Abstract
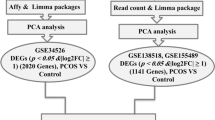
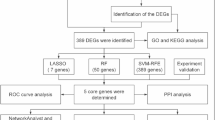
Granulosa cell (GC) apoptosis is intrinsically linked to the ovarian dysfunction of polycystic ovary syndrome (PCOS). Although oxidative stress and apoptosis in GCs have been detected in PCOS patients, how reactive oxygen species (ROS) links to GC apoptosis in PCOS remains to be further elucidated. Here, by integrating public single-cell RNA-seq data with clinical GC sample validation, we found that the expression of the E3 ubiquitin ligase WWP2 was significantly reduced, whereas its role in PCOS has not been previously reported. Notably, we first demonstrated that WWP2 overexpression can effectively antagonize mitochondrial apoptosis and ROS in KGNs. Mechanistically, oxidative stress weakened the interaction between WWP2 and BAK and reduced WWP2 expression, thereby suppressing BAK ubiquitination at Lys113. This inhibition impaired proteasomal degradation and consequently increased BAK protein levels. Consistently, disrupting BAK ubiquitination (BAK-K113R mutant) or knocking down WWP2 facilitated KGN apoptosis, and genetic ablation of Wwp2 in PCOS mice further aggravated GC apoptosis and hormonal disturbances. This study elucidates the molecular mechanism by which oxidative stress modulates GC mitochondrial apoptosis through WWP2-mediated BAK ubiquitination, and establishes WWP2 as a potential therapeutic target for PCOS.
Similar content being viewed by others
Data availability
All data relevant to the study are included in the article.
References
Teede HJ, Moran LJ, Morman R, Gibson M, Dokras A, Berry L, et al. Polycystic ovary syndrome perspectives from patients and health professionals on clinical features, current name, and renaming: a longitudinal international online survey. eClinicalMedicine. 2025;84:103287.
Mizgier M, Sansoni V, Więckowska B, Jarząbek-Bielecka G, Formanowicz D, Kędzia W, et al. The correlations between serum bone biomarkers and those related to metabolic and hormonal profile, low-grade inflammation and redox balance, in lean and overweight PCOS adolescent girls. Front Nutr. 2025;12:1477992.
Balen AH, Morley LC, Misso M, Franks S, Legro RS, Wijeyaratne CN, et al. The management of anovulatory infertility in women with polycystic ovary syndrome: an analysis of the evidence to support the development of global WHO guidance. Hum Reprod Update. 2016;22:687–708.
Moreira MV, Vale-Fernandes E, Albergaria IC, Alves MG, Monteiro MP. Follicular fluid composition and reproductive outcomes of women with polycystic ovary syndrome undergoing in vitro fertilization: A systematic review. Rev Endocr Metab Disord. 2023;24:1045–73.
Chen Y, Dai H, Mao F, Li Y, Feng R, Qian Y. Long noncoding RNA NONHSAT233728.1 promotes ROS accumulation and granulosa cell apoptosis by regulating the MAPK/ERK1/2 signaling pathway. FASEB J. 2025;39:e70681.
Liu Y, Jiang J-J, Du S-Y, Mu L-S, Fan J-J, Hu J-C, et al. Artemisinins ameliorate polycystic ovarian syndrome by mediating LONP1-CYP11A1 interaction. Science. 2024;384:eadk5382.
Qiao J, Feng HL. Extra- and intra-ovarian factors in polycystic ovary syndrome: impact on oocyte maturation and embryo developmental competence. Hum Reprod Update. 2011;17:17–33.
Yu H, Zhang W, Wang D, Shi B, Zhu Y, Hu W, et al. Exposure to 6PPD-Q induces dysfunctions of ovarian granulosa cells: Its potential role in PCOS. J Hazard Mater. 2025;486:137037.
Huang J-C, Duan C-C, Jin S, Sheng C-B, Wang Y-S, Yue Z-P, et al. HB-EGF induces mitochondrial dysfunction via estrogen hypersecretion in granulosa cells dependent on cAMP-PKA-JNK/ERK-Ca2+-FOXO1 pathway. Int J Biol Sci. 2022;18:2047–59.
Bas D, Abramovich D, Hernandez F, Tesone M. Altered expression of Bcl-2 and Bax in follicles within dehydroepiandrosterone-induced polycystic ovaries in rats. Cell Biol Int. 2011;35:423–9.
Wang Z, Wu H, Chang X, Song Y, Chen Y, Yan Z, et al. CKMT1 deficiency contributes to mitochondrial dysfunction and promotes intestinal epithelial cell apoptosis via reverse electron transfer-derived ROS in colitis. Cell Death Dis. 2025;16:177.
Czabotar PE, Garcia-Saez AJ. Mechanisms of BCL-2 family proteins in mitochondrial apoptosis. Nat Rev Mol Cell Biol. 2023;24:732–48.
Cheng P, Hou Y, Bian M, Fang X, Liu Y, Rao Y, et al. Parkin-mediated ubiquitination inhibits BAK apoptotic activity by blocking its canonical hydrophobic groove. Commun Biol. 2023;6:1260.
Schweighofer SV, Jans DC, Keller-Findeisen J, Folmeg A, Ilgen P, Bates M, et al. Endogenous BAX and BAK form mosaic rings of variable size and composition on apoptotic mitochondria. Cell Death Differ. 2024;31:469–78.
Hockings C, Alsop AE, Fennell SC, Lee EF, Fairlie WD, Dewson G, et al. Mcl-1 and Bcl-xL sequestration of bak confers differential resistance to BH3-only proteins. Cell Death Differ. 2018;25:721–34.
Bernardini JP, Brouwer JM, Tan IK, Sandow JJ, Huang S, Stafford CA, et al. Parkin inhibits BAK and BAX apoptotic function by distinct mechanisms during mitophagy. EMBO J. 2019;38:e99916.
Huang AS, Chin HS, Reljic B, Djajawi TM, Tan IKL, Gong J-N, et al. Mitochondrial E3 ubiquitin ligase MARCHF5 controls BAK apoptotic activity independently of BH3-only proteins. Cell Death Differ. 2023;30:632–46.
Someya S, Xu J, Kondo K, Ding D, Salvi RJ, Yamasoba T, et al. Age-related hearing loss in C57BL/6J mice is mediated by Bak-dependent mitochondrial apoptosis. Proc Natl Acad Sci USA. 2009;106:19432–7.
Wu H, Zhao B, Yao Q, Kang J. Dehydroepiandrosterone-induced polycystic ovary syndrome mouse model requires continous treatments to maintain reproductive phenotypes. J Ovarian Res. 2023;16:207.
Zhang N, Zhang Y, Xu J, Wang P, Wu B, Lu S, et al. α-myosin heavy chain lactylation maintains sarcomeric structure and function and alleviates the development of heart failure. Cell Res. 2023;33:679–98.
Li T, Zhang T, Gao H, Liu R, Gu M, Yang Y, et al. Tempol ameliorates polycystic ovary syndrome through attenuating intestinal oxidative stress and modulating of gut microbiota composition-serum metabolites interaction. Redox Bio. 2021;41:101886.
You S, Xu J, Yin Z, Wu B, Wang P, Hao M, et al. Down-regulation of WWP2 aggravates type 2 diabetes mellitus-induced vascular endothelial injury through modulating ubiquitination and degradation of DDX3X. Cardiovasc Diabetol. 2023;22:107.
Pervushin NV, Nilov DK, Zhivotovsky B, Kopeina GS. Bcl-2 modifying factor (bmf): ‘a mysterious stranger’ in the bcl-2 family proteins. Cell Death Differ. 2025 https://doi.org/10.1038/s41418-025-01562-z.
Holloway A, Simmonds M, Azad A, Fox JL, Storey A. Resistance to UV-induced apoptosis by β-HPV5 E6 involves targeting of activated BAK for proteolysis by recruitment of the HERC1 ubiquitin ligase. Int J Cancer. 2015;136:2831–43.
Iyer S, Bell F, Westphal D, Anwari K, Gulbis J, Smith BJ, et al. Bak apoptotic pores involve a flexible C-terminal region and juxtaposition of the C-terminal transmembrane domains. Cell Death Differ. 2015;22:1665–75.
You S, Xu J, Guo Y, Guo X, Zhang Y, Zhang N, et al. E3 ubiquitin ligase WWP2 as a promising therapeutic target for diverse human diseases. Mol Aspects Med. 2024;96:101257.
Cosentino K, Hertlein V, Jenner A, Dellmann T, Gojkovic M, Peña-Blanco A, et al. The interplay between BAX and BAK tunes apoptotic pore growth to control mitochondrial-DNA-mediated inflammation. Mol Cell. 2022;82:933–.e9.
Shang F, Taylor A. Ubiquitin-proteasome pathway and cellular responses to oxidative stress. Free Radic Biol Med. 2011;51:5–16.
Delrue C, Speeckaert MM. Renal implications of dysregulated protein homeostasis: insights into ubiquitin-proteasome and autophagy systems. Biomolecules. 2025;15:349.
Yang L-K, Ma W-J, Wang X, Chen H-R, Jiang Y-N, Sun H. Apoptosis in polycystic ovary syndrome: mechanisms and therapeutic implications. Life Sci. 2025;363:123394.
Yan H, Wang L, Zhang G, Li N, Zhao Y, Liu J, et al. Oxidative stress and energy metabolism abnormalities in polycystic ovary syndrome: from mechanisms to therapeutic strategies. Reprod Biol Endocrinol. 2024;22:159.
Li Y, Song H, Xu J, Wang Y, Bai L, Wang H, et al. TIGAR relieves PCOS by inhibiting granulosa cell apoptosis and oxidative stress through activating Nrf2. Mol Cell Endocrinol. 2024;594:112381.
Canipari R. Oocyte-granulosa cell interactions. Hum Reprod Update. 2000;6:279–89.
Wang H. The role of granulosa cells in oocyte development and aging: mechanisms and therapeutic opportunities. Semin Cell Dev Biol. 2025;171:103614.
Gong Y, Luo S, Fan P, Zhu H, Li Y, Huang W. Growth hormone activates PI3K/akt signaling and inhibits ROS accumulation and apoptosis in granulosa cells of patients with polycystic ovary syndrome. Reprod Biol Endocrinol. 2020;18:121.
Czabotar PE, Lessene G, Strasser A, Adams JM. Control of apoptosis by the BCL-2 protein family: implications for physiology and therapy. Nat Rev Mol Cell Biol. 2014;15:49–63.
Sun T, Liu Z, Yang Q. The role of ubiquitination and deubiquitination in cancer metabolism. Mol Cancer. 2020;19:146.
Liu X, Sun C, Zou K, Li C, Chen X, Gu H, et al. Novel PGK1 determines SKP2-dependent AR stability and reprograms granular cell glucose metabolism facilitating ovulation dysfunction. eBioMedicine. 2020;61:103058.
Zhang N, Zhang Y, Qian H, Wu S, Cao L, Sun Y. Selective targeting of ubiquitination and degradation of PARP1 by E3 ubiquitin ligase WWP2 regulates isoproterenol-induced cardiac remodeling. Cell Death Differ. 2020;27:2605–19.
Funding
This research was supported by Noncommunicable Chronic Diseases-National Science and Technology Major Project (2025ZD0547800), the National Natural Science Foundation of China (82371647, 82470283, 82271615), National Key Research and Development Program (No. 2024YFC2706900), the Science Foundation for Outstanding Youth of Liaoning Province (No. 2024JH3/50100023), Scientific Research Fund of Liaoning Provincial Education Department (No. LJ222410159094) and the Liaoning Revitalization Talents Program (XLYC2403104).
Author information
Authors and Affiliations
Contributions
Wenke Wang: Writing - original draft, Writing - review & editing, Software, Methodology, Formal analysis, Data curation. Wenjie Wu: Investigation, Software, Formal analysis, Validation, Methodology, Data curation. Mingjun Hao: Writing - original draft, Software, Formal analysis, Validation, Data curation. Shenshen Cui: Writing - review & editing, Methodology, Formal analysis, Data curation. Siqi Zhao: Resources, Investigation, Data curation. Jian-Fei Pei: Writing - review & editing, Visualization, Validation, Supervision, Conceptualization. Naijin Zhang: Writing - original draft, Supervision, Funding acquisition, Conceptualization. Da Li: Writing - review & editing, Writing - original draft, Visualization, Validation, Supervision, Project administration, Methodology, Investigation, Funding acquisition, Formal analysis, Data curation, Conceptualization.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
All experimental procedures involving human participants and animals were performed in accordance with the relevant guidelines and regulations. The human study was approved by the Ethics Committee of Shengjing Hospital, China Medical University, Shenyang (Approval No. SZCS2025016), and informed consent was obtained from all IVF participants for the collection of ovarian granulosa cells. The animal study protocol was approved by the Animal Ethics Committee of China Medical University (License number CMU2023296).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Edited by Professor Mauro Piacentini
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wang, W., Wu, W., Hao, M. et al. WWP2 underlies ROS-induced granulosa cell apoptosis by promoting ubiquitination of BAK in polycystic ovary syndrome. Cell Death Dis (2026). https://doi.org/10.1038/s41419-026-08500-y
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41419-026-08500-y