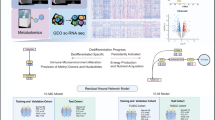
Abstract
One of the fastest-growing malignant tumors in the world is thyroid cancer (TC), and there are currently no effective treatments for its aggressive subtypes, such as anaplastic carcinoma and radioactive iodine-refractory differentiated thyroid carcinoma. Recent investigations have shown that DNA methylation and metabolic reprogramming are not independent events, but rather create a closely interconnected, mutually reinforcing network of carcinogenic processes. On the one hand, metabolic reprogramming influences the methylation status of tumor suppressor genes and thyroid function genes by dynamically regulating the activity of DNA methyltransferases and demethylases through important metabolites (such as S-adenosylmethionine, or SAM, and α-KG) and oncogenic signaling pathways (like PI3K/AKT). Conversely, DNA methylation systematically remodels cellular glucose, lipid, and amino acid metabolism by directly silencing metabolic enzyme genes (such as FASN and GLS) and thyroid differentiation markers (such as NIS) to fulfill its proliferative demands. Tumor growth, treatment resistance, and the development of an immunosuppressive microenvironment are all fueled by this ongoing bidirectional interaction, which creates a self-reinforcing oncogenic cycle. As a result, the limitations of earlier discrete debates on DNA methylation or metabolic reprogramming are overcome in this review. To methodically clarify their crosstalk mechanisms, a theoretical framework based on the “DNA methylation-metabolism axis” is suggested. Additionally, it suggests multimodal therapy approaches that focus on this axis. Incorporating biomimetic delivery technologies, combined with epigenetic, metabolic, and immunotherapies, to lay the groundwork for comprehending TC causes and creating targeted treatments.

Similar content being viewed by others
Data availability
Not Applicable.
References
Siegel RL, Miller KD, Wagle NS, Jemal A. Cancer statistics, 2023. CA: A Cancer J Clin. 2023;73:17–48.
Voelker R. What Is Thyroid Cancer? JAMA. 2024;332:346.
Boucai L, Zafereo M, Cabanillas ME. Thyroid Cancer: A Review. JAMA. 2024;331:425–35.
Evans LK, Chen H, Taki Labib M, Cronkite DA, Yu AC, Ashendouek M, et al. Improved Survival of Advanced-Stage Anaplastic Thyroid Cancer With Systemic Therapy. The Laryngoscope [Internet]. 2025 Jan [cited 2025 Nov 13];135. Available from: https://pubmed.ncbi.nlm.nih.gov/39162326/.
Volante M, Lam AK, Papotti M, Tallini G. Molecular pathology of poorly differentiated and anaplastic thyroid cancer: what do pathologists need to know?. Endocr Pathol. 2021;32:63–76.
Bao L, Xu T, Lu X, Huang P, Pan Z, Ge M. Metabolic reprogramming of thyroid cancer cells and crosstalk in their microenvironment. Front Oncol. 2021;11:773028.
Ju SH, Song M, Lim JY, Kang YE, Yi HS, Shong M. Metabolic Reprogramming in Thyroid Cancer. Endocrinol Metab. 2024;39:425–44.
Ge T, Gu X, Jia R, Ge S, Chai P, Zhuang A, et al. Crosstalk between metabolic reprogramming and epigenetics in cancer: updates on mechanisms and therapeutic opportunities. Cancer Commun. 2022;42:1049–82.
Zhang L, Feng Q, Wang J, Tan Z, Li Q, Ge M. Molecular basis and targeted therapy in thyroid cancer: Progress and opportunities. Biochim Biophys Acta Rev Cancer. 2023;1878:188928.
Díaz-Gago S, Vicente-Gutiérrez J, Ruiz-Rodríguez JM, Calafell J, Álvarez-Álvarez A, Lasa M, et al. Autophagy sustains mitochondrial respiration and determines resistance to BRAFV600E inhibition in thyroid carcinoma cells. Autophagy. 2024;20:1383–97.
Zhang Y, Wang J. Targeting uptake transporters for cancer imaging and treatment. Acta Pharm Sin B. 2020;10:79–90.
Yang J, Xu J, Wang W, Zhang B, Yu X, Shi S. Epigenetic regulation in the tumor microenvironment: molecular mechanisms and therapeutic targets. Signal Transduct Target Ther. 2023;8:210.
Lyko F. The DNA methyltransferase family: a versatile toolkit for epigenetic regulation. Nat Rev Genet. 2018;19:81–92.
Hodges AJ, Hudson NO, Buck-Koehntop BA. Cys2His2 Zinc Finger Methyl-CpG Binding Proteins: Getting a Handle on Methylated DNA. J Mol Biol. 2020;432:1640–60.
Mensah IK, Norvil AB, AlAbdi L, McGovern S, Petell CJ, He M, et al. Misregulation of the expression and activity of DNA methyltransferases in cancer. NAR Cancer. 2021;3:zcab045.
Newar K, Abdulla AZ, Salari H, Fanchon E, Jost D. Dynamical modeling of the H3K27 epigenetic landscape in mouse embryonic stem cells. PLoS Comput Biol. 2022;18:e1010450.
Drohat AC, Coey CT. Role of Base Excision ‘Repair’ Enzymes in Erasing Epigenetic Marks from DNA. Chem Rev. 2016;116:12711–29.
Li J, Wang X. Functional roles of conserved lncRNAs and circRNAs in eukaryotes. Noncoding RNA Res. 2024;9:1271–9.
Bandargal S, Rajab M, Forest VI, Pusztaszeri MP, Hier MP, da Silva SD, et al. Characteristics of PTEN Mutation in Thyroid Tumours: A Retrospective Chart Review. Cancers. 2023;15:1575.
Wei F, Wu Y, Wang Z, Li Y, Wang J, Shao G, et al. Diagnostic significance of DNA methylation of PTEN and DAPK in thyroid tumors. Clin Endocrinol (Oxf). 2020;93:187–95.
Sun M, Zhao M, Li R, Zhang Y, Shi X, Ding C, et al. SHMT2 promotes papillary thyroid cancer metastasis through epigenetic activation of AKT signaling. Cell Death Dis. 2024;15:87.
Brait M, Loyo M, Rosenbaum E, Ostrow KL, Markova A, Papagerakis S, et al. Correlation between BRAF mutation and promoter methylation of TIMP3, RARβ2 and RASSF1A in thyroid cancer. Epigenetics. 2012;7:710–9.
Lee SJ, Lee MH, Kim DW, Lee S, Huang S, Ryu MJ, et al. Cross-regulation between oncogenic BRAF(V600E) kinase and the MST1 pathway in papillary thyroid carcinoma. PLoS One. 2011;6:e16180.
Shou F, Xu F, Li G, Zhao Z, Mao Y, Yang F, et al. RASSF1A promoter methylation is associated with increased risk of thyroid cancer: a meta-analysis. Onco Targets Ther. 2017;10:247–57.
Stojanović Novković S, Šelemetjev S, Krajnović M, Božović A, Kožik B, Prosenc Zmrzljak U, et al. Implication of p16 Promoter Methylation, the BRAFV600E Mutation, and ETS1 Expression Determination on Papillary Thyroid Carcinoma Prognosis and High-Risk Patients’ Selection. Biomedicines. 2025;13:1583.
Cao YM, Gu J, Zhang YS, Wei WJ, Qu N, Wen D, et al. Aberrant hypermethylation of the HOXD10 gene in papillary thyroid cancer with BRAFV600E mutation. Oncol Rep. 2018;39:338–48.
Yang H, Zhou J, Mi J, Ma K, Fan Y, Ning J, et al. HOXD10 acts as a tumor-suppressive factor via inhibition of the RHOC/AKT/MAPK pathway in human cholangiocellular carcinoma. Oncol Rep. 2015;34:1681–91.
Xiao X, Chen M, Sang Y, Xue J, Jiang K, Chen Y, et al. Methylation-Mediated Silencing of ATF3 Promotes Thyroid Cancer Progression by Regulating Prognostic Genes in the MAPK and PI3K/AKT Pathways. Thyroid. 2023;33:1441–54.
Hong K, Cen K, Chen Q, Dai Y, Mai Y, Guo Y. Identification and validation of a novel senescence-related biomarker for thyroid cancer to predict the prognosis and immunotherapy. Front Immunol. 2023;14:1128390.
Zhu X, Xue C, Kang X, Jia X, Wang L, Younis MH, et al. DNMT3B-mediated FAM111B methylation promotes papillary thyroid tumor glycolysis, growth and metastasis. Int J Biol Sci. 2022;18:4372–87.
Guan H, Ji M, Hou P, Liu Z, Wang C, Shan Z, et al. Hypermethylation of the DNA mismatch repair gene hMLH1 and its association with lymph node metastasis and T1799A BRAF mutation in patients with papillary thyroid cancer. Cancer. 2008;113:247–55.
Li S, Hu G, Chen Y, Sang Y, Tang Q, Liu R. TERT upstream promoter methylation regulates TERT expression and acts as a therapeutic target in TERT promoter mutation-negative thyroid cancer. Cancer Cell Int. 2024;24:271.
Wu M, Wei B, Duan SL, Liu M, Ou-Yang DJ, Huang P, et al. Methylation-Driven Gene PLAU as a Potential Prognostic Marker for Differential Thyroid Carcinoma. Front Cell Dev Biol. 2022;10:819484.
Khan MS, Pandith AA, Masoodi SR, Wani KA, Ul Hussain M, Mudassar S. Epigenetic silencing of TSHR gene in thyroid cancer patients in relation to their BRAF V600E mutation status. Endocrine. 2014;47:449–55.
Qu M, Wan S, Ren B, Wu H, Liu L, Shen H. Association between TSHR gene methylation and papillary thyroid cancer: a meta-analysis. Endocrine. 2020;69:508–15.
Ye C, Jiang N, Zheng J, Zhang S, Zhang J, Zhou J. Epigenetic therapy: Research progress of Decitabine in the treatment of solid tumors. Biochim Biophys Acta (BBA) - Rev Cancer. 2024;1879:189066.
Coelho RG, Cazarin J, de M, Cavalcanti de Albuquerque JPA, de Andrade BM, Carvalho DP. Differential glycolytic profile and Warburg effect in papillary thyroid carcinoma cell lines. Oncol Rep. 2016;36:3673–81.
Zarkesh M, Arab N, Abooshahab R, Heydarzadeh S, Sheikholeslami S, Nozhat Z, et al. CpG island status as an epigenetic alteration for NIS promoter in thyroid neoplasms; a cross-sectional study with a systematic review. Cancer Cell Int. 2022;22:310.
Oh JM, Ahn BC. Molecular mechanisms of radioactive iodine refractoriness in differentiated thyroid cancer: Impaired sodium iodide symporter (NIS) expression owing to altered signaling pathway activity and intracellular localization of NIS. Theranostics. 2021;11:6251–77.
Martínez-Reyes I, Chandel NS. Cancer metabolism: looking forward. Nat Rev Cancer. 2021;21:669–80.
Fendt SM. 100 years of the Warburg effect: A cancer metabolism endeavor. Cell. 2024;187:3824–8.
Fagin JA, Krishnamoorthy GP, Landa I. Pathogenesis of cancers derived from thyroid follicular cells. Nat Rev Cancer. 2023;23:631–50.
Nagayama Y, Hamada K. Reprogramming of Cellular Metabolism and Its Therapeutic Applications in Thyroid Cancer. Metabolites. 2022;12:1214.
Liu CL, Yang PS, Wang TY, Huang SY, Kuo YH, Cheng SP. PGC1α downregulation and glycolytic phenotype in thyroid cancer. J Cancer. 2019;10:3819–29.
Liu CL, Hsu YC, Lee JJ, Chen MJ, Lin CH, Huang SY, et al. Targeting the pentose phosphate pathway increases reactive oxygen species and induces apoptosis in thyroid cancer cells. Mol Cell Endocrinol. 2020;499:110595.
Liu K, Du Y, Li H, Lin X. Identification of super-enhancer-associated transcription factors regulating glucose metabolism in poorly differentiated thyroid carcinoma. Genet Mol Biol. 2022;45:e20210370.
Liu C, Zhou X, Pan Y, Liu Y, Zhang Y. Pyruvate carboxylase promotes thyroid cancer aggressiveness through fatty acid synthesis. BMC Cancer. 2021;21:722.
Strickaert A, Corbet C, Spinette SA, Craciun L, Dom G, Andry G, et al. Reprogramming of Energy Metabolism: Increased Expression and Roles of Pyruvate Carboxylase in Papillary Thyroid Cancer. Thyroid. 2019;29:845–57.
Anderson NM, Mucka P, Kern JG, Feng H. The emerging role and targetability of the TCA cycle in cancer metabolism. Protein Cell. 2018;9:216–37.
Lee S, Rauch J, Kolch W. Targeting MAPK Signaling in Cancer: Mechanisms of Drug Resistance and Sensitivity. Int J Mol Sci. 2020;21:1102.
Yang Y, Wu J, Zhu H, Shi X, Liu J, Li Y, et al. Effect of hypoxia‑HIF‑1α‑periostin axis in thyroid cancer. Oncol Rep. 2024;51:57.
Song H, Qiu Z, Wang Y, Xi C, Zhang G, Sun Z, et al. HIF-1α/YAP signaling rewrites glucose/iodine metabolism program to promote papillary thyroid cancer progression. Int J Biol Sci. 2023;19:225–41.
Lukasiewicz M, Zwara A, Kowalski J, Mika A, Hellmann A. The role of lipid metabolism disorders in the development of thyroid cancer. Int J Mol Sci. 2024;25:7129.
von Roemeling CA, Marlow LA, Pinkerton AB, Crist A, Miller J, Tun HW, et al. Aberrant lipid metabolism in anaplastic thyroid carcinoma reveals stearoyl CoA desaturase 1 as a novel therapeutic target. J Clin Endocrinol Metab. 2015;100:E697–709.
Dube G, Tiamiou A, Bizet M, Boumahd Y, Gasmi I, Crake R, et al. Methylglyoxal: a novel upstream regulator of DNA methylation. J Exp Clin Cancer Res. 2023;42:78.
Chi J, Zhao J, Wei S, Li Y, Zhi J, Wang H, et al. A CRISPR-Cas9-Based Near-Infrared Upconversion-Activated DNA Methylation Editing System. ACS Appl Mater Interfaces. 2021;13:6043–52.
Valvo V, Iesato A, Kavanagh TR, Priolo C, Zsengeller Z, Pontecorvi A, et al. Fine-Tuning Lipid Metabolism by Targeting Mitochondria-Associated Acetyl-CoA-Carboxylase 2 in BRAF(V600E) Papillary Thyroid Carcinoma. Thyroid. 2021;31:1335–58.
Röhrig F, Schulze A. The multifaceted roles of fatty acid synthesis in cancer. Nat Rev Cancer. 2016;16:732–49.
Liang Z, He H, Zhang B, Kai Z, Zong L. Hypoxia expedites the progression of papillary thyroid carcinoma by promoting the CPT1A-mediated fatty acid oxidative pathway. Drug Dev Res. 2024;85:e22168.
Lu J, Zhang Y, Sun M, Ding C, Zhang L, Kong Y, et al. Multi-Omics Analysis of Fatty Acid Metabolism in Thyroid Carcinoma. Front Oncol. 2021;11:737127.
Tu Y, Chen Y, Mo L, Yan G, Xie J, Ji X, et al. Multi-omic analysis reveals a lipid metabolism gene signature and predicts prognosis and chemotherapy response in thyroid carcinoma. Cancer Med. 2025;14:e70819.
Häfliger P, Graff J, Rubin M, Stooss A, Dettmer MS, Altmann KH, et al. The LAT1 inhibitor JPH203 reduces growth of thyroid carcinoma in a fully immunocompetent mouse model. J Exp Clin Cancer Res. 2018;37:234.
Meng H, Xiao Z, Wang Q, Li D, Li Z. Genetic variations in amino acid metabolism-related genes are associated with risk of papillary thyroid carcinoma: a case-control study. BMC Endocr Disord. 2025;25:214.
Ducker GS, Rabinowitz JD. One-Carbon Metabolism in Health and Disease. Cell Metab. 2017;25:27–42.
Zhu X, Xuan Z, Chen J, Li Z, Zheng S, Song P. How DNA methylation affects the Warburg effect. Int J Biol Sci. 2020;16:2029–41.
Murugan AK, Bojdani E, Xing M. Identification and functional characterization of isocitrate dehydrogenase 1 (IDH1) mutations in thyroid cancer. Biochem Biophys Res Commun. 2010;393:555–9.
Xu W, Yang H, Liu Y, Yang Y, Wang P, Kim SH, et al. Oncometabolite 2-hydroxyglutarate is a competitive inhibitor of α-ketoglutarate-dependent dioxygenases. Cancer Cell. 2011;19:17–30.
Wang P, Dong Q, Zhang C, Kuan PF, Liu Y, Jeck WR, et al. Mutations in isocitrate dehydrogenase 1 and 2 occur frequently in intrahepatic cholangiocarcinomas and share hypermethylation targets with glioblastomas. Oncogene. 2013;32:3091–100.
Pusch S, Schweizer L, Beck AC, Lehmler JM, Weissert S, Balss J, et al. D-2-Hydroxyglutarate producing neo-enzymatic activity inversely correlates with frequency of the type of isocitrate dehydrogenase 1 mutations found in glioma. Acta Neuropathol Commun. 2014;2:19.
Qian J, Liu SX. CRISPR/dCas9-Tet1-Mediated DNA Methylation Editing. Bio Protoc. 2024;14:e4976.
Tao T, Yan J, Liao Z, Zhang W, Xu C, Li R. Oxidative low-density lipoprotein oxLDL induces pyroptosis in trophoblast cells HTR-8/Svneo by downregulating TET2. J Holist Integr Pharm. 2022;3:380–7.
Li Q, Wang W, Duan F, Wang Y, Chen S, Shi K, et al. DNMT3B Alleviates Liver Steatosis Induced by Chronic Low-grade LPS via Inhibiting CIDEA Expression. Cell Mol Gastroenterol Hepatol. 2024;17:59–77.
Cao K, Lv W, Wang X, Dong S, Liu X, Yang T, et al. Hypermethylation of Hepatic Mitochondrial ND6 Provokes Systemic Insulin Resistance. Adv Sci. 2021;8:2004507.
Zeng Q, Song J, Sun X, Wang D, Liao X, Ding Y, et al. A negative feedback loop between TET2 and leptin in adipocyte regulates body weight. Nat Commun. 2024;15:2825.
Fu R, Sun F, Wang W, Wang R, Zhang H, He X, et al. SHMT proteins: An emerging set of serine hydroxymethyltransferase in cancer. Cell Signal. 2025;135:111977.
Lee SE, Park S, Yi S, Choi NR, Lim MA, Chang JW, et al. Unraveling the role of the mitochondrial one-carbon pathway in undifferentiated thyroid cancer by multi-omics analyses. Nat Commun. 2024;15:1163.
Maddocks ODK, Labuschagne CF, Adams PD, Vousden KH. Serine Metabolism Supports the Methionine Cycle and DNA/RNA Methylation through De Novo ATP Synthesis in Cancer Cells. Mol Cell. 2016;61:210–21.
Schoultz E, Dahlberg J, Nilsson LM, Dzanan JJ, Carlsson T, Dahr N, et al. Involvement of KEAP1/NRF2 pathway in non-BRAF mutated squamous cell carcinoma of the thyroid. J Pathol. 2025;266:481–94.
Joshi K, Liu S, Breslin SJP, Zhang J. Mechanisms that regulate the activities of TET proteins. Cell Mol Life Sci. 2022;79:363.
Lopez-Serra P, Marcilla M, Villanueva A, Ramos-Fernandez A, Palau A, Leal L, et al. A DERL3-associated defect in the degradation of SLC2A1 mediates the Warburg effect. Nat Commun. 2014;5:3608.
Liu X, Fu J, Bi H, Ge A, Xia T, Liu Y, et al. DNA methylation of SFRP1, SFRP2, and WIF1 and prognosis of postoperative colorectal cancer patients. BMC Cancer. 2019;19:1212.
Veeck J, Niederacher D, An H, Klopocki E, Wiesmann F, Betz B, et al. Aberrant methylation of the Wnt antagonist SFRP1 in breast cancer is associated with unfavourable prognosis. Oncogene. 2006;25:3479–88.
Pate KT, Stringari C, Sprowl-Tanio S, Wang K, TeSlaa T, Hoverter NP, et al. Wnt signaling directs a metabolic program of glycolysis and angiogenesis in colon cancer. EMBO J. 2014;33:1454–73.
Gou R, Chen S, Lei Y, Wu P, Chen X, Zhang Q. Hypoxia Inhibitor Improves Iodine Uptake Disorder in Thyroid Cancer Through the hsa_circ_0023990/miR-448/DNMT1/NIS Axis. Cancer Sci. 2025;116:2113–24.
Li Q, Wang Y, Meng X, Wang W, Duan F, Chen S, et al. METTL16 inhibits papillary thyroid cancer tumorigenicity through m6A/YTHDC2/SCD1-regulated lipid metabolism. Cell Mol Life Sci. 2024;81:81.
Pham DV, Park PH. Adiponectin triggers breast cancer cell death via fatty acid metabolic reprogramming. J Exp Clin Cancer Res. 2022;41:9.
Tan SK, Hougen HY, Merchan JR, Gonzalgo ML, Welford SM. Fatty acid metabolism reprogramming in ccRCC: mechanisms and potential targets. Nat Rev Urol. 2023;20:48–60.
Zhang M, Yu L, Sun Y, Hao L, Bai J, Yuan X, et al. Comprehensive Analysis of FASN in Tumor Immune Infiltration and Prognostic Value for Immunotherapy and Promoter DNA Methylation. Int J Mol Sci. 2022;23:15603.
Nakamura MT, Yudell BE, Loor JJ. Regulation of energy metabolism by long-chain fatty acids. Prog Lipid Res. 2014;53:124–44.
Maréchal L, Laviolette M, Rodrigue-Way A, Sow B, Brochu M, Caron V, et al. The CD36-PPARγ Pathway in Metabolic Disorders. Int J Mol Sci. 2018;19:1529.
Christiansen C, Tomlinson M, Eliot M, Nilsson E, Costeira R, Xia Y, et al. Adipose methylome integrative-omic analyses reveal genetic and dietary metabolic health drivers and insulin resistance classifiers. Genome Med. 2022;14:75.
Bott AJ, Peng IC, Fan Y, Faubert B, Zhao L, Li J, et al. Oncogenic Myc Induces Expression of Glutamine Synthetase through Promoter Demethylation. Cell Metab. 2015;22:1068–77.
Dong J, Xiao D, Zhao Z, Ren P, Li C, Hu Y, et al. Epigenetic silencing of microRNA-137 enhances ASCT2 expression and tumor glutamine metabolism. Oncogenesis. 2017;6:e356.
Zhang K, Wang J, He Z, Qiu X, Sa R, Chen L. Epigenetic Targets and Their Inhibitors in Thyroid Cancer Treatment. Pharmaceuticals. 2023;16.
Provenzano MJ, Fitzgerald MP, Krager K, Domann FE. Increased iodine uptake in thyroid carcinoma after treatment with sodium butyrate and decitabine (5-Aza-dC). Otolaryngol–head Neck Surg. 2007;137:722–8.
Choi YW, Kim HJ, Kim YH, Park SH, Chwae YJ, Lee J, et al. B-RafV600E inhibits sodium iodide symporter expression via regulation of DNA methyltransferase 1. Exp Mol Med. 2014;46:e120–e120.
Laranjeira ABA, Hollingshead MG, Nguyen D, Kinders RJ, Doroshow JH, Yang SX. DNA damage, demethylation and anticancer activity of DNA methyltransferase (DNMT) inhibitors. Sci Rep. 2023;13:5964.
Plumb JA, Strathdee G, Sludden J, Kaye SB, Brown R. Reversal of drug resistance in human tumor xenografts by 2’-deoxy-5-azacytidine-induced demethylation of the hMLH1 gene promoter. Cancer Res. 2000;60:6039–44.
Vitale G, Dicitore A, Pepe D, Gentilini D, Grassi ES, Borghi MO, et al. Synergistic activity of everolimus and 5-aza-2’-deoxycytidine in medullary thyroid carcinoma cell lines. Mol Oncol. 2017;11:1007–22.
Zhang G, Gao X, Zhao X, Wu H, Yan M, Li Y, et al. Decitabine inhibits the proliferation of human T-cell acute lymphoblastic leukemia molt4 cells and promotes apoptosis partly by regulating the PI3K/AKT/mTOR pathway. Oncol Lett. 2021;21:340.
Hasegawa T, Adachi R, Iwakata H, Takeno T, Sato K, Sakamaki T. ErbB2 signaling epigenetically suppresses microRNA-205 transcription via Ras/Raf/MEK/ERK pathway in breast cancer. FEBS Open Bio. 2017;7:1154–65.
Gonçalves J, Emmons MF, Faião-Flores F, Aplin AE, Harbour JW, Licht JD, et al. Decitabine limits escape from MEK inhibition in uveal melanoma. Pigment Cell Melanoma Res. 2020;33:507–14.
Arosio G, Sharma GG, Villa M, Mauri M, Crespiatico I, Fontana D, et al. Synergistic Drug Combinations Prevent Resistance in ALK+ Anaplastic Large Cell Lymphoma. Cancers. 2021;13:4422.
Wang Y, Tong C, Dai H, Wu Z, Han X, Guo Y, et al. Low-dose decitabine priming endows CAR T cells with enhanced and persistent antitumour potential via epigenetic reprogramming. Nat Commun. 2021;12:409.
Wen SS, Wu YJ, Wang JY, Ni ZX, Dong S, Xie XJ, et al. BRAFV600E/p-ERK/p-DRP1(Ser616) Promotes Tumor Progression and Reprogramming of Glucose Metabolism in Papillary Thyroid Cancer. Thyroid. 2024;34:1246–59.
Zhao B, Aggarwal A, Im SY, Viswanathan K, Landa I, Nehs MA. Effect of Lactate Export Inhibition on Anaplastic Thyroid Cancer Growth and Metabolism. J Am Coll Surg. 2022;234:1044–50.
Halford S, Veal GJ, Wedge SR, Payne GS, Bacon CM, Sloan P, et al. A Phase I Dose-escalation Study of AZD3965, an Oral Monocarboxylate Transporter 1 Inhibitor, in Patients with Advanced Cancer. Clin Cancer Res. 2023;29:1429–39.
Mehibel M, Ortiz-Martinez F, Voelxen N, Boyers A, Chadwick A, Telfer BA, et al. Statin-induced metabolic reprogramming in head and neck cancer: a biomarker for targeting monocarboxylate transporters. Sci Rep. 2018;8:16804.
Huang T, Feng Q, Wang Z, Li W, Sun Z, Wilhelm J, et al. Tumor-Targeted Inhibition of Monocarboxylate Transporter 1 Improves T-Cell Immunotherapy of Solid Tumors. Adv Health Mater. 2021;10:e2000549.
Schlaepfer IR, Joshi M. CPT1A-mediated Fat Oxidation, Mechanisms, and Therapeutic Potential. Endocrinology. 2020;161:bqz046.
McGuffee RM, McCommis KS, Ford DA. Etomoxir: an old dog with new tricks. J Lipid Res. 2024;65:100604.
Lanman TA, Youssef G, Huang R, Rahman R, DeSalvo M, Flood T, et al. Ivosidenib for the treatment of IDH1-mutant glioma, grades 2-4: Tolerability, predictors of response, and outcomes. Neurooncol Adv. 2025;7:vdae227.
Rothenberg SM, McFadden DG, Palmer EL, Daniels GH, Wirth LJ. Redifferentiation of iodine-refractory BRAF V600E-mutant metastatic papillary thyroid cancer with dabrafenib. Clin Cancer Res. 2015;21:1028–35.
Subbiah V, Kreitman RJ, Wainberg ZA, Cho JY, Schellens JHM, Soria JC, et al. Dabrafenib and Trametinib Treatment in Patients With Locally Advanced or Metastatic BRAF V600-Mutant Anaplastic Thyroid Cancer. J Clin Oncol. 2018;36:7–13.
Ho AL, Grewal RK, Leboeuf R, Sherman EJ, Pfister DG, Deandreis D, et al. Selumetinib-enhanced radioiodine uptake in advanced thyroid cancer. N Engl J Med. 2013;368:623–32.
Schneider TC, de Wit D, Links TP, van Erp NP, van der Hoeven JJM, Gelderblom H, et al. Everolimus in Patients With Advanced Follicular-Derived Thyroid Cancer: Results of a Phase II Clinical Trial. J Clin Endocrinol Metab. 2017;102:698–707.
de Wit D, Schneider TC, Moes DJaR, Roozen CFM, den Hartigh J, Gelderblom H, et al. Everolimus pharmacokinetics and its exposure-toxicity relationship in patients with thyroid cancer. Cancer Chemother Pharm. 2016;78:63–71.
Lin QY, Qi QL, Hou S, Chen Z, Jiang N, Zhang L, et al. Silencing of AHNAK2 restricts thyroid carcinoma progression by inhibiting the Wnt/β-catenin pathway. Neoplasma. 2021;68:1063–71.
Zou Z, Zhong L. Anaplastic thyroid cancer: Genetic roles, targeted therapy, and immunotherapy. Genes Dis. 2025;12:101403.
Sheikholeslami S, Zarif-Yeganeh M, Farashi S, Azizi F, Kia SK, Teimoori-Toolabi L, et al. Promoter Methylation of Tumor Suppressors in Thyroid Carcinoma: A Systematic Review. Iran J Public Health. 2021;50:2461–72.
Yuan J, Dong X, Yap J, Hu J. The MAPK and AMPK signalings: interplay and implication in targeted cancer therapy. J Hematol Oncol. 2020;13:113.
Zeng JD, Wu WKK, Wang HY, Li XX. Serine and one-carbon metabolism, a bridge that links mTOR signaling and DNA methylation in cancer. Pharmacol Res. 2019;149:104352.
Li M, Wu C, Yang Y, Zheng M, Yu S, Wang J, et al. 3-Phosphoglycerate dehydrogenase: a potential target for cancer treatment. Cell Oncol. 2021;44:541–56.
Tajan M, Hennequart M, Cheung EC, Zani F, Hock AK, Legrave N, et al. Serine synthesis pathway inhibition cooperates with dietary serine and glycine limitation for cancer therapy. Nat Commun. 2021;12:366.
Orvain C, Tavitian S, Mediavilla C, Boyer F, Santagostino A, Venton G, et al. Outcome of patients with accelerated and blast-phase myeloproliferative neoplasms not eligible for intensive chemotherapy or allogeneic hematopoietic cell transplantation treated by azacitidine alone or in combination-A FIM study. Hemasphere. 2025;9:e70202.
Mina A, McGraw KL, Cunningham L, Kim N, Jen EY, Calvo KR, et al. Advancing drug development in myelodysplastic syndromes. Blood Adv. 2025;9:1095–104.
Montesinos P, Recher C, Vives S, Zarzycka E, Wang J, Bertani G, et al. Ivosidenib and Azacitidine in IDH1-Mutated Acute Myeloid Leukemia. N Engl J Med. 2022;386:1519–31.
Harba D, Corell A, Mansouri A, Brandal P, Blomstrand M, Jakola AS. A scoping review of proton radiation therapy and mutant-isocitrate dehydrogenase-inhibitors in IDH mutated lower-grade gliomas: pushing beyond surrogate end-points. Acta Neurochir (Wien). 2025;167:196.
Zhang J, Hu L, Wang H, Zhi J, Hou X, Wu Y, et al. Functional analysis and clinical significance of the isocitrate dehydrogenase 2 gene in papillary thyroid carcinoma. Cancer Manag Res. 2019;11:3765–77.
Khan ZM, Real AM, Marsiglia WM, Chow A, Duffy ME, Yerabolu JR, et al. Structural basis for the action of the drug trametinib at KSR-bound MEK. Nature. 2020;588:509–14.
Klieser E, Neumayer B, Di Fazio P, Mayr C, Neureiter D, Kiesslich T. HDACs as an emerging target in endocrine tumors: a comprehensive review. Expert Rev Endocrinol Metab. 2023;18:143–54.
Hamidi S, Ning MS, Phan J, Zafereo ME, Gule-Monroe MK, Dadu R. Recurrent Poorly Differentiated Thyroid Cancer Successfully Treated With Radiation and Immunotherapy. JCEM Case Rep. 2025;3:luaf015.
Li G, Hu J, Cho C, Cui J, Li A, Ren P, et al. Everolimus combined with PD-1 blockade inhibits progression of triple-negative breast cancer. Cellular signalling [Internet]. 2023 Sept [cited 2025 Mar 1];109. Available from: https://pubmed.ncbi.nlm.nih.gov/37257766/.
Dierks C, Seufert J, Aumann K, Ruf J, Klein C, Kiefer S, et al. Combination of Lenvatinib and Pembrolizumab Is an Effective Treatment Option for Anaplastic and Poorly Differentiated Thyroid Carcinoma. Thyroid. 2021;31:1076–85.
Ahmad U, Islam A, Khan MM, Akhtar J. Nanotechnology-driven Epigenetic Cancer Therapy: Precision Delivery and Sustained Release of DNA Methylation Modulators. Yale J Biol Med. 2025;98:227–35.
Yang CL, Chao YJ, Wang HC, Hou YC, Chen CG, Chang CC, et al. Local ablation of gastric cancer by reconstituted apolipoprotein B lipoparticles carrying epigenetic drugs. Nanomedicine. 2021;37:102450.
Niu Q, Liu Y, Zheng Y, Tang Z, Qian Y, Qi R, et al. Co-delivery of nigericin and decitabine using hexahistidine-metal nanocarriers for pyroptosis-induced immunotherapeutics. Acta Pharm Sin B. 2022;12:4458–71.
Kapoor DU, Patel G, Prajapati BG. Nanomedicine-Based Treatments for Rare and Aggressive Ocular Cancers: Advances in Drug Delivery. Curr Treat Options Oncol. 2025;26:569–86.
Zhao X, Qin X, Wang R, Wang Y, He Y, Wang Y, et al. An Epigenetic Nanoagonist Facilitates T Cell Priming, Recruitment, and Reinvigoration in Tumors Resistant to PD-L1 Therapy. Adv Mater. 2025;e02800.
Tost J, Gut IG. DNA methylation analysis by pyrosequencing. Nat Protoc. 2007;2:2265–75.
Hattori N, Liu YY, Ushijima T. DNA Methylation Analysis. Methods Mol Biol. 2023;2691:165–83.
Schubert L, Mariko ML, Clerc J, Huillard O, Groussin L. MAPK Pathway Inhibitors in Thyroid Cancer: Preclinical and Clinical Data. Cancers (Basel). 2023;15:710.
Subbiah V, Kreitman RJ, Wainberg ZA, Gazzah A, Lassen U, Stein A, et al. Dabrafenib plus trametinib in BRAFV600E-mutated rare cancers: the phase 2 ROAR trial. Nat Med. 2023;29:1103–12.
Weber M, Kersting D, Riemann B, Brandenburg T, Führer-Sakel D, Grünwald F, et al. Enhancing Radioiodine Incorporation into Radioiodine-Refractory Thyroid Cancer with MAPK Inhibition (ERRITI): A Single-Center Prospective Two-Arm Study. Clin Cancer Res. 2022;28:4194–202.
Dorman A, Shendler G, Warshavsky A, Muhanna N, Horowitz G, Furman LK, et al. Surgical Outcomes Following Neoadjuvant-targeted Therapy For Advanced Differentiated Thyroid Cancer-real-world Data. Clin Endocrinol. 2025 Nov;
Busaidy NL, Konda B, Wei L, Wirth LJ, Devine C, Daniels GA, et al. Dabrafenib Versus Dabrafenib + Trametinib in BRAF-Mutated Radioactive Iodine Refractory Differentiated Thyroid Cancer: Results of a Randomized, Phase 2, Open-Label Multicenter Trial. Thyroid. 2022;32:1184–92.
Zhao K, Chen L. Immune checkpoint inhibitors against thyroid cancer. Int J Surg. 2025;111:7346–9.
Basté N, Mora M, Grau JJ. Emerging systemic antitarget treatment for differentiated thyroid carcinoma. Curr Opin Oncol. 2021;33:184–95.
Murugan AK, Qasem E, Al-Hindi H, Alzahrani AS. Analysis of ALK, IDH1, IDH2 and MMP8 somatic mutations in differentiated thyroid cancers. Mol Clin Oncol. 2021;15:210.
Lee AV, Nestler KA, Chiappinelli KB. Therapeutic targeting of DNA methylation alterations in cancer. Pharm Ther. 2024;258:108640.
Vishakha S, Navneesh N, Kurmi BD, Gupta GD, Verma SK, Jain A, et al. An Expedition on Synthetic Methodology of FDA-approved Anticancer Drugs (2018-2021). Anticancer Agents Med Chem. 2024;24:590–626.
Ji X, Liang W, Lv G, Ding C, Lai H, Li L, et al. Efficacy and safety of targeted therapeutics for patients with radioiodine-refractory differentiated thyroid cancer: Systematic review and network meta-analysis. Front Pharm. 2022;13:933648.
Ho AL, Dedecjus M, Wirth LJ, Tuttle RM, Inabnet WB, Tennvall J, et al. Selumetinib Plus Adjuvant Radioactive Iodine in Patients With High-Risk Differentiated Thyroid Cancer: A Phase III, Randomized, Placebo-Controlled Trial (ASTRA). J Clin Oncol. 2022;40:1870–8.
Iyer PC, Dadu R, Gule-Monroe M, Busaidy NL, Ferrarotto R, Habra MA, et al. Salvage pembrolizumab added to kinase inhibitor therapy for the treatment of anaplastic thyroid carcinoma. J Immunother Cancer. 2018;6:68.
Siegel RL, Kratzer TB, Giaquinto AN, Sung H, Jemal A. Cancer statistics, 2025. CA Cancer J Clin. 2025;75:10–45.
Gunda V, Bucur O, Varnau J, Vanden Borre P, Bernasconi MJ, Khosravi-Far R, et al. Blocks to thyroid cancer cell apoptosis can be overcome by inhibition of the MAPK and PI3K/AKT pathways. Cell Death Dis. 2014;5:e1104.
Huang X, Kong G, Li Y, Zhu W, Xu H, Zhang X, et al. Decitabine and 5-azacitidine both alleviate LPS induced ARDS through anti-inflammatory/antioxidant activity and protection of glycocalyx and inhibition of MAPK pathways in mice. Biomed Pharmacother. 2016;84:447–53.
Acknowledgements
The authors acknowledge that all figures were supported by BioRender (https://biorender.com).
Funding
This work was supported by the following grants: Natural Science Foundation of Gansu Province, China [No. 17JR5RA272; No. 22JR5RA923] and Research Fund project of The First Hospital of Lanzhou University [No. Ldyyyn2021-120; No. Ldyyyn2020-98; No. Ldyyyn2021-30].
Author information
Authors and Affiliations
Contributions
Tianying Zhang–Writing–Original Draft, Conceptualization. Hengtong Han–Writing–Review & Editing. Yating Zhang–Supervision. Tingting Zhang–Supervision. Libin Ma–Supervision. Ze Yang–Supervision. Yong-xun Zhao–Writing–Review & Editing, Conceptualization, Supervision. All authors have read and agreed to the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
Not Applicable.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhang, T., Han, H., Zhang, Y. et al. The molecular mechanisms and potential therapeutic implications of the crosstalk between DNA methylation and metabolic reprogramming in thyroid cancer. Cell Death Discov. (2026). https://doi.org/10.1038/s41420-026-02981-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41420-026-02981-8