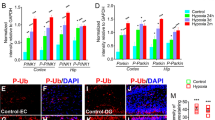
Abstract
Hypoxic stress contributes to brain disorders by causing neuronal injury, making it crucial to understand neuronal hypoxic response mechanisms for disease resistance. In the early stage of stress, neurons initiate a series of compensatory pathways to resist cell damage, but the underlying mechanisms have not been fully elucidated. In this study, we found that hypoxia transiently activates PTEN-induced kinase 1 (PINK1)-dependent mitophagy in the early stage before cell damage and neurological dysfunction. When PINK1-dependent mitophagy is inhibited, neuronal injury begins to exacerbate. Under hypoxia, overexpression of PINK1 can resist neuronal injury, while knockdown of PINK1 aggravates neuronal injury, revealing that PINK1-dependent mitophagy plays a key role in neuronal compensatory hypoxia response. Mechanistically, in the early stage of hypoxia, the nuclear translocation of HIF-1α increases, mediating the transcription of its downstream target molecule STOML2. STOML2 translocates to the outer mitochondrial membrane and participates in the cleavage of PGAM5. These processes initiate PINK1-dependent mitophagy. Knockdown of HIF-1α, STOML2, or PGAM5 inhibits mitophagy and worsens hypoxia-induced dysfunction, highlighting this pathway’s importance. Intermittent hypoxia, a conditioning strategy, stimulates endogenous protection. Notably, it activates the HIF-1α/STOML2 axis, inducing PINK1-dependent mitophagy and protecting neurons. In conclusion, our study reveals a new “self-protection” mechanism of neurons against hypoxic stress and discovers that intermittent hypoxia can effectively activate this pathway to resist neuronal injury, providing new insights into the mechanisms and interventions of hypoxia-related nerve injury.

Similar content being viewed by others
Data availability
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
References
Li S, Hafeez A, Noorulla F, Geng X, Shao G, Ren C, et al. Preconditioning in neuroprotection: from hypoxia to ischemia. Prog Neurobiol. 2017;157:79–91.
Li B, Yang WW, Yao BC, Chen QL, Zhao LL, Song YQ, et al. Liriodendrin alleviates myocardial ischemia‑reperfusion injury via partially attenuating apoptosis, inflammation and mitochondria damage in rats. Int J Mol Med. 2025;55:1–12.
Alshial EE, Abdulghaney MI, Wadan AS, Abdellatif MA, Ramadan NE, Suleiman AM, et al. Mitochondrial dysfunction and neurological disorders: A narrative review and treatment overview. Life Sci. 2023;334:122257.
Schmitt, LO & Gaspar, JM obesity-induced brain neuroinflammatory and mitochondrial changes. Metabolites. 2023;13. https://doi.org/10.3390/metabo13010086
Hoffmann L, Waclawczyk MS, Tang S, Hanschmann EM, Gellert M, Rust MB, et al. Cofilin1 oxidation links oxidative distress to mitochondrial demise and neuronal cell death. Cell Death Dis. 2021;12:953.
Liang R, Hou X, Zhou D, Zhu L, Teng L, Song W, et al. Exercise preconditioning mitigates Ischemia-Reperfusion injury in rats by enhancing mitochondrial respiration. Neuroscience. 2024;562:64–74.
Wen P, Sun Z, Gou F, Wang J, Fan Q, Zhao D, et al. Oxidative stress and mitochondrial impairment: Key drivers in neurodegenerative disorders. Ageing Res Rev. 2025;104:102667.
Almeida VN. Somatostatin and the pathophysiology of Alzheimer’s disease. Ageing Res Rev. 2024;96:102270.
Szczepanowska K, Trifunovic A. Mitochondrial matrix proteases: quality control and beyond. FEBS J. 2022;289:7128–46.
Tian RZ, Zhuang DL, Vong CT, He X, Ouyang Q, Liang JH, et al. Role of Autophagy in Myocardial Remodeling After Myocardial Infarction. J Cardiovasc Pharm. 2025;85:1–11.
Cheng Y, Gu W, Wu X, Tian W, Mu Z, Ye Y, et al. Allicin alleviates traumatic brain injury-induced neuroinflammation by enhancing PKC-δ-mediated mitophagy. Phytomedicine. 2025;139:156500.
Meng Q, Mi Y, Xu L, Liu Y, Liang D, Wang Y, et al. A quinolinyl analog of resveratrol improves neuronal damage after ischemic stroke by promoting Parkin-mediated mitophagy. Chin J Nat Med. 2025;23:214–24.
Wu H, Chen Q. Hypoxia activation of mitophagy and its role in disease pathogenesis. Antioxid Redox Signal. 2015;22:1032–46.
Liu L, Feng D, Chen G, Chen M, Zheng Q, Song P, et al. Mitochondrial outer-membrane protein FUNDC1 mediates hypoxia-induced mitophagy in mammalian cells. Nat Cell Biol. 2012;14:177–85.
Kuang Y, Ma K, Zhou C, Ding P, Zhu Y, Chen Q, et al. Structural basis for the phosphorylation of FUNDC1 LIR as a molecular switch of mitophagy. Autophagy. 2016;12:2363–73.
Ge P, Dawson VL, Dawson TM. PINK1 and Parkin mitochondrial quality control: a source of regional vulnerability in Parkinson’s disease. Mol Neurodegener. 2020;15:20.
Linqing L, Yuhan Q, Erfei L, Yong Q, Dong W, Chengchun T, et al. Hypoxia-induced PINK1/Parkin-mediated mitophagy promotes pulmonary vascular remodeling. Biochem Biophys Res Commun. 2021;534:568–75.
Shao Q, Liu J, Li G, Gu Y, Guo M, Guan Y, et al. Proteomic analysis reveals that mitochondria dominate the hippocampal hypoxic response in mice. Int J Mol Sci. 2022;23:14094.
Cheng M, Lin N, Dong D, Ma J, Su J, Sun L. PGAM5: A crucial role in mitochondrial dynamics and programmed cell death. Eur J Cell Biol. 2021;100:151144.
Zeb A, Choubey V, Gupta R, Kuum M, Safiulina D, Vaarmann A, et al. A novel role of KEAP1/PGAM5 complex: ROS sensor for inducing mitophagy. Redox Biol. 2021;48:102186.
Yan C, Gong L, Chen L, Xu M, Abou-Hamdan H, Tang M, et al. PHB2 (prohibitin 2) promotes PINK1-PRKN/Parkin-dependent mitophagy by the PARL-PGAM5-PINK1 axis. Autophagy. 2020;16:419–34.
Fan R, Jiang H, Hu Y, Xu Y, Zhou Y, Chen G, et al. Stomatin-like protein-2 attenuates macrophage pyroptosis and H9c2 cells apoptosis by protecting mitochondrial function. Biochem Biophys Res Commun. 2022;636:112–20.
Christie DA, Mitsopoulos P, Blagih J, Dunn SD, St-Pierre J, Jones RG, et al. Stomatin-like protein 2 deficiency in T cells is associated with altered mitochondrial respiration and defective CD4+ T cell responses. J Immunol. 2012;189:4349–60.
Guo H, Liang S, Wang Y, Zhou S, Yin D, Zhang S, et al. Cytochrome B5 type A alleviates HCC metastasis via regulating STOML2 related autophagy and promoting sensitivity to ruxolitinib. Cell Death Dis. 2022;13:623.
Ma W, Chen Y, Xiong W, Li W, Xu Z, Wang Y, et al. STOML2 interacts with PHB through activating MAPK signaling pathway to promote colorectal Cancer proliferation. J Exp Clin Cancer Res. 2021;40:359.
Wai T, Saita S, Nolte H, Müller S, König T, Richter-Dennerlein R, et al. The membrane scaffold SLP2 anchors a proteolytic hub in mitochondria containing PARL and the i-AAA protease YME1L. EMBO Rep. 2016;17:1844–56.
Tang T, Hu LB, Ding C, Zhang Z, Wang N, Wang T, et al. Src inhibition rescues FUNDC1-mediated neuronal mitophagy in ischaemic stroke. Stroke Vasc Neurol. 2024;9:367–79.
Chen G, Cizeau J, Vande Velde C, Park JH, Bozek G, Bolton J, et al. Nix and Nip3 form a subfamily of pro-apoptotic mitochondrial proteins. J Biol Chem. 1999;274:7–10.
Hu S, Zhang C, Ni L, Huang C, Chen D, Shi K, et al. Stabilization of HIF-1α alleviates osteoarthritis via enhancing mitophagy. Cell Death Dis. 2020;11:481.
Randle RK, Amara VR, Popik W. IFI16 is indispensable for promoting HIF-1α-mediated APOL1 expression in human podocytes under hypoxic conditions. Int J Mol Sci. 2024;25:3324.
Jewell UR, Kvietikova I, Scheid A, Bauer C, Wenger RH, Gassmann M. Induction of HIF-1alpha in response to hypoxia is instantaneous. FASEB J. 2001;15:1312–4.
Liu Y, Liu WC, Sun Y, Shen X, Wang X, Shu H, et al. Normobaric hyperoxia extends neuro- and vaso-protection of N-acetylcysteine in transient focal ischemia. Mol Neurobiol. 2017;54:3418–27.
Zhang Z, Yan J, Shi H. Role of hypoxia inducible factor 1 in hyperglycemia-exacerbated blood-brain barrier disruption in ischemic stroke. Neurobiol Dis. 2016;95:82–92.
Zheng Y, Huang C, Lu L, Yu K, Zhao J, Chen M, et al. STOML2 potentiates metastasis of hepatocellular carcinoma by promoting PINK1-mediated mitophagy and regulates sensitivity to lenvatinib. J Hematol Oncol. 2021;14:16.
Lazarou M, Sliter DA, Kane LA, Sarraf SA, Wang C, Burman JL, et al. The ubiquitin kinase PINK1 recruits autophagy receptors to induce mitophagy. Nature. 2015;524:309–14.
Pedrera L, Prieto Clemente L, Dahlhaus A, Lotfipour Nasudivar S, Tishina S, Olmo González D, et al. Ferroptosis triggers mitochondrial fragmentation via Drp1 activation. Cell Death Dis. 2025;16:40.
Qin X, Wang R, Xu H, Tu L, Chen H, Li H, et al. Identification of an autoinhibitory, mitophagy-inducing peptide derived from the transmembrane domain of USP30. Autophagy. 2022;18:2178–97.
Ling Z, Ge X, Jin C, Song Z, Zhang H, Fu Y, et al. Copper doped bioactive glass promotes matrix vesicles-mediated biomineralization via osteoblast mitophagy and mitochondrial dynamics during bone regeneration. Bioact Mater. 2025;46:195–212.
Li K, Xia X, Tong Y. Multiple roles of mitochondrial autophagy receptor FUNDC1 in mitochondrial events and kidney disease. Front Cell Dev Biol. 2024;12:1453365.
Martinez A, Sanchez-Martinez A, Pickering JT, Twyning MJ, Terriente-Felix A, Chen PL, et al. Mitochondrial CISD1/Cisd accumulation blocks mitophagy and genetic or pharmacological inhibition rescues neurodegenerative phenotypes in Pink1/parkin models. Mol Neurodegeneration. 2024;19:12.
Yi J, Wang HL, Lu G, Zhang H, Wang L, Li ZY, et al. Spautin-1 promotes PINK1-PRKN-dependent mitophagy and improves associative learning capability in an alzheimer disease animal model. Autophagy. 2024;20:2655–76.
Eldeeb MA, Fallahi A, Soumbasis A, Bayne AN, Trempe JF, Fon EA. Mitochondrial import stress and PINK1-mediated mitophagy: the role of the PINK1-TOMM-TIMM23 supercomplex. Autophagy. 2024;20:1903–5.
Shin HS, Park GH, Choi ES, Park SY, Kim DS, Chang J, et al. RNF213 variant and autophagic impairment: a pivotal link to endothelial dysfunction in moyamoya disease. J Cereb Blood Flow Metab : Off J Int Soc Cereb Blood Flow Metab. 2024;44:1801–15.
Lan XY, Li D, Cui Y, Nguyen TN, Li S, Chen HS. Proteomic analysis of jugular venous blood in acute large vessel occlusion stroke with futile recanalization. J Cereb Blood Flow Metab. 2024;44:702–11.
Zhao YH, Liang Y, Wang KJ, Jin SN, Yu XM, Zhang Q, et al. Endothelial lincRNA-p21 alleviates cerebral ischemia/reperfusion injury by maintaining blood-brain barrier integrity. J Cereb Blood Flow Metab. 2024;44:1532–50.
Wong Zhang DE, Gibson Hughes TA, Figueiredo Galvao HB, Lo C, Dinh QN, Zhang SR, et al. Post-stroke cognitive impairment and brain hemorrhage are augmented in hypertensive mice. J Cereb Blood Flow Metab. 2024;44:1517–34.
Zhou K, Tan Y, Zhang G, Li J, Xing S, Chen X, et al. Loss of SARM1 ameliorates secondary thalamic neurodegeneration after cerebral infarction. J Cereb Blood Flow Metab. 2024;44:224–38.
Gu R, Bai L, Yan F, Zhang S, Zhang X, Deng R, et al. Thioredoxin-1 decreases alpha-synuclein induced by MPTP through promoting autophagy-lysosome pathway. Cell Death Discov. 2024;10:93.
Park J, Jin L, Song HC, Chen Y, Jang EY, Park GH, et al. CO confers neuroprotection via activating the PERK-calcineurin pathway and inhibiting necroptosis. Cell Death Discov. 2025;11:254.
Su Y, Ke C, Li C, Huang C, Wan C. Intermittent hypoxia promotes the recovery of motor function in rats with cerebral ischemia by regulating mitochondrial function. Exp Biol Med (Maywood). 2022;247:1364–78.
Peng W, Ma H, Zhao R, Xu S, Lv M, Jing B, et al. Role of intermittent hypoxic training combined with methazolamide in the prevention of high-altitude cerebral edema in rats. Sci Rep. 2024;14:30252.
Wang X, Gong L, Wei C, Zhao Y, Ran L, Li P, et al. Inhibition of NSUN6 protects against intermittent hypoxia-induced oxidative stress and inflammatory response in adipose tissue through suppressing macrophage ferroptosis and M1 polarization. Life Sci. 2025;364:123433.
Panerai RB, Davies A, Clough RH, Beishon LC, Robinson TG, Minhas JS. The effect of hypercapnia on the directional sensitivity of dynamic cerebral autoregulation and the influence of age and sex. J Cereb Blood Flow Metab. 2024;44:272–83.
Sawan H, Li C, Buch S, Bernitsas E, Haacke EM, Ge Y, et al. Reduced oxygen extraction fraction in deep cerebral veins associated with cognitive impairment in multiple sclerosis. J Cereb Blood Flow Metab. 2024;44:1298–305.
Cheng X, Potenza DM, Brenna A, Ajalbert G, Yang Z, Ming XF. Aging increases hypoxia-induced endothelial permeability and blood-brain barrier dysfunction by upregulating arginase-II. Aging Dis. 2024;15:2710–5415.
Kataura T, Wilson N, Ma G, Korolchuk VI. Mitophagy as a guardian against cellular aging. Autophagy. 2025;21:249–51.
Funding
This research was supported by the National Natural Science Foundation of China (Grant number: 32100925, 82571493), National Scientific and Technological Innovation 2030 of China-Major project (2023ZD0505300, 2023ZD0505304), Outstanding Youth Project of Capital Medical University (Grant number: B2403), the Beijing Nova Program (Grant number: 20230484436), the Chinese Institutes for Medical Research (Grant number: CX23YQ01) and Beijing-Tianjin-Hebei Basic Research Cooperation Project, No. 22JCZXJC00190.
Author information
Authors and Affiliations
Contributions
Yuning Li performed most of the experiments in this study and drafted the majority of the manuscript content; Zirui Xu contributed to the experiments and undertook the revision of the manuscript; Zhengming Tian undertook the revision of the manuscript and helped to analyze the study data; Yakun Gu investigated the experiments process; Yingxia Liu helped design the methodology; Feiyang Jin contributed to the experiments; Mengyuan Guo participated in the experiments and helped write the manuscript; Qianqian Shao provided the materials, animals; Xunming Ji contributed to the development of experimental ideas; Jia Liu designed the experiments and supervise the research activity execution.
Corresponding author
Ethics declarations
Ethics approval
All animal experiments were approved by the Animal Care and Use Committee of the Institute of Animal Management, Capital Medical University (permit no. AEEI-2022-073), and conducted in accordance with ethical requirements and ARRIVE guidelines.
Consent for publication
Consent for publication was obtained from the participants.
Competing interests
The authors declare no competing interests.
Declaration of Artificial Intelligence (AI)
During the writing process of this article, we used ChatGPT-4o for language refinement and optimization to enhance readability and fluency. However, all research content, data analysis, and conclusions were independently conducted by the authors. ChatGPT-4o was solely used for language enhancement and did not influence the scientific integrity or authenticity of the study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Li, Y., Xu, Z., Tian, Z. et al. Novel mechanism of neuronal hypoxia response: HIF-1α/STOML2 mediated PINK1-dependent mitophagy activation against neuronal injury. Cell Death Discov. (2026). https://doi.org/10.1038/s41420-026-02960-z
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41420-026-02960-z