Abstract
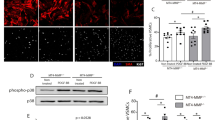
Platelet-derived growth factor (PDGF-BB) released from the injured intima induces the proliferation and migration of vascular smooth muscle cells (VSMCs), which is the key mechanism of neointimal hyperplasia. Zinc finger 36 (ZFP36), a widespread RNA-binding protein, is important for pathological processes in many diseases. In this study we investigated the role of ZFP36 in VSMCs proliferation, migration and neointimal hyperplasia in mice. We generated smooth muscle-specific Zfp36 knockout (Zfp36SMKO) mice, and established restenosis mouse models by ligation of left carotid artery in Zfp36SMKO mice. We showed that the expression levels of ZFP36 were significantly decreased in human atherosclerotic coronary arteries and murine injured carotid arteries compared with controls. Compared to control Zfp36fl/fl mice, Zfp36SMKO mice displayed accelerated neointimal hyperplasia. In cultured mouse VSMCs, PDGF-BB (20 ng/mL) significantly downregulated ZFP36 expression through KLF4 binding site in Zfp36 promoter. We revealed that ZFP36 could bind to the mRNA of cell migration-inducing protein (CEMIP) and promoted its degradation in VSMCs, thereby reducing the expression of CEMIP protein. Knockdown of Cemip inhibited VSMCs proliferation and migration induced by Zfp36 knockout, thereby suppressing neointimal hyperplasia in Zfp36SMKO mice. We conclude that vascular smooth muscle ZFP36 has a protective effect against neointimal hyperplasia by reducing CEMIP expression. ZFP36 is downregulated by vascular injury and PDGF-BB treatment, which promotes VSMCs proliferation and migration and neointima formation. The results suggest that targeting ZFP36 may represent a novel therapeutic strategy for preventing or treating neointimal hyperplasia and related cardiovascular diseases.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others

Data availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request. The RNA-seq datasets have been deposited in the NCBI Gene Expression Omnibus and are accessible through the accession number GSE286906.
References
Glagov S. Intimal hyperplasia, vascular modeling, and the restenosis problem. Circulation. 1994;89:2888–91.
Sedding DG, Boyle EC, Demandt JAF, Sluimer JC, Dutzmann J, Haverich A, et al. Vasa vasorum angiogenesis: key player in the initiation and progression of atherosclerosis and potential target for the treatment of cardiovascular disease. Front Immunol. 2018;9:706.
Melnik T, Jordan O, Corpataux JM, Delie F, Saucy F. Pharmacological prevention of intimal hyperplasia: A state-of-the-art review. Pharmacol Ther. 2022;235:108157.
Jaminon A, Reesink K, Kroon A, Schurgers L. The role of vascular smooth muscle cells in arterial remodeling: focus on calcification-related processes. Int J Mol Sci. 2019;20:5694.
Marmur JD, Poon M, Rossikhina M, MB Taubman. Induction of PDGF-responsive genes in vascular smooth muscle. Implications for the early response to vessel injury. Circulation. 1992;86:III53–60.
Pidkovka NA, Cherepanova OA, Yoshida T, Alexander MR, Deaton RA, Thomas JA, et al. Oxidized phospholipids induce phenotypic switching of vascular smooth muscle cells in vivo and in vitro. Circ Res. 2007;101:792–801.
Ferns GA, Raines EW, Sprugel KH, Motani AS, Reidy MA, Ross R. Inhibition of neointimal smooth muscle accumulation after angioplasty by an antibody to PDGF. Science. 1991;253:1129–32.
Tanizawa S, Ueda M, van der Loos CM, van der Wal AC, Becker AE. Expression of platelet derived growth factor B chain and beta receptor in human coronary arteries after percutaneous transluminal coronary angioplasty: an immunohistochemical study. Heart. 1996;75:549–56.
Liu Y, Sinha S, McDonald OG, Shang Y, Hoofnagle MH, Owens GK. Kruppel-like factor 4 abrogates myocardin-induced activation of smooth muscle gene expression. J Biol Chem. 2005;280:9719–27.
Yoshida T, Kaestner KH, Owens GK. Conditional deletion of Kruppel-like factor 4 delays downregulation of smooth muscle cell differentiation markers but accelerates neointimal formation following vascular injury. Circ Res. 2008;102:1548–57.
Xue Q, Wang X, Deng X, Huang Y, Tian W. CEMIP regulates the proliferation and migration of vascular smooth muscle cells in atherosclerosis through the WNT-beta-catenin signaling pathway. Biochem Cell Biol. 2020;98:249–57.
Rodriguez-Gomez G, Paredes-Villa A, Cervantes-Badillo MG, Gomez-Sonora JP, Jorge-Perez JH, Cervantes-Roldan R, et al. Tristetraprolin: A cytosolic regulator of mRNA turnover moonlighting as transcriptional corepressor of gene expression. Mol Genet Metab. 2021;133:137–47.
Makita S, Takatori H, Nakajima H. Post-transcriptional regulation of immune responses and inflammatory diseases by RNA-binding ZFP36 family proteins. Front Immunol. 2021;12:711633.
Saini Y, Chen J, Patial S. The tristetraprolin family of RNA-binding proteins in cancer: progress and future prospects. Cancers (Basel). 2020;12:1539.
Snyder BL, Blackshear PJ. Clinical implications of tristetraprolin (TTP) modulation in the treatment of inflammatory diseases. Pharmacol Ther. 2022;239:108198.
Ji E, Hu S, Lu Q, Zhang M, Jiang M. Hydrogen peroxide positively regulates ABA signaling via oxidative modification of the C2H2-type zinc finger protein ZFP36 in rice. Plant Physiol Biochem. 2024;213:108844.
Taylor GA, Carballo E, Lee DM, Lai WS, Thompson MJ, Patel DD, et al. A pathogenetic role for TNF alpha in the syndrome of cachexia, arthritis, and autoimmunity resulting from tristetraprolin (TTP) deficiency. Immunity. 1996;4:445–54.
Carballo E, Blackshear PJ. Roles of tumor necrosis factor-alpha receptor subtypes in the pathogenesis of the tristetraprolin-deficiency syndrome. Blood. 2001;98:2389–95.
Park J, Rah SY, An HS, Lee JY, Roh GS, Ryter SW, et al. Metformin-induced TTP mediates communication between Kupffer cells and hepatocytes to alleviate hepatic steatosis by regulating lipophagy and necroptosis. Metabolism. 2023;141:155516.
Saaoud F, Wang J, Iwanowycz S, Wang Y, Altomare D, Shao Y, et al. Bone marrow deficiency of mRNA decaying protein tristetraprolin increases inflammation and mitochondrial ROS but reduces hepatic lipoprotein production in LDLR knockout mice. Redox Biol. 2020;37:101609.
Kafasla P, Skliris A, Kontoyiannis DL. Post-transcriptional coordination of immunological responses by RNA-binding proteins. Nat Immunol. 2014;15:492–502.
Lai WS, Carballo E, Strum JR, Kennington EA, Phillips RS, Blackshear PJ. Evidence that tristetraprolin binds to AU-rich elements and promotes the deadenylation and destabilization of tumor necrosis factor alpha mRNA. Mol Cell Biol. 1999;19:4311–23.
Zhao W, Liu M, D’Silva NJ, Kirkwood KL. Tristetraprolin regulates interleukin-6 expression through p38 MAPK-dependent affinity changes with mRNA 3’ untranslated region. J Interferon Cytokine Res. 2011;31:629–37.
Cicchetto AC, Jacobson EC, Sunshine H, Wilde BR, Krall AS, Jarrett KE, et al. ZFP36-mediated mRNA decay regulates metabolism. Cell Rep. 2023;42:112411.
Kim DJ, Vo MT, Choi SH, Lee JH, Jeong SY, Hong CH, et al. Tristetraprolin-mediated hexokinase 2 expression regulation contributes to glycolysis in cancer cells. Mol Biol Cell. 2019;30:542–53.
Jang JH, Kim DJ, Ham SY, Vo MT, Jeong SY, Choi SH, et al. Tristetraprolin posttranscriptionally downregulates PFKFB3 in cancer cells. Biochem Biophys Res Commun. 2020;521:389–94.
Liu J, Yan W, Han P, Tian D. The emerging role of KIAA1199 in cancer development and therapy. Biomed Pharmacother. 2021;138:111507.
Zhao L, Zhang D, Shen Q, Jin M, Lin Z, Ma H, et al. KIAA1199 promotes metastasis of colorectal cancer cells via microtubule destabilization regulated by a PP2A/stathmin pathway. Oncogene. 2019;38:935–49.
Zhang D, Zhao L, Shen Q, Lv Q, Jin M, Ma H, et al. Down-regulation of KIAA1199/CEMIP by miR-216a suppresses tumor invasion and metastasis in colorectal cancer. Int J Cancer. 2017;140:2298–309.
Wang H, Zhang B, Li R, Chen J, Xu G, Zhu Y, et al. KIAA1199 drives immune suppression to promote colorectal cancer liver metastasis by modulating neutrophil infiltration. Hepatology. 2022;76:967–81.
Xu Y, Xu H, Li M, Wu H, Guo Y, Chen J, et al. KIAA1199 promotes sorafenib tolerance and the metastasis of hepatocellular carcinoma by activating the EGF/EGFR-dependent epithelial-mesenchymal transition program. Cancer Lett. 2019;454:78–89.
Ohtsuki T, Hatipoglu OF, Asano K, Inagaki J, Nishida K, Hirohata S. Induction of CEMIP in chondrocytes by inflammatory cytokines: underlying mechanisms and potential involvement in osteoarthritis. Int J Mol Sci. 2020;21:3140.
Xie G, Dong P, Chen H, Xu L, Liu Y, Ma Y, et al. Decreased expression of ATF3, orchestrated by beta-catenin/TCF3, miR-17-5p and HOXA11-AS, promoted gastric cancer progression via increased beta-catenin and CEMIP. Exp Mol Med. 2021;53:1706–22.
Yu Y, Liu B, Li X, Lu D, Yang L, Chen L, et al. ATF4/CEMIP/PKCalpha promotes anoikis resistance by enhancing protective autophagy in prostate cancer cells. Cell Death Dis. 2022;13:46.
Cloud AS, Vargheese AM, Gunewardena S, Shimak RM, Ganeshkumar S, Kumaraswamy E, et al. Loss of REST in breast cancer promotes tumor progression through estrogen sensitization, MMP24 and CEMIP overexpression. BMC Cancer. 2022;22:180.
Hsieh IY, He J, Wang L, Lin B, Liang Z, Lu B, et al. H3K27me3 loss plays a vital role in CEMIP mediated carcinogenesis and progression of breast cancer with poor prognosis. Biomed Pharmacother. 2020;123:109728.
Evensen NA, Li Y, Kuscu C, Liu J, Cathcart J, Banach A, et al. Hypoxia promotes colon cancer dissemination through up-regulation of cell migration-inducing protein (CEMIP). Oncotarget. 2015;6:20723–39.
Newby AC, Zaltsman AB. Molecular mechanisms in intimal hyperplasia. J Pathol. 2000;190:300–9.
Ji Z, Li J, Wang J. Jujuboside B inhibits neointimal hyperplasia and prevents vascular smooth muscle cell dedifferentiation, proliferation, and migration via activation of AMPK/PPAR-gamma signaling. Front Pharmacol. 2021;12:672150.
Huang L, Zhang SM, Zhang P, Zhang XJ, Zhu LH, Chen K, et al. Interferon regulatory factor 7 protects against vascular smooth muscle cell proliferation and neointima formation. J Am Heart Assoc. 2014;3:e001309.
Jain M, Dhanesha N, Doddapattar P, Chorawala MR, Nayak MK, Cornelissen A, et al. Smooth muscle cell-specific fibronectin-EDA mediates phenotypic switching and neointimal hyperplasia. J Clin Invest. 2020;130:295–314.
Huang F, Zhang F, Huang L, Zhu XB, Huang C, Li N, et al. Inositol 1,4,5-trisphosphate receptors regulate vascular smooth muscle cell proliferation and neointima formation in mice. J Am Heart Assoc. 2024;13:e034203.
Acknowledgements
This study was supported by grants from the National Natural Science Foundation of China (No. 82470499, 82270457, 82300486), the Natural Science Foundation of Shandong Province (ZR2023QH398, ZR2022QH288, ZR2024ZD23), the Taishan Scholar Project of Shandong Province of China (No. tstp20240852), and the Postdoctoral Innovation Project of Shandong Province (SDCX-ZG-202400016).
Author information
Authors and Affiliations
Contributions
YL and WCZ developed and designed the study; LW, LFH, XX, ZNW, MT and YLZ developed the methodology and wrote, reviewed and revised the paper; HXL and XPJ acquired, analyzed, interpreted, and statistically analyzed the data; and GQC and PK provided technical and material support. All the authors read and approved the final paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, L., He, Lf., Xiong, X. et al. Deletion of smooth muscle ZFP36 promotes neointimal hyperplasia in mice. Acta Pharmacol Sin 46, 1317–1328 (2025). https://doi.org/10.1038/s41401-024-01473-8
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41401-024-01473-8