Abstract
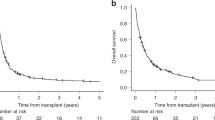
Myeloid sarcoma (MS) is a rare hematological neoplasm with poor prognosis, posing a significant clinical challenge due to the absence of effective and standardized treatments. We conducted a retrospective analysis of 162 MS patients treated at 12 centers to compare outcomes between intensive chemotherapy and allogeneic hematopoietic stem cell transplantation (allo-HSCT). Our analysis revealed that allo-HSCT demonstrated superior overall survival (OS) within the initial 36 months compared to intensive chemotherapy alone (p = 0.037). However, beyond 36 months (36–60 months), a reverse trend was observed (p = 0.056). Subgroup analysis revealed potential benefit for isolated MS patients with allo-HSCT, but not for those with leukemic MS. Additionally, in patients achieving first complete remission (CR1) after induction chemotherapy, allo-HSCT did not significantly improve 5-year OS compared with intensive chemotherapy alone (p = 0.25). Conversely, allo-HSCT significantly improved 5-year OS in non-CR1 patients (p < 0.001). Notably, HLA-matched HSCT and haploidentical HSCT showed comparable outcomes in terms of OS, disease-free survival, and cumulative incidence of relapse. In conclusion, allo-HSCT improved outcomes for MS patients within 36 months of disease onset, and haploidentical HSCT emerged as a viable treatment option for patients without matched donors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that support the findings of this study are available on request from the corresponding author, Xiao-Hui Zhang (zhangxh@bjmu.edu.cn). The data are not publicly available due to their containing information that could compromise the privacy of research participants.
References
Khoury JD, Solary E, Abla O, Akkari Y, Alaggio R, Apperley JF, et al. The 5th edition of the World Health Organization classification of haematolymphoid tumours: myeloid and histiocytic/dendritic neoplasms. Leukemia. 2022;36:1703–19.
Bakst RL, Tallman MS, Douer D, Yahalom J. How I treat extramedullary acute myeloid leukemia. Blood. 2011;118:3785–93.
Magdy M, Abdel Karim N, Eldessouki I, Gaber O, Rahouma M, Ghareeb M. Myeloid sarcoma. Oncol Res Treat. 2019;42:224–9.
Yilmaz AF, Saydam G, Sahin F, Baran Y. Granulocytic sarcoma: a systematic review. Am J Blood Res. 2013;3:265–70.
Chevallier P, Labopin M, Cornelissen J, Socie G, Rocha V, Mohty M, et al. Allogeneic hematopoietic stem cell transplantation for isolated and leukemic myeloid sarcoma in adults: a report from the Acute Leukemia Working Party of the European Group for Blood and Marrow Transplantation. Haematologica. 2011;96:1391–4.
Chevallier P, Mohty M, Lioure B, Michel G, Contentin N, Deconinck E, et al. Allogeneic hematopoietic stem-cell transplantation for myeloid sarcoma: a retrospective study from the SFGM-TC. J Clin Oncol. 2008;26:4940–3.
Lazzarotto D, Candoni A, Filì C, Forghieri F, Pagano L, Busca A, et al. Clinical outcome of myeloid sarcoma in adult patients and effect of allogeneic stem cell transplantation. Results from a multicenter survey. Leuk Res. 2017;53:74–81.
Shimizu H, Saitoh T, Tanaka M, Mori T, Sakura T, Kawai N, et al. Allogeneic hematopoietic stem cell transplantation for adult AML patients with granulocytic sarcoma. Leukemia. 2012;26:2469–73.
Pileri SA, Ascani S, Cox MC, Campidelli C, Bacci F, Piccioli M, et al. Myeloid sarcoma: clinico-pathologic, phenotypic and cytogenetic analysis of 92 adult patients. Leukemia. 2007;21:340–50.
Yu WJ, Sun YQ, Han TT, Ye PP, Zhang XH, Xu LP, et al. Haploidentical hematopoietic stem cell transplantation for patients with myeloid sarcoma: a single center retrospective study. Ann Hematol. 2021;100:799–808.
Lu DP, Dong L, Wu T, Huang XJ, Zhang MJ, Han W, et al. Conditioning including antithymocyte globulin followed by unmanipulated HLA-mismatched/haploidentical blood and marrow transplantation can achieve comparable outcomes with HLA-identical sibling transplantation. Blood. 2006;107:3065–73.
Huang XJ, Liu DH, Liu KY, Xu LP, Chen H, Han W, et al. Haploidentical hematopoietic stem cell transplantation without in vitro T-cell depletion for the treatment of hematological malignancies. Bone Marrow Transplant. 2006;38:291–7.
Wang Y, Fu G, Xu L, Wang Y, Cheng Y, Zhang Y, et al. Risk factors for positive post-transplantation measurable residual disease in patients with acute lymphoblastic leukemia. Chin Med J. 2024. https://doi.org/10.1097/CM9.0000000000003150. Epub ahead of print.
Zhang XH, Chen J, Han MZ, Huang H, Jiang EL, Jiang M, et al. The consensus from The Chinese Society of Hematology on indications, conditioning regimens and donor selection for allogeneic hematopoietic stem cell transplantation: 2021 update. J Hematol Oncol. 2021;14:145.
Wang Y, Chen H, Chen J, Han M, Hu J, Jiong H, et al. The consensus on the monitoring, treatment, and prevention of leukemia relapse after allogeneic hematopoietic stem cell transplantation in China. Cancer Lett. 2018;438:63–75.
Dohner H, Wei AH, Appelbaum FR, Craddock C, DiNardo CD, Dombret H, et al. Diagnosis and management of AML in adults: 2022 recommendations from an international expert panel on behalf of the ELN. Blood. 2022;140:1345–77.
Tsimberidou AM, Kantarjian HM, Estey E, Cortes JE, Verstovsek S, Faderl S, et al. Outcome in patients with nonleukemic granulocytic sarcoma treated with chemotherapy with or without radiotherapy. Leukemia. 2003;17:1100–3.
Gordon MY, Blackett NM. Reconstruction of the hematopoietic system after stem cell transplantation. Cell Transplant. 1998;7:339–44.
Schoemans HM, Lee SJ, Ferrara JL, Wolff D, Levine JE, Schultz KR, et al. EBMT-NIH-CIBMTR Task Force position statement on standardized terminology & guidance for graft-versus-host disease assessment. Bone Marrow Transplant. 2018;53:1401–15.
Lee SJ, Wolff D, Kitko C, Koreth J, Inamoto Y, Jagasia M, et al. Measuring therapeutic response in chronic graft-versus-host disease. National Institutes of Health consensus development project on criteria for clinical trials in chronic graft-versus-host disease: IV. The 2014 Response Criteria Working Group report. Biol Blood Marrow Transplant. 2015;21:984–99.
Shan M, Lu Y, Yang M, Wang P, Lu S, Zhang L, et al. Characteristics and transplant outcome of myeloid sarcoma: a single-institute study. Int J Hematol. 2021;113:682–92.
Zhao H, Dong Z, Wan D, Cao W, Xing H, Liu Z, et al. Clinical characteristics, treatment, and prognosis of 118 cases of myeloid sarcoma. Sci Rep. 2022;12:6752.
Almond LM, Charalampakis M, Ford SJ, Gourevitch D, Desai A. Myeloid sarcoma: presentation, diagnosis, and treatment. Clin Lymphoma Myeloma Leuk. 2017;17:263–7.
Untaaveesup S, Trithiphen S, Kulchutisin K, Rungjirajittranon T, Leelakanok N, Panyoy S, et al. Genetic alterations in myeloid sarcoma among acute myeloid leukemia patients: insights from 37 cohort studies and a meta-analysis. Front Oncol. 2024;14:1325431.
Yang Y, Shu Y, Tang Y, Zhao S, Jia Y, Ji J, et al. RNA sequencing of myeloid sarcoma, shed light on myeloid sarcoma stratification. Cancer Med. 2023;12:9156–66.
Eckardt JN, Stolzel F, Kunadt D, Rollig C, Stasik S, Wagenfuhr L, et al. Molecular profiling and clinical implications of patients with acute myeloid leukemia and extramedullary manifestations. J Hematol Oncol. 2022;15:60.
Dalland JC, Meyer R, Ketterling RP, Reichard KK. Myeloid sarcoma with CBFB-MYH11 fusion (inv(16) or t(16;16)) prevails in the abdomen. Am J Clin Pathol. 2020;153:333–41.
Sen F, Zhang XX, Prieto VG, Shea CR, Qumsiyeh MB. Increased incidence of trisomy 8 in acute myeloid leukemia with skin infiltration (leukemia cutis). Diagn Mol Pathol. 2000;9:190–4.
Schwyzer R, Sherman GG, Cohn RJ, Poole JE, Willem P. Granulocytic sarcoma in children with acute myeloblastic leukemia and t(8;21). Med Pediatr Oncol. 1998;31:144–9.
Acknowledgements
We thank all patients and the data collection team who participated in this study.
Funding
This work was supported by the National Key Research and Development Program of China (No. 2023YFC2507803), the Key Program of National Natural Science Foundation of China (No. 82230004) and the Beijing Natural Science Foundation (No. 7242154).
Author information
Authors and Affiliations
Contributions
XHZ, JS and YCZ contributed to the study conception and design. JS, JW, YZ, AQW, YZ, FQL, CL, JHQ, JW, RX, QG, XPL and LLS collected the data. YJX, YHL, LF, ZXJ, MJ, PCH, XBH, SJG, SBW and SJF reviewed the cases and provided the data. JS and YCZ analyzed the data and wrote the original manuscript. LPX, XJH and XHZ reviewed the structure and statistical section of the paper. All the authors critically reviewed and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sun, J., Zhang, YC., Wei, J. et al. Outcomes of allogeneic hematopoietic stem cell transplantation versus intensive chemotherapy in patients with myeloid sarcoma: a nationwide representative multicenter study. Bone Marrow Transplant 60, 319–325 (2025). https://doi.org/10.1038/s41409-024-02485-y
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/s41409-024-02485-y