow-up. No significant group differences were found for MCCB total scores; however, a significant Group × Time interaction emerged for 0-back accuracy during EEG recording (P = 0.029, Cohen’s d = 1.19). These findings demonstrate that 40 Hz tACS can enhance and preserve gamma synchrony in frontoparietal circuits during working memory. The maintained delta-gamma coupling in our exploratory findings on visual learning may suggest a relationship to sustained improvements in cognition over time, but needs additional confirmation.
Similar content being viewed by others
Data availability
The datasets generated and/or analyzed during the current study are not publicly available due to patient privacy and ethical restrictions but are available from the corresponding author upon reasonable request.
Code availability
The analysis code used in this study is not publicly available but can be obtained from the corresponding author upon reasonable request.
References
Wilk CM, Gold JM, McMahon RP, Humber K, Iannone VN, Buchanan RW. No, it is not possible to be schizophrenic yet neuropsychologically normal. Neuropsychology. 2005;19:778–86.
Keefe RSE, Fenton WS. How should DSM-V criteria for schizophrenia include cognitive impairment?. Schizophr Bull. 2007;33:912–20.
Carrión RE, Walder DJ, Auther AM, McLaughlin D, Zyla HO, Adelsheim S, et al. From the psychosis prodrome to the first-episode of psychosis: No evidence of a cognitive decline. J Psychiatr Res. 2018;96:231–8.
Bilder RM, Goldman RS, Robinson D, Reiter G, Bell L, Bates JA, et al. Neuropsychology of first-episode schizophrenia: Initial characterization and clinical correlates. Am J Psychiatry. 2000;157:549–59.
Rodriguez M, Knížková K, Keřková B, Siroňová A, Šustová P, Jonáš J, et al. The relationships between cognitive reserve, cognitive functioning and quality of life in first-episode schizophrenia spectrum disorders. Psychiatry Res. 2022;310:114479.
González-Ortega I, de Los Mozos V, Echeburúa E, Mezo M, Besga A, Ruiz de Azúa S, et al. Working memory as a predictor of negative symptoms and functional outcome in first episode psychosis. Psychiatry Res. 2013;206:8–16.
Mahmood Z, Burton CZ, Vella L, Twamley EW. Neuropsychological predictors of performance-based measures of functional capacity and social skills in individuals with severe mental illness. J Psychiatr Res. 2018;102:201–6.
Cowman M, Holleran L, Lonergan E, O’Connor K, Birchwood M, Donohoe G. Cognitive predictors of social and occupational functioning in early psychosis: A systematic review and meta-analysis of cross-sectional and longitudinal data. Schizophr Bull. 2021;47:1243–53.
Baldez DP, Biazus TB, Rabelo-da-Ponte FD, Nogaro GP, Martins DS, Kunz M, et al. The effect of antipsychotics on the cognitive performance of individuals with psychotic disorders: Network meta-analyses of randomized controlled trials. Neurosci Biobehav Rev. 2021;126:265–75.
Nakayama R, Motoyoshi I. Attention periodically binds visual features as single events depending on neural oscillations phase-locked to action. J Neurosci. 2019;39:4153–61.
Canolty RT, Knight RT. The functional role of cross-frequency coupling. Trends Cogn Sci. 2010;14:506–15.
Esghaei M, Treue S, Vidyasagar TR. Dynamic coupling of oscillatory neural activity and its roles in visual attention. Trends Neurosci. 2022;45:323–35.
Helfrich RF, Knight RT. Oscillatory dynamics of prefrontal cognitive control. Trends Cogn Sci. 2016;20:916–30.
Reinhart RMG, Nguyen JA. Working memory revived in older adults by synchronizing rhythmic brain circuits. Nat Neurosci. 2019;22:820–7.
Fell J, Axmacher N. The role of phase synchronization in memory processes. Nat Rev Neurosci. 2011;12:105–18.
Barr MS, Rajji TK, Zomorrodi R, Radhu N, George TP, Blumberger DM, et al. Impaired theta-gamma coupling during working memory performance in schizophrenia. Schizophr Res. 2017;189:104–10.
Fries P. Rhythms for Cognition: Communication through Coherence. Neuron. 2015;88:220–35.
Salazar RF, Dotson NM, Bressler SL, Gray CM. Content-specific fronto-parietal synchronization during visual working memory. Science. 2012;338:1097–1100.
Kopp F, Schröger E, Lipka S. Synchronized brain activity during rehearsal and short-term memory disruption by irrelevant speech is affected by recall mode. Int J Psychophysiol. 2006;61:188–203.
Griesmayr B, Berger B, Stelzig-Schoeler R, Aichhorn W, Bergmann J, Sauseng P. EEG theta phase coupling during executive control of visual working memory investigated in individuals with schizophrenia and in healthy controls. Cogn Affect Behav Neurosci. 2014;14:1340–55.
Berger B, Minarik T, Griesmayr B, Stelzig-Schoeler R, Aichhorn W, Sauseng P. Brain oscillatory correlates of altered executive functioning in positive and negative symptomatic schizophrenia patients and healthy controls. Front Psychol. 2016;7:705.
Tavakoli AV, Yun K. Transcranial Alternating Current Stimulation (tACS) mechanisms and protocols. Front Cell Neurosci. 2017;11:214.
Schutter DJLG, Wischnewski M. A meta-analytic study of exogenous oscillatory electric potentials in neuroenhancement. Neuropsychologia. 2016;86:110–8.
Vosskuhl J, Strüber D, Herrmann CS. Non-invasive brain stimulation: A paradigm shift in understanding brain oscillations. Front Hum Neurosci. 2018;12:211.
Haller N, Hasan A, Padberg F, Brunelin J, da Costa Lane Valiengo L, Palm U. Gamma transcranial alternating current stimulation in patients with negative symptoms in schizophrenia: A case series. Neurophysiol Clin. 2020;50:301–4.
Haller N, Hasan A, Padberg F, da Costa Lane Valiengo L, Brunelin J, Palm U. Gamma transcranial alternating current stimulation for treatment of negative symptoms in schizophrenia: Report of two cases. Asian J Psychiatr. 2020;54:102423.
Sreeraj VS, Shivakumar V, Sowmya S, Bose A, Nawani H, Narayanaswamy JC, et al. Online theta frequency transcranial alternating current stimulation for cognitive remediation in schizophrenia: A case report and review of literature. J ECT. 2019;35:139–43.
Cao X, Liu Y, Lu Y, Jin H, Sershen H, Davis JM, et al. Effects of transcranial alternating current stimulation on measures of cognition and symptom scores in Chinese patients with schizophrenia. J Psychiatr Res. 2025;183:10–15.
Nuechterlein KH, Green MF, Kern RS, Baade LE, Barch DM, Cohen JD, et al. The MATRICS consensus cognitive battery, part 1: Test selection, reliability, and validity. Am J Psychiatry. 2008;165:203–13.
Shi C, Kang L, Yao S, Ma Y, Li T, Liang Y, et al. The MATRICS Consensus Cognitive Battery (MCCB): Co-norming and standardization in China. Schizophr Res. 2015;169:109–15.
Gronwall DM. Paced auditory serial-addition task: A measure of recovery from concussion. Percept Mot Skills. 1977;44:367–73.
Cohen JD, Forman SD, Braver TS, Casey BJ, Servan-Schreiber D, Noll DC. Activation of the prefrontal cortex in a nonspatial working memory task with functional MRI. Hum Brain Mapp. 1994;1:293–304.
Lachaux JP, Rodriguez E, Martinerie J, Varela FJ. Measuring phase synchrony in brain signals. Hum Brain Mapp. 1999;8:194–208.
di Biase L, Ricci L, Caminiti ML, Pecoraro PM, Carbone SP, Di Lazzaro V. Quantitative high density eeg brain connectivity evaluation in Parkinson’s disease: The Phase Locking Value (PLV). J Clin Med. 2023;12:1450.
Cho RY, Konecky RO, Carter CS. Impairments in frontal cortical gamma synchrony and cognitive control in schizophrenia. Proc Natl Acad Sci USA. 2006;103:19878–83.
Uhlhaas PJ, Singer W. Abnormal neural oscillations and synchrony in schizophrenia. Nat Rev Neurosci. 2010;11:100–13.
Kanai R, Chaieb L, Antal A, Walsh V, Paulus W. Frequency-dependent electrical stimulation of the visual cortex. Curr Biol. 2008;18:1839–43.
Nardone R, Höller Y, Leis S, Höller P, Thon N, Thomschewski A, et al. Invasive and non-invasive brain stimulation for treatment of neuropathic pain in patients with spinal cord injury: a review. J Spinal Cord Med. 2014;37:19–31.
Gevins A, Smith ME, McEvoy L, Yu D. High-resolution EEG mapping of cortical activation related to working memory: effects of task difficulty, type of processing, and practice. Cereb Cortex. 1997;7:374–85.
Engel AK, Fries P. Beta-band oscillations–signalling the status quo?. Curr Opin Neurobiol. 2010;20:156–65.
Luo H, Ye X, Cai H-T, Wang M, Wang Y, Liu Q, et al. Frequency-specific and state-dependent neural responses to brain stimulation. Mol Psychiatry. 2025;30:2880–90.
Sauseng P, Klimesch W, Heise KF, Gruber WR, Holz E, Karim AA, et al. Brain oscillatory substrates of visual short-term memory capacity. Curr Biol. 2009;19:1846–52.
Roux F, Wibral M, Mohr HM, Singer W, Uhlhaas PJ. Gamma-band activity in human prefrontal cortex codes for the number of relevant items maintained in working memory. J Neurosci. 2012;32:12411–20.
Polanía R, Nitsche MA, Korman C, Batsikadze G, Paulus W. The importance of timing in segregated theta phase-coupling for cognitive performance. Curr Biol. 2012;22:1314–8.
Reato D, Rahman A, Bikson M, Parra LC. Effects of weak transcranial alternating current stimulation on brain activity-a review of known mechanisms from animal studies. Front Hum Neurosci. 2013;7:687.
Fries P. A mechanism for cognitive dynamics: Neuronal communication through neuronal coherence. Trends Cogn Sci. 2005;9:474–80.
Sauseng P, Klimesch W, Schabus M, Doppelmayr M. Fronto-parietal EEG coherence in theta and upper alpha reflect central executive functions of working memory. Int J Psychophysiol. 2005;57:97–103.
Palva S, Palva JM. Functional roles of alpha-band phase synchronization in local and large-scale cortical networks. Front Psychol. 2011;2:204.
Zaehle T, Rach S, Herrmann CS. Transcranial alternating current stimulation enhances individual alpha activity in human EEG. PLoS ONE. 2010;5:e13766.
Voytek B, Canolty RT, Shestyuk A, Crone NE, Parvizi J, Knight RT. Shifts in gamma phase-amplitude coupling frequency from theta to alpha over posterior cortex during visual tasks. Front Hum Neurosci. 2010;4:191.
Hyafil A, Giraud A-L, Fontolan L, Gutkin B. Neural cross-frequency coupling: Connecting architectures, mechanisms, and functions. Trends Neurosci. 2015;38:725–40.
Varela F, Lachaux JP, Rodriguez E, Martinerie J. The brainweb: Phase synchronization and large-scale integration. Nat Rev Neurosci. 2001;2:229–39.
Herrmann CS, Rach S, Neuling T, Strüber D. Transcranial alternating current stimulation: a review of the underlying mechanisms and modulation of cognitive processes. Front Hum Neurosci. 2013;7:279.
Cohen JR. The behavioral and cognitive relevance of time-varying, dynamic changes in functional connectivity. Neuroimage. 2018;180:515–25.
Polanía R, Nitsche MA, Ruff CC. Studying and modifying brain function with non-invasive brain stimulation. Nat Neurosci. 2018;21:174–87.
Vossen A, Gross J, Thut G. Alpha Power Increase After Transcranial Alternating Current Stimulation at Alpha Frequency (α-tACS) Reflects Plastic Changes Rather Than Entrainment. Brain Stimul. 2015;8:499–508.
Krause MR, Vieira PG, Csorba BA, Pilly PK, Pack CC. Transcranial alternating current stimulation entrains single-neuron activity in the primate brain. Proc Natl Acad Sci USA. 2019;116:5747–55.
Funding
Funding for this study was provided through several grants. The National Natural Science Foundation of China (82201656, 82101543), the Science and Technology Commission of Shanghai Municipality (19411969400), Shanghai Clinical Research Center for Mental Health (19MC1911100), and the Feixiang Program of Shanghai Mental Health Center (2022-FX-04).
Author information
Authors and Affiliations
Contributions
YL: Formal analysis, investigation, visualization, funding acquisition, writing-original draft, and writing-review and editing. XYC: Investigation, funding acquisition, and writing-review and editing. HJ: Conceptualization, methodology, and writing-review and editing. WL: Data acquisition, and writing-review and editing. FYY: Writing-review and editing. THZ: Methodology, and writing-review and editing. YYT: Methodology, and writing-review and editing. JJW: Methodology, and writing-review and editing. JMD: Conceptualization, methodology, and writing-review and editing. SHH: Writing-review and editing. RCS: Conceptualization, methodology, project administration, and writing-review and editing. CBL: Conceptualization, supervision, methodology, project administration, funding acquisition, and writing-review and editing.
Corresponding authors
Ethics declarations
Competing interests
The authors assert no conflicts of interest related to the work performed in this study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Liu, Y., Cao, X., Jin, H. et al. Effects of 40 Hz transcranial alternating current stimulation on neural synchronization and cognitive correlates in schizophrenia: An EEG study. Transl Psychiatry (2026). https://doi.org/10.1038/s41398-026-03917-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41398-026-03917-7