Abstract
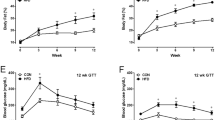
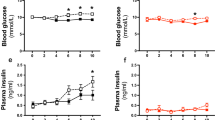
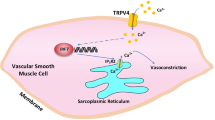
Transient receptor potential channel TRPV4 and nicotinamide adenine dinucleotide phosphate oxidase (Nox2) are involved in oxidative stress that increases endothelial permeability. It has been shown that obesity enhances the physical association of TRPV4 and Nox2, but the role of TRPV4–Nox2 association in obesity has not been clarified. In this study we investigated the function of TRPV4–Nox2 complex in reducing oxidative stress and regulating abnormal vascular permeability in obesity. Obesity was induced in mice by feeding a high-fat diet (HFD) for 14 weeks. The physical interaction between TRPV4 and Nox2 was measured using FRET, co-immunoprecipitation and GST pull-down assays. The functional interaction was measured by rhodamine phalloidin, CM-H2DCFDA in vitro, the fluorescent dye dihydroethidium (DHE) staining assay, and the Evans blue permeability assay in vivo. We demonstrated that TRPV4 physically and functionally associated with Nox2, and this physical association was enhanced in aorta of obese mice. Furthermore, we showed that interrupting TRPV4–Nox2 coupling by TRPV4 knockout, or by treatment with a specific Nox2 inhibitor Nox2 dstat or a specific TRPV4 inhibitor HC067046 significantly attenuated obesity-induced ROS overproduction in aortic endothelial cells, and reversed the abnormal endothelial cytoskeletal structure. In order to discover small molecules disrupting the over-coupling of TPRV4 and Nox2 in obesity, we performed molecular docking analysis and found that compound M12 modulated TRPV4–Nox2 association, reduced ROS production, and finally reversed disruption of the vascular barrier in obesity. Together, this study, for the first time, provides evidence for the TRPV4 physically interacting with Nox2. TRPV4–Nox2 complex is a potential drug target in improving oxidative stress and disruption of the vascular barrier in obesity. Compound M12 targeting TRPV4–Nox2 complex can improve vascular barrier function in obesity.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Mundi S, Massaro M, Scoditti E, Carluccio MA, van Hinsbergh VWM, Iruela-Arispe ML, et al. Endothelial permeability, LDL deposition, and cardiovascular risk factors. Cardiovasc Res. 2018;114:35–52.
Ohara Y, Peterson TE, Harrison DG. Hypercholesterolemia increases endothelial superoxide anion production. J Clin Invest. 1993;91:2546–51.
Xu L, Nagata N, Ota T. Glucoraphanin: a broccoli sprout extract that ameliorates obesity-induced inflammation and insulin resistance. Adipocyte. 2018;7:218–25.
Filosa JA, Yao X, Rath G. TRPV4 and the regulation of vascular tone. J Cardiovasc Pharmacol. 2013;61:113–9.
Wu QF, Qian C, Zhao N, Dong Q, Li J, Wang BB, et al. Activation of transient receptor potential vanilloid 4 involves in hypoxia/reoxygenation injury in cardiomyocytes. Cell Death Dis. 2017;8:e2828.
Hamanaka K, Jian MY, Townsley MI, King JA, Liedtke W, Weber DS, et al. TRPV4 channels augment macrophage activation and ventilator-induced lung injury. Am J Physiol Lung Cell Mol Physiol. 2010;299:L353–62.
Bubolz AH, Mendoza SA, Zheng X, Zinkevich NS, Li R, Gutterman DD, et al. Activation of endothelial TRPV4 channels mediates flow-induced dilation in human coronary arterioles: role of Ca2+ entry and mitochondrial ROS signaling. Am J Physiol Heart Circ Physiol. 2012;302:H634–42.
Burgoyne JR, Mongue-Din H, Eaton P, Shah AM. Redox signaling in cardiac physiology and pathology. Circ Res. 2012;111:1091–106.
Song Q, Zhang Y. Application of high-fat cell model in steady-state regulation of vascular function. Saudi J Biol Sci. 2019;26:2132–5.
Gao M, Han J, Zhu Y, Tang C, Liu L, Xiao W, et al. Blocking endothelial TRPV4-Nox2 interaction helps reduce ROS production and inflammation, and improves vascular function in obese mice. J Mol Cell Cardiol. 2021;157:66–76.
Suzuki M, Mizuno A, Kodaira K, Imai M. Impaired pressure sensation in mice lacking TRPV4. J Biol Chem. 2003;278:22664–8.
Meoli L, Isensee J, Zazzu V, Nabzdyk CS, Soewarto D, Witt H, et al. Sex- and age-dependent effects of Gpr30 genetic deletion on the metabolic and cardiovascular profiles of diet-induced obese mice. Gene. 2014;540:210–6.
Ma X, Du J, Zhang P, Deng J, Liu J, Lam FF, et al. Functional role of TRPV4-KCa2.3 signaling in vascular endothelial cells in normal and streptozotocin-induced diabetic rats. Hypertension. 2013;62:134–9.
Adebiyi A, Zhao G, Narayanan D, Thomas-Gatewood CM, Bannister JP, Jaggar JH. Isoform-selective physical coupling of TRPC3 channels to IP3 receptors in smooth muscle cells regulates arterial contractility. Circ Res. 2010;106:1603–12.
Kropf DL, Berge SK, Quatrano RS. Actin localization during fucus embryogenesis. Plant Cell. 1989;1:191–200.
Lee DH, Park JS, Lee YS, Han J, Lee DK, Kwon SW, et al. SQSTM1/p62 activates NFE2L2/NRF2 via ULK1-mediated autophagic KEAP1 degradation and protects mouse liver from lipotoxicity. Autophagy. 2020;16:1949–73.
Cao C, Edwards A, Sendeski M, Lee-Kwon W, Cui L, Cai CY, et al. Intrinsic nitric oxide and superoxide production regulates descending vasa recta contraction. Am J Physiol Ren Physiol. 2010;299:F1056–64.
Shao J, Han J, Zhu Y, Mao A, Wang Z, Zhang K, et al. Curcumin induces endothelium-dependent relaxation by activating endothelial TRPV4 channels. J Cardiovasc Transl Res. 2019;12:600–7.
Salazar G, Cullen A, Huang J, Zhao Y, Serino A, Hilenski L, et al. SQSTM1/p62 and PPARGC1A/PGC-1alpha at the interface of autophagy and vascular senescence. Autophagy. 2020;16:1092–110.
Choi N, Kim WS, Oh SH, Sung JH. Epiregulin promotes hair growth via EGFR-medicated epidermal and ErbB4-mediated dermal stimulation. Cell Prolif. 2020;53:e12881.
Thengchaisri N, Hein TW, Ren Y, Kuo L. Endothelin-1 impairs coronary arteriolar dilation: Role of p38 kinase-mediated superoxide production from NADPH oxidase. J Mol Cell Cardiol. 2015;86:75–84.
Zhou Z, Rajamani U, Labazi H, Tilley SL, Ledent C, Teng B, et al. Involvement of NADPH oxidase in A2A adenosine receptor-mediated increase in coronary flow in isolated mouse hearts. Purinergic Signal. 2015;11:263–73.
El-Awady MS, Rajamani U, Teng B, Tilley SL, Mustafa SJ. Evidence for the involvement of NADPH oxidase in adenosine receptors-mediated control of coronary flow using A1 and A3 knockout mice. Physiol Rep. 2013;1:e00070.
Chen S, Jiang H, Wu X, Fang J. Therapeutic effects of quercetin on inflammation, obesity, and type 2 diabetes. Mediators Inflamm. 2016;2016:9340637.
Vieira AA, Michels M, Florentino D, Nascimento DZ, Rezin GT, Leffa DD, et al. Obesity promotes oxidative stress and exacerbates sepsis-induced brain damage. Curr Neurovasc Res. 2015;12:147–54.
Timmerman I, Heemskerk N, Kroon J, Schaefer A, van Rijssel J, Hoogenboezem M, et al. A local VE-cadherin and Trio-based signaling complex stabilizes endothelial junctions through Rac1. J Cell Sci. 2015;128:3514.
Barry AK, Wang N, Leckband DE. Local VE-cadherin mechanotransduction triggers long-ranged remodeling of endothelial monolayers. J Cell Sci. 2015;128:1341–51.
Barry AK, Tabdili H, Muhamed I, Wu J, Shashikanth N, Gomez GA, et al. Alpha-catenin cytomechanics-role in cadherin-dependent adhesion and mechanotransduction. J Cell Sci. 2014;127:1779–91.
Giannotta M, Trani M, Dejana E. VE-cadherin and endothelial adherens junctions: active guardians of vascular integrity. Dev Cell. 2013;26:441–54.
Yan M, Zhang X, Chen A, Gu W, Liu J, Ren X, et al. Endothelial cell SHP-2 negatively regulates neutrophil adhesion and promotes transmigration by enhancing ICAM-1-VE-cadherin interaction. FASEB J. 2017;31:4759–69.
Domazetovic V, Bonanomi AG, Stio M, Vincenzini MT, Iantomasi T. Resveratrol decreases TNFalpha-induced ICAM-1 expression and release by Sirt-1-independent mechanism in intestinal myofibroblasts. Exp Cell Res. 2019;382:111479.
Bodiga VL, Kudle MR, Bodiga S. Silencing of PKC-alpha, TRPC1 or NF-kappaB expression attenuates cisplatin-induced ICAM-1 expression and endothelial dysfunction. Biochem Pharmacol. 2015;98:78–91.
Bodiga VL, Inapurapu SP, Vemuri PK, Kudle MR, Bodiga S. Intracellular zinc status influences cisplatin-induced endothelial permeability through modulation of PKCalpha, NF-kappaB and ICAM-1 expression. Eur J Pharmacol. 2016;791:355–68.
Vitoria WO, Thome LS, Kanashiro-Galo L, Carvalho LV, Penny R, Santos WLC, et al. Upregulation of intercellular adhesion molecule-1 and vascular cell adhesion molecule-1 in renal tissue in severe dengue in humans: Effects on endothelial activation/dysfunction. Rev Soc Bras Med Trop. 2019;52:e20180353.
Kim Y, Lee S, Zhang H, Lee S, Kim H, Kim Y, et al. CLEC14A deficiency exacerbates neuronal loss by increasing blood-brain barrier permeability and inflammation. J Neuroinflammation. 2020;17:48.
Shi Y, Xiong Y, Lei Y, Li Z, Yan H, Yuan J, et al. Protective effect of COMP-angiopoietin-1 on peritoneal vascular permeability and peritoneal transport function in uremic peritoneal dialysis rats. Am J Transl Res. 2019;11:5932–43.
Chen H, Tong X, Lang L, Jacobson O, Yung BC, Yang X, et al. Quantification of tumor vascular permeability and blood volume by positron emission tomography. Theranostics. 2017;7:2363–76.
Zhao H, Zhu Y, Zhang J, Wu Y, Xiang X, Zhang Z, et al. The beneficial effect of hes on vascular permeability and its relationship with endothelial glycocalyx and intercellular junction after hemorrhagic shock. Front Pharmacol. 2020;11:597.
Jeong J, Lee J, Lim J, Cho S, An S, Lee M, et al. Soluble RAGE attenuates AngII-induced endothelial hyperpermeability by disrupting HMGB1-mediated crosstalk between AT1R and RAGE. Exp Mol Med. 2019;51:1–15.
Lundeberg E, Van Der Does AM, Kenne E, Soehnlein O, Lindbom L. Assessing large-vessel endothelial permeability using near-infrared fluorescence imaging. Arterioscler Thromb Vasc Biol. 2015;35:783–6.
Suresh K, Servinsky L, Jiang H, Bigham Z, Yun X, Kliment C, et al. Reactive oxygen species induced Ca2+ influx via TRPV4 and microvascular endothelial dysfunction in the SU5416/hypoxia model of pulmonary arterial hypertension. Am J Physiol Lung Cell Mol Physiol. 2018;314:L893–907.
Lyons JS, Joca HC, Law RA, Williams KM, Kerr JP, Shi G, et al. Microtubules tune mechanotransduction through NOX2 and TRPV4 to decrease sclerostin abundance in osteocytes. Sci Signal. 2017;10:506.
Wu Q, Lu K, Zhao Z, Wang B, Liu H, Zhang S, et al. Blockade of transient receptor potential vanilloid 4 enhances antioxidation after myocardial ischemia/reperfusion. Oxid Med Cell Longev. 2019;2019:7283683.
Hong Z, Tian Y, Yuan Y, Qi M, Li Y, Du Y, et al. Enhanced oxidative stress is responsible for TRPV4-induced neurotoxicity. Front Cell Neurosci. 2016;10:232.
Suresh K, Servinsky L, Reyes J, Baksh S, Undem C, Caterina M, et al. Hydrogen peroxide-induced calcium influx in lung microvascular endothelial cells involves TRPV4. Am J Physiol Lung Cell Mol Physiol. 2015;309:L1467–77.
Naziroglu M, Oz A, Yildizhan K. Selenium and neurological diseases: focus on peripheral pain and TRP channels. Curr Neuropharmacol. 2020;18:501–17.
Martner A, Aydin E, Hellstrand K. NOX2 in autoimmunity, tumor growth and metastasis. J Pathol. 2019;247:151–4.
Kroller Schon S, Daiber A, Steven S, Oelze M, Frenis K, Kalinovic S, et al. Crucial role for Nox2 and sleep deprivation in aircraft noise-induced vascular and cerebral oxidative stress, inflammation, and gene regulation. Eur Heart J. 2018;39:3528–39.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (82025005, 81622007, 81700437, 81870362, 81960662, 82000291, and 91939301), the Chang Jiang Scholars Program (Q2015106), Fundamental Research Funds for the Central Universities (JUSRP51704A), and the National First-Class Discipline Program of Food Science and Technology (JUFSTR20180101). We thank Prof. Iain C. Bruce (University of Hong Kong, China) for his guidance on paper writing.
Author information
Authors and Affiliations
Contributions
MRG and XM designed the research; JH, PZ, HK, XPH, and HJL performed the research; CLT contributed new reagents or analytic tools; MRG and YFZ analyzed the data and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
XM, CLT, MRG, and JH have applied to the State Intellectual Property Office of China for the patent “A compound for reducing TRPV4–Nox2 excessive interaction and its application in anti-abnormal vascular permeability” (2020114600923).
Supplementary information
Rights and permissions
About this article
Cite this article
Gao, Mr., Zhang, P., Han, J. et al. Small molecule compound M12 reduces vascular permeability in obese mice via blocking endothelial TRPV4–Nox2 interaction. Acta Pharmacol Sin 43, 1430–1440 (2022). https://doi.org/10.1038/s41401-021-00780-8
Received:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/s41401-021-00780-8