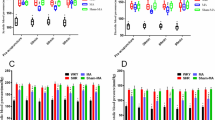
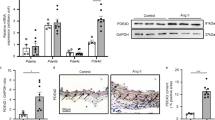
Abstract
Sunitinib is an oral small molecule multitargeted tyrosine kinase inhibitor, which is currently used to treat severe cancers. Clinical research has shown that patients treated with sunitinib develop hypertension. As soon as sunitinib-induced hypertension appears, it is usual to administer anti-hypertension agent. But this treatment may cause acute blood pressure fluctuation which may lead to additional cardiovascular risk. The aim of this study is to establish a mathematical model for managing sunitinib-induced hypertension and blood pressure fluctuation. A mechanism-based PK/PD model was developed based on animal experiments. Then this model was used to perform simulations, thus to propose an anti-hypertension indication, according to which the anti-hypertension treatment might yield relative low-level AUC and fluctuation of blood pressure. The simulation results suggest that the anti-hypertension agent may yield low-level AUC and fluctuation of blood pressure when relative ET-1 level ranges from −15% to 5% and relative NO level is more than 10% compared to control group. Finally, animal experiments were conducted to verify the simulation results. Macitentan (30 mg/kg) was administered based on the above anti-hypertension indication. Compared with the untreated group, the optimized treatment significantly reduced the AUC of blood pressure; meanwhile the fluctuation of blood pressure in optimized treatment group was 70% less than that in immediate treatment group. This work provides a novel model with potential translational value for managing sunitinib-induced hypertension.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Hansson EK, Ma G, Amantea MA, French J, Milligan PA, Friberg LE, et al. PKPD modeling of predictors for adverse effects and overall survival in sunitinib-treated patients with GIST. CPT Pharmacomet Syst Pharm. 2013;2:e85.
Kappers MH, van Esch JH, Sleijfer S, Danser AH, van den Meiracker AH. Cardiovascular and renal toxicity during angiogenesis inhibition: clinical and mechanistic aspects. J Hypertens. 2009;27:2297–309.
Lankhorst S, Kappers MH, van Esch JH, Smedts FM, Sleijfer S, Mathijssen RH, et al. Treatment of hypertension and renal injury induced by the angiogenesis inhibitor sunitinib: preclinical study. Hypertension. 2014;64:1282–9.
Bamias A, Manios E, Karadimou A, Michas F, Lainakis G, Constantinidis C, et al. The use of 24-h ambulatory blood pressure monitoring (ABPM) during the first cycle of sunitinib improves the diagnostic accuracy and management of hypertension in patients with advanced renal cancer. Eur J Cancer. 2011;47:1660–8.
Lankhorst S, Kappers MHW, van Esch JHM, Danser AHJ, van den Meiracker AH. Hypertension during vascular endothelial growth factor inhibition: focus on nitric oxide, endothelin-1, and oxidative stress. Antioxid Redox Signal. 2014;20:135–45.
Grisk O, Koenen A, Meissner T, Donner A, Braun D, Steinbach A, et al. Rho kinase inhibition mitigates sunitinib-induced rise in arterial pressure and renal vascular resistance but not increased renal sodium reabsorption. J Hypertens. 2014;32:2199–210.
Kappers MH, Smedts FM, Horn T, van Esch JH, Sleijfer S, Leijten F, et al. The vascular endothelial growth factor receptor inhibitor sunitinib causes a preeclampsia-like syndrome with activation of the endothelin system. Hypertension. 2011;58:295–302.
Witte J, Mühlbauer M, Braun D, Steinbach A, Golchert J, Rettig R, et al. Renal soluble guanylate cyclase is downregulated in sunitinib-induced hypertension. J Am Heart Assoc. 2018;7:e009557.
Matsui Y, Ishikawa J, Eguchi K, Shibasaki S, Shimada K, Kario K. Maximum value of home blood pressure a novel indicator of target organ damage in hypertension. Hypertension. 2011;57:1087–93.
Parati G, Ochoa JE, Lombardi C, Bilo G. Assessment and management of blood-pressure variability. Nat Rev Cardiol. 2013;10:143.
Hansson EK, Amantea MA, Westwood P, Milligan PA, Houk BE, French J, et al. PKPD modeling of VEGF, sVEGFR-2, sVEGFR-3, and sKIT as predictors of tumor dynamics and overall survival following sunitinib treatment in GIST. CPT Pharmacomet Syst Pharm. 2013;2:e84.
Kappers MH, van Esch JH, Sluiter W, Sleijfer S, Danser AH, van den Meiracker AH. Hypertension induced by the tyrosine kinase inhibitor sunitinib is associated with increased circulating endothelin-1 levels. Hypertension. 2010;56:675–81.
Lankhorst S, Baelde HJ, Kappers MH, Smedts FM, Hansen A, Clahsen-van Groningen MC, et al. Greater sensitivity of blood pressure than renal toxicity to tyrosine kinase receptor inhibition with sunitinib. Hypertension. 2015;66:543–9.
Thijs AM, van Herpen CM, Verweij V, Pertijs J, van den Broek PH, van der Graaf WT, et al. Impaired endothelium-dependent vasodilation does not initiate the development of sunitinib-associated hypertension. J Hypertens. 2015;33:2075–82.
Whitesall SE, Hoff JB, Vollmer AP, D’Alecy LG. Comparison of simultaneous measurement of mouse systolic arterial blood pressure by radiotelemetry and tail-cuff methods. Am J Physiol-Heart Circulatory Physiol. 2004;286:H2408–H15.
Kubota Y, Umegaki K, Kagota S, Tanaka N, Nakamura K, Kunitomo M, et al. Evaluation of blood pressure measured by tail-cuff methods (without heating) in spontaneously hypertensive rats. Biol Pharm Bull. 2006;29::1756–8.
Musijowski J, Piorkowska E, Rudzki PJ. Determination of sunitinib in human plasma using liquid chromatography coupled with mass spectrometry. J Sep Sci. 2014;37:2652–8.
Oberoi RK, Mittapalli RK, Fisher J, Elmquist WF. Sunitinib LC-MS/MS assay in mouse plasma and brain tissue: application in CNS distribution studies. Chromatographia. 2013;76:1657–65.
Moshage H, Kok B, Huizenga JR, Jansen PLM. Nitrite and nitrate determinations in plasma - a critical-evaluation. Clin Chem. 1995;41:892–6.
Speed B, Bu HZ, Pool WF, Peng GW, Wu EY, Patyna S, et al. Pharmacokinetics, distribution, and metabolism of C-14 sunitinib in rats, monkeys, and humans. Drug Metab Disposition. 2012;40:539–55.
Cui W, Zhang ZJ, Hu SQ, Mak SH, Xu DP, Choi CL, et al. Sunitinib produces neuroprotective effect via inhibiting nitric oxide overproduction. CNS Neurosci Ther. 2014;20:244–52.
Eechoute K, van der Veldt AAM, Oosting S, Kappers MHW, Wessels JAM, Gelderblom H, et al. Polymorphisms in endothelial nitric oxide synthase (eNOS) and vascular endothelial growth factor (VEGF) predict sunitinib-induced hypertension. Clin Pharmacol Ther. 2012;92:503–10.
Plato CF, Pollock DM, Garvin JL. Endothelin inhibits thick ascending limb chloride flux via ET(B) receptor-mediated NO release. Am J Physiol-Ren Physiol. 2000;279:F326–F33.
Lee JH, Kim EJ, Kwon JW, Yoo M, Lee MG. Negligible pharmacokinetic interaction between oral DA-8159, a new erectogenic, and amlodipine in rats. Biopharm Drug Dispos. 2006;27:125–31.
Bruderer S, Hopfgartner G, Seiberling M, Wank J, Sidharta PN, Treiber A, et al. Absorption, distribution, metabolism, and excretion of macitentan, a dual endothelin receptor antagonist, in humans. Xenobiotica. 2012;42:901–10.
Mena L, Pintos S, Queipo NV, Aizpurua JA, Maestre G, Sulbaran T. A reliable index for the prognostic significance of blood pressure variability. J Hypertens. 2005;23:505–11.
de Miguel AG, Jimenez-Garcia R, San Martin M, Gonzalez IF, Campo AV, Gonzalez JM, et al. Drug surveillance study of amlodipine in patients with hypertension not controlled with drug therapy: NORCON study. Curr Ther Res-Clin Exp. 2000;61:863–70.
Sidharta PN, van Giersbergen PL, Halabi A, Dingemanse J. Macitentan: entry-into-humans study with a new endothelin receptor antagonist. Eur J Clin Pharmacol. 2011;67:977–84.
Dhaun N, Webb DJ. Receptor tyrosine kinase inhibition, hypertension, and proteinuria: is endothelin the smoking gun? Hypertension. 2010;56:575–7.
Dhaun N, Goddard J, Kohan DE, Pollock DM, Schiffrin EL, Webb DJ. Role of endothelin-1 in clinical hypertension: 20 years on. Hypertension. 2008;52:452–9.
Fishbein L, Cohen DL. Systemic therapies for malignant pheochromocytoma and paraganglioma can exacerbate hypertension. J Clin Hypertens. 2013;15:513–4.
Aparicio-Gallego G, Afonso-Afonso FJ, Leon-Mateos L, Firvida-Perez JL, Vazquez-Estevez S, Lazaro-Quintela M, et al. Molecular basis of hypertension side effects induced by sunitinib. Anticancer Drugs. 2011;22:1–8.
Acknowledgements
This study was supported by the National Natural Science Foundation of China (no. 81773806 and no. 81503145), “Double First-Class” University Project (no. CPU2018GY19) and the Fundamental Research Funds for the Central Universities (no. 2632018ZD10).
Author information
Authors and Affiliations
Contributions
HCL, XTZ, XQL, and HH designed the experiments. HCL, XTZ, and YSZ conducted the animal experiments. XTZ analyzed the samples. HCL developed the mathematic model and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Liu, Hc., Zhou, Xt., Zheng, Ys. et al. PK/PD modeling based on NO-ET homeostasis for improving management of sunitinib-induced hypertension in rats. Acta Pharmacol Sin 41, 719–728 (2020). https://doi.org/10.1038/s41401-019-0331-8
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41401-019-0331-8