Abstract
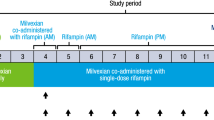
Almonertinib is a novel third-generation EGFR tyrosine kinase inhibitor. It is mainly metabolized by CYP3A in vitro, and N-desmethylated almonertinib (HAS-719) is the major active metabolite in human plasma. In this study, we investigated the effects of CYP3A inhibitor itraconazole and CYP3A inducer rifampicin on the pharmacokinetics of almonertinib and HAS-719 in 64 healthy volunteers. We found that when co-administered with itraconazole, the maximal plasma concentration (Cmax) and the plasma exposure (AUC0–t) of almonertinib were increased by 56.3% and 2.38-fold, respectively, whereas the Cmax and AUC0–t of HAS-719 were reduced by 86.8% and 71.8%, respectively. Co-administration with rifampicin reduced the Cmax and AUC0–t of almonertinib by 79.3% and 92.6%, but the AUC0–t of HAS-719 was unexpectedly decreased by 72.5%. In vitro assays showed that both almonertinib and HAS-719 were substrates of CYP3A and P-gp. Co-administration of rifampicin in Beagle dogs reduced the fecal recovery of almonertinib and HAS-719, and markedly increased the levels of metabolites derived from further metabolism of HAS-719, which was consistent with human plasma data, suggesting that although rifampicin was also a potent inducer of P-gp, the pharmacokinetic alternation of HAS-719 was mainly due to its further metabolism but not excretion changes. Moreover, we revealed that almonertinib was a moderately sensitive substrate of CYP3A in vivo. Special attention should be paid to the interaction between almonertinib and drugs or food affecting CYP3A activity in the clinical application of almonertinib.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Sigismund S, Avanzato D, Lanzetti L. Emerging functions of the EGFR in cancer. Mol Oncol. 2018;12:3–20.
Janne PA, Yang JC, Kim DW, Planchard D, Ohe Y, Ramalingam SS, et al. AZD9291 in EGFR inhibitor-resistant non-small-cell lung cancer. N Engl J Med. 2015;372:1689–99.
Roskoski R Jr. Small molecule inhibitors targeting the EGFR/ErbB family of protein-tyrosine kinases in human cancers. Pharmacol Res. 2019;139:395–411.
Pao W, Chmielecki J. Rational, biologically based treatment of EGFR-mutant non-small-cell lung cancer. Nat Rev Cancer. 2010;10:760–74.
Yun CH, Mengwasser KE, Toms AV, Woo MS, Greulich H, Wong KK, et al. The T790M mutation in EGFR kinase causes drug resistance by increasing the affinity for ATP. Proc Natl Acad Sci U S A. 2008;105:2070–5.
Jackman D, Pao W, Riely GJ, Engelman JA, Kris MG, Janne PA, et al. Clinical definition of acquired resistance to epidermal growth factor receptor tyrosine kinase inhibitors in non-small-cell lung cancer. J Clin Oncol. 2010;28:357–60.
Yu HA, Arcila ME, Rekhtman N, Sima CS, Zakowski MF, Pao W, et al. Analysis of tumor specimens at the time of acquired resistance to EGFR-TKI therapy in 155 patients with EGFR-mutant lung cancers. Clin Cancer Res. 2013;19:2240–7.
Choo JR, Tan CS, Soo RA. Treatment of EGFR T790M-positive non-small cell lung cancer. Target Oncol. 2018;13:141–56.
Han W, Du Y. Recent development of the second and third generation irreversible epidermal growth factor receptor inhibitors. Chem Biodivers. 2017;14: 201600372.
Jiang T, Luo Y, Wang B. Almonertinib-induced interstitial lung disease: a case report. Medicine (Baltimore). 2021;100:e24393.
Yang JC, Camidge DR, Yang CT, Zhou J, Guo R, Chiu CH, et al. Safety, efficacy, and pharmacokinetics of almonertinib (HS-10296) in pretreated patients with EGFR-mutated advanced NSCLC: a multicenter, open-label, phase 1 trial. J Thorac Oncol. 2020;15:1907–18.
Lutz JD, Kirby BJ, Wang L, Song Q, Ling J, Massetto B, et al. Cytochrome P450 3A induction predicts p-glycoprotein induction; part 1: establishing induction relationships using ascending dose rifampin. Clin Pharmacol Ther. 2018;104:1182–90.
Hanke N, Frechen S, Moj D, Britz H, Eissing T, Wendl T, et al. PBPK models for CYP3A4 and P-gp DDI prediction: a modeling network of rifampicin, itraconazole, clarithromycin, midazolam, alfentanil, and digoxin. CPT Pharmacomet Syst Pharmacol. 2018;7:647–59.
Benet LZ. The drug transporter-metabolism alliance: uncovering and defining the interplay. Mol Pharmacol. 2009;6:1631–43.
Benet LZ, Cummins CL, Wu CY. Transporter-enzyme interactions: implications for predicting drug-drug interactions from in vitro data. Curr Drug Metab. 2003;4:393–8.
Masters JC, Shah MM, Feist AA. Drug interaction between sirolimus and ranolazine in a kidney transplant patient. Case Rep Transpl. 2014;2014:548243.
Hylton AC, Ezekiel TO. Rhabdomyolysis in a patient receiving ranolazine and simvastatin. Am J Health Syst Pharm. 2010;67:1829–31.
Cummins CL, Jacobsen W, Christians U, Benet LZ. CYP3A4-transfected Caco-2 cells as a tool for understanding biochemical absorption barriers: studies with sirolimus and midazolam. J Pharmacol Exp Ther. 2004;308:143–55.
Chen J, Tran C, Xiao L, Palamanda J, Klapmuts T, Kumari P, et al. Co-induction of CYP3A12 and 3A26 in dog liver slices by xenobiotics: species difference between human and dog CYP3A induction. Drug Metab Lett. 2009;3:61–6.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Grant 82073924). We thank clinical staff of Ji-nan Central Hospital for their participating in the discussion of the clinical protocol, and being responsible for the management of the subjects and the collection of plasma samples. We are grateful for the contributions of the study sponsor, Hansoh Pharmaceutical Group Co. Ltd.
Author information
Authors and Affiliations
Contributions
LL and XYC participated in research design: LL, ZTG, NJX, HX, LY and WL conducted experiments: LL and XYC wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Liu, L., Li, W., Yang, L. et al. Itraconazole and rifampicin, as CYP3A modulators but not P-gp modulators, affect the pharmacokinetics of almonertinib and active metabolite HAS-719 in healthy volunteers. Acta Pharmacol Sin 43, 1082–1090 (2022). https://doi.org/10.1038/s41401-021-00710-8
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41401-021-00710-8
Keywords
This article is cited by
-
Almonertinib and alflutinib show novel inhibition on rare EGFR S768I mutant cells
Clinical and Translational Oncology (2024)