Abstract
Thermal behavior, molecular aggregation structure, and water repellency for poly{2-[(perfluorooctylethyl)carbamate]ethyl}acrylate (PFAUr-C8) were studied by differential scanning calorimetry (DSC), polarized optical microscopy (POM), wide-angle X-ray diffraction (WAXD), grazing-incidence wide-angle X-ray diffraction (GI-WAXD), contact angle measurements, and X-ray photoelectron spectroscopy (XPS). The impact of the carbamate linker was verified by comparison with poly[2-(perfluorooctyl)ethyl]acrylate (PFA-C8). PFAUr-C8 exhibited two phase transitions at 357 K and 416 K, whereas PFA-C8 showed a single-phase transition at 347 K. Both PFAUr-C8 and PFA-C8 formed a bilayered hexatic smectic-B phase at room temperature. Although the lamellar structure for the fluoroalkyl (Rf) groups was immediately lost in PFA-C8 after disordering of the lateral translational order, PFAUr-C8 maintained a stacked lamellar structure without lateral translational order. PFAUr-C8 exhibited excellent water repellency at room temperature similar to PFA-C8. The outstanding thermal stability of the lamellar structure in PFAUr-C8 is attributed to the cohesive interaction of the carbamate linkage.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Volkov VV, Platé NA, Takahara A, Kajiyama T, Amaya N, Murata Y. Aggregation state and mesophase structure of comb-shaped polymers with fluorocarbon side groups. Polymer. 1992;33:1316–20.
Pittman AG, Ludwig BA. Effect of polymer crystallinity on the wetting properties of certain fluoroalkyl acrylates. J Polym Sci A-1 Polym Chem. 1969;7:3053–66.
Smith DW, Lacono ST, Lyer SS. Handbook of fluoropolymer science and technology. 1st ed. Hoboken, New Jersey, USA: John Wiley & Sons, Inc.; 2014.
Shibasaki Y, Zhu Z, Fukuda K. Molecular organization of comb-like polymers containing long fluorocarbon chains in the thin films formed by the LB and lift-up methods. Mol Cryst Liq Cryst Sci Technol Sect A. 1999;337:165–8.
Hasegawa T. Physicochemical nature of perfluoroalkyl compounds induced by fluorine. Chem Rec. 2017;17:903–17.
Banerjee S, Tawade BV, Ladmiral V, Dupuy LX, MacDonald MP, Améduri B. Poly(fluoroacrylate)s with tunable surface hydrophobicity via radical copolymerization of 2, 2, 2-trifluoroethyl α-fluoroacrylate and 2-(trifluoromethyl) acrylic acid. Polym Chem. 2017;8:1978–88.
Oikawa Y, Saito T, Yamada S, Sugiya M, Sawada H. Preparation and surface property of fluoroalkyl end-capped vinyltrimethoxysilane oligomer/talc composite-encapsulated organic compounds: application for the separation of oil and water. ACS Appl Mater Interfaces. 2015;7:13782–93.
Ishige R, Shinohara T, White KL, Meskini A, Raihane M, Takahara A, et al. Unique difference in transition temperature of two similar fluorinated side chain polymers forming hexatic smectic phase: poly{2-(perfluorooctyl) ethyl acrylate} and poly{2-(perfluorooctyl) ethyl vinyl ether}. Macromolecules. 2014;47:3860–70.
Honda K, Yakabe H, Koga T, Sasaki S, Sakata O, Otsuka H, et al. Molecular aggregation structure of poly(fluoroalkyl acrylate) thin films evaluated by synchrotron-sourced grazing-incidence X-ray diffraction. Chem Lett. 2005;34:1024–5.
Roussel F, Saidi S, Guittard F, Geribaldi S. Thermophysical properties of fluorinated acrylate homopolymers: mixing and phase separation. Eur Phys J E. 2002;8:283–8.
Honda K, Morita M, Otsuka H, Takahara A. Molecular aggregation structure and surface properties of poly (fluoroalkyl acrylate) thin films. Macromolecules. 2005;38:5699–705.
Honda K, Morita M, Sakata O, Sasaki S, Takahara A. Effect of surface molecular aggregation state and surface molecular motion on wetting behavior of water on poly(fluoroalkyl methacrylate) thin films. Macromolecules. 2010;43:454–60.
Honda K, Yamamoto I, Morita M, Yamaguchi H, Arita H, Ishige R, et al. Effect of α-substituents on molecular motion and wetting behaviors of poly(fluoroalkyl acrylate) thin films with short fluoroalkyl side chains. Polymer. 2014;55:6303–8.
Ramharack R, Nguyen TH. Fluoropolymers of very low surface energies. J Polym Sci Part C Polym Lett. 1987;25:93–98.
Saïdi S, Guittard F, Guimon C, Géribaldi S. Fluorinated comblike homopolymers: the effect of spacer lengths on surface properties. J Polym Sci A. 2005;43:3737–47.
Corpart JM, Girault S, Juhué D. Structure and surface properties of liquid crystalline fluoroalkyl polyacrylates: role of the spacer. Langmuir. 2001;17:7237–44.
Lindstron AB, Strynar MJ, Libelo EL. Polyfluorinated compoundsː past, present, and future. Environ Sci Technol. 2011;45:7954–61.
Wang Z, Dewitt JC, Higgins CP, Cousins IT. A never-ending story of per- and polyfluoroalkyl substances (PFASs). Environ Sci Technol. 2017;51:2508–18.
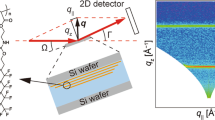
Ogawa H, Masunaga H, Sasaki S, Goto S, Tanaka T, Seike T, et al. Experimental station for multiscale surface structural analyses of soft-material films at SPring-8 via a GISWAX/GIXD/XR-integrated system. Polym J. 2013;45:109–16.
Masunaga H, Ogawa H, Takano T, Sasaki S, Goto S, Tanaka T, et al. Multipurpose softmaterial SAXS/WAXS/GISAXS beamline at SPring-8. Polym J. 2011;43:471–7.
Hatsui T, Omodani M, Kudo T, Kobayashi K, Imamura T, Ohmoto T, et al. A direct-detection X-ray CMOS image sensor with 500 µm thick high resistivity silicon. Proc. Int. Image Sensor Workshop. 2013, 3.05.
Hatsui T, Graafsma H. X-ray imaging detectors for synchrotron and XFEL sources. IUCr J. 2015;2:371–83.
Wang H, Aubuchon SR, Thompson DG, Osborn JC, Marsh AL, Nichols WR, et al. Temperature-dependent dynamic mechanical analysis−Fourier transform infrared study of a poly(ester urethane) copolymer. Macromolecules. 2002;35:8794–801.
Brunette CM, Hsu SL, MacKnight WJ. Hydrogen-bonding properties of hard-segment model compounds in polyurethane block copolymers. Macromolecules. 1982;15:71–77.
Kassis CM, Steehler JK, Betts DE, Guan ZB, Romack TJ, Desimone JM, et al. XPS studies of fluorinated acrylate polymers and block copolymers with polystyrene. Macromolecules. 1996;29:3247–54.
Heide, PVD. X-ray photoelectron spectroscopy. 1st ed. Hoboken, New Jersey, USA: John Wiley & Sons, Inc.; 2012.
Nojima S, Shinohara T, Higaki Y, Ishige R, Ohishi T, Kobayashi D, et al. Precise characterization of outermost surface of crystalline-crystalline diblock copolymer thin films using synchrotron radiation soft X-ray photoelectron spectroscopy. Polym J. 2014;46:637–40.
Shinohara T, Higaki Y, Nojima S, Masunaga H, Ogawa H, Okamoto Y, et al. Molecular aggregation states and wetting behavior of a poly{2-(perfluorooctyl) ethyl acrylate} brush-immobilized nano-imprinted surface. Polymer. 2015;69:10–16.
Saïdi S, Guittard F, Guimon C, Géribaldi S. Synthesis and characterization of copolymers based on styrene and partially fluorinated acrylates. Eur Polym J. 2006;42:702–10.
Cecchet F, Pilling M, Hevesi L, Schergna S, Wong JKY, Clarkson GJ, et al. Grafting of benzylic amide macrocycles onto acid-terminated self-assembled monolayers studied by XPS, RAIRS, and contact angle measurements. J Phys Chem B. 2003;107:10863–72.
Thünemann AF, Lieske A, Paulke BR. Low surface energy coatings from waterborne nano-dispersions of polymer complexes. Adv Mater. 1999;11:321–4.
Acknowledgements
YL wishes to acknowledge the financial support from the China Scholarship Council (Grant Number 201608525036), Guizhou Science and Technical Foundation (Grant Number 20152124), Guizhou Oversea Talent Project (Qianren Xiangmu Zizhu Hetong Number 201414), and the Science Foundation of Guizhou Normal College (Guishiyuan Xiangmu Zizhu Hetong Number 14BS014). This work was supported by the Photon and Quantum Basic Research Coordinated Development Program of the Ministry of Education, Culture, Sports, Science and Technology, Japan. This work was supported by ImPACT Program of Council for Science, Technology, and Innovation (Cabinet Office, Government of Japan). This work was performed under the Cooperative Research Program “Network Joint Research Center for Materials and Devices”. This work was also supported by the MEXT Project “Integrated Research Consortium on Chemical Sciences.” The powder WAXD and GIXD experiments were performed at BL05XU, BL40B2, and BL03XU in SPring-8 with the approval of the Japan Synchrotron Radiation Research Institute (JASRI) (Proposal Numbers 2017B1086 and 2018A1177). We gratefully acknowledge Dr. Kazuki Mita and Mr. Kiminori Uchida (Mitsui Chemicals, Inc.) for kindly giving us the opportunity to perform thin film WAXD measurements at the BL03XU. XPS measurement was performed at BL12 in the Kyushu Synchrotron Light Research Center (Proposal Number 1612115F and 1707063F).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing financial interest.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Liu, Y., Higaki, Y., Mukai, M. et al. Smectic ordered structure and water repellency of a poly(fluoroalkyl acrylate) with a carbamate linker. Polym J 51, 189–198 (2019). https://doi.org/10.1038/s41428-018-0139-2
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41428-018-0139-2