Abstract
Background
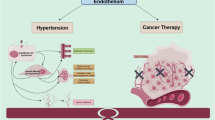
Vascular endothelial growth factor inhibitors (VEGFIs) are effective anticancer agents which often induce hypertension. VEGFI-induced hypertension is sodium-sensitive in animal studies. Therefore, the efficacy of dietary sodium restriction (DSR) to prevent VEGFI-induced hypertension in cancer patients was studied.
Methods
Cancer patients with VEGFI-induced hypertension (day mean >135/85 mmHg or a rise in systolic and/or diastolic BP ≥ 20 mmHg) were treated with DSR (aiming at <4 g salt/day). The primary endpoint was the difference in daytime mean arterial blood pressure (MAP) increase between the treatment cycle with and without DSR.
Results
During the first VEGFI treatment cycle without DSR, mean daytime MAP increased from 95 to 110 mmHg. During the subsequent treatment cycle with DSR, mean daytime MAP increased from 94 to 102 mmHg. Therefore, DSR attenuated the increase in mean daytime MAP by 7 mmHg (95% CI 1.3–12.0, P = 0.009). DSR prevented the rise in the endothelin-1/renin ratio that normally accompanies VEGFI-induced hypertension (P = 0.020) and prevented the onset of proteinuria: 0.15 (0.10–0.25) g/24 h with DSR versus 0.19 (0.11–0.32) g/24 h without DSR; P = 0.005.
Discussion
DSR significantly attenuated VEGFI induced BP rise and proteinuria and thus is an effective non-pharmacological intervention.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Data will be made available upon reasonable request.
References
Ferrara N, Adamis AP. Ten years of anti-vascular endothelial growth factor therapy. Nat Rev Drug Discov. 2016;15:385–403.
Moslehi JJ. Cardiovascular toxic effects of targeted cancer therapies. N Engl J Med. 2016;375:1457–67.
van Dorst DCH, Dobbin SJH, Neves KB, Herrmann J, Herrmann SM, Versmissen J, et al. Hypertension and prohypertensive antineoplastic therapies in cancer patients. Circ Res. 2021;128:1040–61.
Lankhorst S, Baelde HF, Clahsen-van Groningen MC, Smedts FM, Danser AH, van den Meiracker AH. Effect of high salt diet on blood pressure and renal damage during vascular endothelial growth factor inhibition with sunitinib. Nephrol Dial Transplant. 2016;31:914–21.
Gu JW, Manning RD Jr., Young E, Shparago M, Sartin B, Bailey AP. Vascular endothelial growth factor receptor inhibitor enhances dietary salt-induced hypertension in Sprague-Dawley rats. Am J Physiol Regul Integr Comp Physiol. 2009;297:R142–8.
Machnik A, Neuhofer W, Jantsch J, Dahlmann A, Tammela T, Machura K, et al. Macrophages regulate salt-dependent volume and blood pressure by a vascular endothelial growth factor-C-dependent buffering mechanism. Nat Med. 2009;15:545–52.
Selvarajah V, Mäki-Petäjä KM, Pedro L, Bruggraber SFA, Burling K, Goodhart AK, et al. Novel mechanism for buffering dietary salt in humans: effects of salt loading on skin sodium, vascular endothelial growth factor C, and blood pressure. Hypertension. 2017;70:930–7.
Karlsen TV, Nikpey E, Han J, Reikvam T, Rakova N, Castorena-Gonzalez JA, et al. High-salt diet causes expansion of the lymphatic network and increased lymph flow in skin and muscle of rats. Arterioscler Thromb Vasc Biol. 2018;38:2054–64.
Wiig H, Schröder A, Neuhofer W, Jantsch J, Kopp C, Karlsen TV, et al. Immune cells control skin lymphatic electrolyte homeostasis and blood pressure. J Clin Invest. 2013;123:2803–15.
Totzeck M, Mincu RI, Mrotzek S, Schadendorf D, Rassaf T. Cardiovascular diseases in patients receiving small molecules with anti-vascular endothelial growth factor activity: a meta-analysis of approximately 29,000 cancer patients. Eur J Prev Cardiol. 2018;25:482–94.
Versmissen J, Mirabito Colafella KM, Koolen SLW, Danser AHJ. Vascular cardio-oncology: vascular endothelial growth factor inhibitors and hypertension. Cardiovasc Res. 2019;115:904–14.
Zhu X, Stergiopoulos K, Wu S. Risk of hypertension and renal dysfunction with an angiogenesis inhibitor sunitinib: systematic review and meta-analysis. Acta Oncol. 2009;48:9–17.
Kappers MH, Smedts FM, Horn T, Van Esch JH, Sleijfer S, Leijten F, et al. The vascular endothelial growth factor receptor inhibitor sunitinib causes a preeclampsia-like syndrome with activation of the endothelin system. Hypertension. 2011;58:295–302.
Kappers MH, Van Esch JHM, Sluiter W, Sleijfer S, Danser AHJ, Van den Meiracker AHJ. Hypertension induced by the tyrosine kinase inhibitor sunitinib is associated with increased circulating endothelin-1 levels. Hypertension. 2010;56:675–81.
Levine RJ, Maynard SE, Qian C, Lim KH, England LJ, Yu KF, et al. Circulating angiogenic factors and the risk of preeclampsia. N. Engl J Med. 2004;350:672–83.
Maynard SE, Min JY, Merchan J, Lim KH, Li J, Mondal S, et al. Excess placental soluble fms-like tyrosine kinase 1 (sFlt1) may contribute to endothelial dysfunction, hypertension, and proteinuria in preeclampsia. J Clin Investig. 2003;111:649–58.
Mirabito Colafella KM, Neuman RI, Visser W, Danser AHJ, Versmissen J. Aspirin for the prevention and treatment of pre-eclampsia: a matter of COX-1 and/or COX-2 inhibition? Basic Clin Pharmacol Toxicol. 2020;127:132–41.
Mirabito Colafella KM, Neves KB, Montezano AC, Garrelds IM, Van Veghel R, De, et al. Selective ETA versus dual ETA/B receptor blockade for the prevention of sunitinib-induced hypertension and albuminuria in WKY rats. Cardiovasc Res. 2019;116:1779–90.
de Jesus-Gonzalez N, Robinson E, Moslehi J, Humphreys BD. Management of antiangiogenic therapy-induced hypertension. Hypertension. 2012;60:607–15.
Bossi P, Delrio P, Mascheroni A, Zanetti M. The spectrum of malnutrition/cachexia/sarcopenia in oncology according to different cancer types and settings: a narrative review. Nutrients. 2021;13:1980.
Azizi M, Chedid A, Oudard S. Home blood-pressure monitoring in patients receiving sunitinib. N Engl J Med. 2008;358:95–7.
Parati G, Stergiou G, O’Brien E, Asmar R, Beilin L, Bilo G, et al. European Society of Hypertension practice guidelines for ambulatory blood pressure monitoring. J Hypertens. 2014;32:1359–66.
Bovée DM, Visser WJ, Middel I, De Mik-van Egmond A, Greupink R, Masereeuw R, et al. A randomized trial of distal diuretics versus dietary sodium restriction for hypertension in chronic kidney disease. J Am Soc Nephrol. 2020;31:650–62.
Kloth JS, Klümpen HF, Yu H, Eechoute K, Samer CF, Kam BLR, et al. Predictive value of CYP3A and ABCB1 phenotyping probes for the pharmacokinetics of sunitinib: the ClearSun study. Clin Pharmacokinet. 2014;53:261–9.
Lankhorst S, Kappers MHW, Van Esch JHM, Smedts FMM, Sleijfer S, Mathijssen RHJ, et al. Treatment of hypertension and renal injury induced by the angiogenesis inhibitor sunitinib: preclinical study. Hypertension. 2014;64:1282–9.
Khalil RA. Modulators of the vascular endothelin receptor in blood pressure regulation and hypertension. Curr Mol Pharmacol. 2011;4:176–86.
Pollock DM, Pollock JS. Evidence for endothelin involvement in the response to high salt. Am J Physiol Ren Physiol. 2001;281:F144–150.
Markó L, Dörr A, Linz P, Van den Meiracker AH, Garrelds IM, Kuehne T, et al. Effect of sunitinib treatment on skin sodium accumulation in patients with renal cancer: a pilot study. Hypertension. 2022;79:e103–e105.
Dahlmann A, Linz P, Zucker I, Haag V, Jantsch J, Dienemann T, et al. Reduction of tissue Na(+) accumulation after renal transplantation. Kidney Int Rep. 2021;6:2338–47.
Lankhorst S, Severs D, Markó L, Rakova N, Titze J, Müller D N et al. Salt sensitivity of angiogenesis inhibition-induced blood pressure rise: role of interstitial sodium accumulation? Hypertension. 2017;69:919–26.
Kohan DE, Rossi NF, Inscho EW, Pollock DM. Regulation of blood pressure and salt homeostasis by endothelin. Physiol Rev. 2011;91:1–77.
Wagner OF, Christ G, Wojta J, Vierhapper H, Parzer S, Nowotny PJ, et al. Polar secretion of endothelin-1 by cultured endothelial cells. J Biol Chem. 1992;267:16066–8.
Verdonk K, Saleh L, Lankhorst S, Smilde JEI, Van Ingen MM, Garrelds IM, et al. Association studies suggest a key role for endothelin-1 in the pathogenesis of preeclampsia and the accompanying renin-angiotensin-aldosterone system suppression. Hypertension. 2015;65:1316–23.
Matsuda H, Ito Y, Hosono K, Tsuru S, Inoue T, Nakamoto S, et al. Roles of thromboxane receptor signaling in enhancement of lipopolysaccharide-induced lymphangiogenesis and lymphatic drainage function in diaphragm. Arterioscler Thromb Vasc Biol. 2021;41:1390–407.
Kashiwagi S, Hosono K, Suzuki T, Takeda A, Uchinuma E, Majima M. Role of COX-2 in lymphangiogenesis and restoration of lymphatic flow in secondary lymphedema. Lab Invest. 2011;91:1314–25.
Mirabito Colafella KM, Van Dorst DCH, Neuman RI, Van Doorn L, Neves KB, Montezano AC, et al. Differential effects of cyclo-oxygenase 1 and 2 inhibition on angiogenesis inhibitor-induced hypertension and kidney damage. Clin Sci. 2022;13:675–94.
Verdonk K, Visser W, Van den Meiracker AH, Danser AHJ. Are aldosterone levels inappropriately low in preeclampsia? Hypertension. 2013;62:e39.
Verdonk K, Visser W, Van Den Meiracker AH, Danser AH. The renin-angiotensin-aldosterone system in pre-eclampsia: the delicate balance between good and bad. Clin Sci. 2014;126:537–44.
Roberge S, Giguére Y, Villa P, Nicolaides K, Vanio M, Forest JC, et al. Early administration of low-dose aspirin for the prevention of severe and mild preeclampsia: a systematic review and meta-analysis. Am J Perinatol. 2012;29:551–6.
Birukov A, Andersen LB, Herse F, Rakova N, Kitlen G, Kyhl HB, et al. Aldosterone, salt, and potassium intakes as predictors of pregnancy outcome, including preeclampsia. Hypertension. 2019;74:391–8.
Moutquin JM, Garner PR, Burrows RF, Rey E, Helewa ME, Lange IR, et al. Report of the Canadian Hypertension Society Consensus Conference: 2. Nonpharmacologic management and prevention of hypertensive disorders in pregnancy. CMAJ. 1997;157:907–19.
Versmissen J, Van Doorn L, Mirabito Colafella KM, Mathijssen RH, Danser AHJ. Sunitinib-induced blood pressure rise does not involve aldosterone: observations in a patient after bilateral adrenalectomy. J Hypertens. 2018;36:2279–80.
Acknowledgements
We thank Frank Geurts for help with the analyses and collecting the urine samples.
Funding
This work was funded by Stichting De Merel, The Hague, the Netherlands. The funders had no role in study design, data collection and analysis, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
LvD helped design the study, included patients, helped in sample collection, performed the analyses, interpreted the results and wrote the first draft of the manuscript. WJV helped design the study, developed the sodium restricted diet and monitoring plan, helped in sample collection and analyses and reviewed the manuscript. DCHvD helped in sample collection, analyses, writing and reviewing the manuscript. KMMC and SLWK helped in analyses and reviewing the manuscript. AvE-dM helped in applying and monitoring the sodium restricted diet and reviewing the manuscript. IMG, DMB and SB helped in data acquirement, analyses and reviewing the manuscript. EO-dH helped in design of the statistical plan and helped in interpreting the results and reviewed the manuscript. FALME included helped in data acquisition, interpreting the results and writing and reviewing the manuscript. EJH helped design the study and reviewed the manuscript. AHJD and RHJM helped design the study and in interpretation of the results and helped in writing and reviewing the manuscript. JV had the original research idea, received funding, designed the study, performed the analyses and helped writing and reviewing the manuscript. All authors approved the final version and agreed to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The study was approved by the Medical Ethics Committee from the Erasmus University Medical Center (MEC-2018-155) and complies with the Declaration of Helsinki. The study was registered at the Dutch trial registry (NTR7556).
Consent for publication
No individual person’s data are given.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
van Doorn, L., Visser, W.J., van Dorst, D.C.H. et al. Dietary sodium restriction prevents vascular endothelial growth factor inhibitor-induced hypertension. Br J Cancer 128, 354–362 (2023). https://doi.org/10.1038/s41416-022-02036-6
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/s41416-022-02036-6
This article is cited by
-
VEGFC ameliorates salt-sensitive hypertension and hypertensive nephropathy by inhibiting NLRP3 inflammasome via activating VEGFR3-AMPK dependent autophagy pathway
Cellular and Molecular Life Sciences (2023)