Abstract
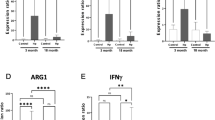
The maintenance of immune homeostasis is critical for tissue health and longevity, yet the regulatory mechanisms linking immune modulation to aging remain poorly understood. Here we found that the transcription factor cAMP response element-binding protein (CREB), activated by JNK signaling in aging guts, transcriptionally suppresses peptidoglycan recognition protein SC2(PGRP-SC2)—a homolog of anti-inflammatory PGLYRP1–4 with amidase activity. 16S rRNA sequencing revealed that CREB modulates not only microbial load but also microbiota composition. Elevated CREB activity decreased the Firmicutes/Bacteroidetes (F/B) ratio—a hallmark of age-associated dysbiosis in animals. Genetic enhancement of PGRP-SC2 rescues age-related gut hyperplasia, microbiota imbalance, and lifespan shortening induced by overactivation of CREB or its coactivator CRTC. Notably, CREB’s regulation of PGRP-SC2 operates independently of canonical immune pathways such as Imd/Relish, revealing a previously unrecognized layer of immune modulation. Our findings establish CREB as a central player in age-associated immune dysregulation and propose targeting the CREB-PGRP-SC2 axis as a potential therapeutic strategy for mitigating gut aging and its systemic consequences.
Similar content being viewed by others
Data availability
The 16S rRNA gene sequence data have been deposited in China National Center for Bioinformation (CNCB) (GSA: CRA018399 and CRA030538) with the link: https://ngdc.cncb.ac.cn/gsa/. All study data are included in the article and supporting information.
References
Hoffmann JA. The immune response of Drosophila. Nature. 2003;426:33–8.
Steiner H. Peptidoglycan recognition proteins: on and off switches for innate immunity. Immunol Rev. 2004;198:83–96.
Paredes JC, Welchman DP, Poidevin M, Lemaitre B. Negative regulation by amidase PGRPs shapes the Drosophila antibacterial response and protects the fly from innocuous infection. Immunity. 2011;35:770–9.
Chen Y, Xu W, Chen Y, Han A, Song J, Zhou X, et al. Renal NF-kappaB activation impairs uric acid homeostasis to promote tumor-associated mortality independent of wasting. Immunity. 2022;55:1594–608.e6.
Royet J, Dziarski R. Peptidoglycan recognition proteins: pleiotropic sensors and effectors of antimicrobial defences. Nat Rev Microbiol. 2007;5:264–77.
Buchon N, Broderick NA, Lemaitre B. Gut homeostasis in a microbial world: insights from Drosophila melanogaster. Nat Rev Microbiol. 2013;11:615–26.
Becker T, Loch G, Beyer M, Zinke I, Aschenbrenner AC, Carrera P, et al. FOXO-dependent regulation of innate immune homeostasis. Nature. 2010;463:369–73.
Guo L, Karpac J, Tran SL, Jasper H. PGRP-SC2 promotes gut immune homeostasis to limit commensal dysbiosis and extend lifespan. Cell. 2014;156:109–22.
Chen H, Zheng X, Zheng Y. Age-associated loss of lamin-B leads to systemic inflammation and gut hyperplasia. Cell. 2014;159:829–43.
Aleman FDD, Valenzano DR. Microbiome evolution during host aging. PLoS Pathog. 2019;15:e1007727.
Kapoor P, Tiwari A, Sharma S, Tiwari V, Sheoran B, Ali U, et al. Effect of anthocyanins on gut health markers, Firmicutes-Bacteroidetes ratio and short-chain fatty acids: a systematic review via meta-analysis. Sci Rep. 2023;13:1729.
Clark RI, Salazar A, Yamada R, Fitz-Gibbon S, Morselli M, Alcaraz J, et al. Distinct shifts in microbiota composition during Drosophila aging impair intestinal function and drive mortality. Cell Rep. 2015;12:1656–67.
Altarejos JY, Montminy M. CREB and the CRTC co-activators: sensors for hormonal and metabolic signals. Nat Rev Mol Cell Biol. 2011;12:141–51.
Yin JC, Del Vecchio M, Zhou H, Tully T. CREB as a memory modulator: induced expression of a dCREB2 activator isoform enhances long-term memory in Drosophila. Cell. 1995;81:107–15.
Mayr B, Montminy M. Transcriptional regulation by the phosphorylation-dependent factor CREB. Nat Rev Mol Cell Biol. 2001;2:599–609.
Yin Y, Ma P, Wang S, Zhang Y, Han R, Huo C, et al. The CRTC-CREB axis functions as a transcriptional sensor to protect against proteotoxic stress in Drosophila. Cell Death Dis. 2022;13:688.
Deng H, Gerencser AA, Jasper H. Signal integration by Ca(2+) regulates intestinal stem-cell activity. Nature. 2015;528:212–7.
Luan B, Yoon YS, Le Lay J, Kaestner KH, Hedrick S, Montminy M. CREB pathway links PGE2 signaling with macrophage polarization. Proc Natl Acad Sci USA. 2015;112:15642–7.
Wen AY, Sakamoto KM, Miller LS. The role of the transcription factor CREB in immune function. J Immunol. 2010;185:6413–9.
Gan T, Fan L, Zhao L, Misra M, Liu M, Zhang M, et al. JNK signaling in Drosophila aging and longevity. Int J Mol Sci. 2021;22:9649.
Wang S, Qi B, Du C, Ma P, Zhang Y, Chen S, et al. CREB-mediated sensing of bacterial membrane vesicles unveils a conserved host defense pathway. Mucosal Immunol. 2025;18:1313–24.
Ma P, Zhang Y, Yin Y, Wang S, Chen S, Liang X, et al. Gut microbiota metabolite tyramine ameliorates high-fat diet-induced insulin resistance via increased Ca(2+) signaling. EMBO J. 2024;43:3466–93.
Biteau B, Hochmuth CE, Jasper H. JNK activity in somatic stem cells causes loss of tissue homeostasis in the aging Drosophila gut. Cell Stem Cell. 2008;3:442–55.
Cha GH, Kim S, Park J, Lee E, Kim M, Lee SB, et al. Parkin negatively regulates JNK pathway in the dopaminergic neurons of Drosophila. Proc Natl Acad Sci USA. 2005;102:10345–50.
Zhang Y, Ma P, Wang S, Chen S, Deng H. Restoring calcium crosstalk between ER and mitochondria promotes intestinal stem cell rejuvenation through autophagy in aged Drosophila. Nat Commun. 2025;16:4909.
McGuire SE, Mao Z, Davis RL. Spatiotemporal gene expression targeting with the TARGET and gene-switch systems in Drosophila. Sci STKE. 2004;2004:pl6.
Rodriguez-Fernandez IA, Tauc HM, Jasper H. Hallmarks of aging Drosophila intestinal stem cells. Mech Ageing Dev. 2020;190:111285.
Ma P, Zhang Y, Yin Y, Wang S, Chen S, Liang X, et al. Gut microbiota metabolite tyramine ameliorates high-fat diet-induced insulin resistance via increased Ca(2+) signaling. EMBO J. 2024;43:3466–93.
Yin JC, Wallach JS, Del Vecchio M, Wilder EL, Zhou H, Quinn WG, et al. Induction of a dominant negative CREB transgene specifically blocks long-term memory in Drosophila. Cell. 1994;79:49–58.
Staubach F, Baines JF, Kunzel S, Bik EM, Petrov DA. Host species and environmental effects on bacterial communities associated with Drosophila in the laboratory and in the natural environment. PLoS ONE. 2013;8:e70749.
Garcia-Lozano M, Haynes J, Lopez-Ortiz C, Natarajan P, Pena-Garcia Y, Nimmakayala P, et al. Effect of pepper-containing diets on the diversity and composition of gut microbiome of Drosophila melanogaster. Int J Mol Sci. 2020;21:945.
Bischoff V, Vignal C, Duvic B, Boneca IG, Hoffmann JA, Royet J. Downregulation of the Drosophila immune response by peptidoglycan-recognition proteins SC1 and SC2. PLoS Pathog. 2006;2:e14.
Wiklund ML, Steinert S, Junell A, Hultmark D, Stoven S. The N-terminal half of the Drosophila Rel/NF-kappaB factor Relish, REL-68, constitutively activates transcription of specific Relish target genes. Dev Comp Immunol. 2009;33:690–6.
Moskalev A, Shaposhnikov M. Pharmacological inhibition of NF-kappaB prolongs lifespan of Drosophila melanogaster. Aging. 2011;3:391–4.
Demontis F, Perrimon N. FOXO/4E-BP signaling in Drosophila muscles regulates organism-wide proteostasis during aging. Cell. 2010;143:813–25.
Tain LS, Mortiboys H, Tao RN, Ziviani E, Bandmann O, Whitworth AJ. Rapamycin activation of 4E-BP prevents Parkinsonian dopaminergic neuron loss. Nat Neurosci. 2009;12:1129–35.
Akagi K, Wilson KA, Katewa SD, Ortega M, Simons J, Hilsabeck TA, et al. Dietary restriction improves intestinal cellular fitness to enhance gut barrier function and lifespan in D. melanogaster. PLoS Genet. 2018;14:e1007777.
Wu SC, Liao CW, Pan RL, Juang JL. Infection-induced intestinal oxidative stress triggers organ-to-organ immunological communication in Drosophila. Cell Host Microbe. 2012;11:410–7.
Wang L, Zhang L, Gong XD, Fu JL, Gan YW, Hou M, et al. PP-1beta and PP-2Aalpha modulate cAMP response element-binding protein (CREB) functions in aging control and stress response through de-regulation of alphaB-crystallin gene and p300-p53 signaling axis. Aging Cell. 2021;20:e13458.
Mair W, Morantte I, Rodrigues AP, Manning G, Montminy M, Shaw RJ, et al. Lifespan extension induced by AMPK and calcineurin is mediated by CRTC-1 and CREB. Nature. 2011;470:404–8.
Dziarski R, Gupta D. Review: mammalian peptidoglycan recognition proteins (PGRPs) in innate immunity. Innate Immun. 2010;16:168–74.
Saha S, Jing X, Park SY, Wang S, Li X, Gupta D, et al. Peptidoglycan recognition proteins protect mice from experimental colitis by promoting normal gut flora and preventing induction of interferon-gamma. Cell Host Microbe. 2010;8:147–62.
Deng H, Takashima S, Paul M, Guo M, Hartenstein V. Mitochondrial dynamics regulates Drosophila intestinal stem cell differentiation. Cell Death Discov. 2018;4:17.
Morris O, Deng H, Tam C, Jasper H. Warburg-like metabolic reprogramming in aging intestinal stem cells contributes to tissue hyperplasia. Cell Rep. 2020;33:108423.
Chen K, Luan X, Liu Q, Wang J, Chang X, Snijders AM, et al. Drosophila histone demethylase KDM5 regulates social behavior through immune control and gut microbiota maintenance. Cell Host Microbe. 2019;25:537–52.e8.
Broderick NA, Buchon N, Lemaitre B. Microbiota-induced changes in drosophila melanogaster host gene expression and gut morphology. mBio. 2014;5:e01117-14.
Acknowledgements
We thank Bloomington Drosophila Stock Center, Vienna Drosophila Stock Center, Tsinghua Fly Center and Dr. Lei Xue and Dr. Heinrich Jasper for stocks and reagents.
Funding
This work was supported by a National Key Research and Development Project [2018YFA0107100], National Natural Science Foundation of China [grant no. 31871371 and 32071147] and Tongji University Basic Scientific Research-Interdisciplinary Fund [grant no. 2000123424] to H.D.
Author information
Authors and Affiliations
Contributions
HD and SW conceived and designed the study. HD prepared the first draft of the paper. SW, BQ, PM, YZ, YY, SC contributed to the experimental work. SW conducted the statistical analysis of the data.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
All methods were performed in accordance with the relevant guidelines and regulations, and consent was obtained from all participants.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wang, S., Qi, B., Ma, P. et al. CREB suppresses PGRP-SC2 to drive age-related immune senescence and gut dysbiosis in Drosophila. Cell Death Discov. (2026). https://doi.org/10.1038/s41420-026-02955-w
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41420-026-02955-w