Abstract
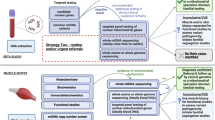
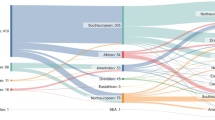
Genomic sequencing has transformed the diagnostic approach for mitochondrial disease, yet integration into standard clinical practice is limited by access and funding. We conducted a post-implementation evaluation of genome sequencing (GS) for mitochondrial disease in Australia, which became publicly funded through the Medicare Benefits Scheme (MBS) in November 2023, to allow for broader access to testing. Test request data, including demographics, phenotypic information, and the diagnostic outcomes, were collected from November 2023 to May 2025 from the Victorian Clinical Genetics Services, the current laboratory provider of the MBS-funded service. Test uptake was 26% of predicted, with lower test rates in regional and remote areas. Over the first 19 months, 300 individuals suspected of mitochondrial disease underwent GS with a median turnaround time of 84 days (8 days–218 days). The diagnostic yield was 20%, with 56% of diagnoses in known mitochondrial disease genes. Of these, 70% (24 of 34) were in mitochondrial DNA. Seventeen diagnoses were in individuals who had prior non-diagnostic testing (exome sequencing or gene panel). We demonstrate that publicly-funded GS can deliver meaningful diagnostic outcomes for mitochondrial disease on a national scale. To maximise its impact, attention must now shift towards ensuring equitable access, particularly for regional and remote areas, and embedding sustainable mainstreaming models that support both genetic and non-genetic clinicians.
Similar content being viewed by others
Data availability
The variants identified in this study have been submitted to ClinVar (Submission IDs: SUB15793101, SUB15793127, SUB15796069, SUB15768047). All submitted data are publicly available.
References
Gorman GS, Chinnery PF, Dimauro S, Hirano M, Koga Y, McFarland R, et al. Mitochondrial diseases. Nat Rev Dis Primers. 2016;2:16080.
Thompson JLP, Karaa A, Pham H, Yeske P, Krischer J, Xiao Y, et al. The evolution of the mitochondrial disease diagnostic odyssey. Orphanet J Rare Dis. 2023;18:157.
Heath O, Hammerl E, Spitzinger A, Wortmann SB. Ending an Odyssey? The psychosocial experiences of parents after the genetic diagnosis of a mitochondrial disease in children. J Pers Med. 2024;14:523.
Mavraki E, Labrum R, Sergeant K, Alston CL, Woodward C, Smith C, et al. Genetic testing for mitochondrial disease: the United Kingdom best practice guidelines. Eur J Hum Genet. 2023;31:148–63.
Stenton SL, Prokisch H. Genetics of mitochondrial diseases: identifying mutations to help diagnosis. EBioMedicine. 2020;56:102784.
Long JC, Gul H, McPherson E, Best S, Augustsson H, Churruca K, et al. A dynamic systems view of clinical genomics: a rich picture of the landscape in Australia using a complexity science lens. BMC Med Genom. 2021;14:63.
Mackley MP, Richer J, Guerin A, Caluseriu O, Armstrong L, Blood KA, et al. Mainstreaming of clinical genetic testing: a conceptual framework. Genet Med. 2025;27:101465.
Ma A, Newing TP, O’Shea R, Gokoolparsadh A, Murdoch E, Hayward J, et al. Genomic multidisciplinary teams: a model for navigating genetic mainstreaming and precision medicine. J Paediatr Child Health. 2024;60:118–24.
Mallett AJ, Ingles J, Goranitis I, Stark Z. Implementation of reimbursement for genomic testing in Australia: early successes and the pathway ahead. Intern Med J. 2024;54:531–4.
Munnich A, Rustin P. Clinical spectrum and diagnosis of mitochondrial disorders. Am J Med Genet. 2001;106:4–17.
Ball M, Bouffler SE, Barnett CB, Freckmann M-L, Hunter MF, Kamien B, et al. Critically unwell infants and children with mitochondrial disorders diagnosed by ultra-rapid genomic sequencing. Genet Med. 2024;27:101293.
Department of Health and Aged Care. Item 73456 Canberra, Australia: medicare benefits schedule; 2023 [updated November 1, 2023. Available from: https://www9.health.gov.au/mbs/fullDisplay.cfm?type=item&q=73456&qt=item&<div class="pi">show [QJ]criteria=73456.
Department of Health and Aged Care. Item 73457 Canberra, Australia: medicare benefits schedule; 2023 [updated November 1, 2023. Available from: https://www9.health.gov.au/mbs/fullDisplay.cfm?type=item&q=73457.
Committee MSA. 1675–Whole genome sequencing for the diagnosis of mitochondrial disease: Commonwealth of Australia; 2022 [updated November 2022. Available from: https://www.msac.gov.au/applications/1675.
Lunke S, Bouffler SE, Patel CV, Sandaradura SA, Wilson M, Pinner J, et al. Integrated multi-omics for rapid rare disease diagnosis on a national scale. Nat Med. 2023;29:1681–91.
MCRI Bioinfomethods Group. Mitoreport Github: Github repository; 2021 [Available from: https://github.com/bioinfomethods/mitoreport/releases.
Richards S, Aziz N, Bale S, Bick D, Das S, Gastier-Foster J, et al. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med. 2015;17:405–24.
McCormick EM, Lott MT, Dulik MC, Shen L, Attimonelli M, Vitale O, et al. Specifications of the ACMG/AMP standards and guidelines for mitochondrial DNA variant interpretation. Hum Mutat. 2020;41:2028–57.
Robinson PN, Kohler S, Bauer S, Seelow D, Horn D, Mundlos S. The Human Phenotype Ontology: a tool for annotating and analyzing human hereditary disease. Am J Hum Genet. 2008;83:610–5.
Services Australia. medicare item reports (MBS Item) [Available from: https://medicarestatistics.humanservices.gov.au/statistics/mbs_item.html.
Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG. Research electronic data capture (REDCap)–a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inf. 2009;42:377–81.
Team RC. R: A language and environment for statistical computing. Vienna: R Foundation for Statistical Computing; 2023.
Statistics ABo. Australian Statistical Geography Standard (ASGS), Edition 3. Canberra: Australian Bureau of Statistics; 2021.
Casauria S, Collins F, White SM, Konings P, Wallis M, Pachter N, et al. Assessing the unmet needs of genomic testing in Australia: a geospatial exploration. Eur J Hum Genet. 2025;33:496–503.
Ellard S, Morgan S, Wynn SL, Walker S, Parrish A, Mein R, et al. Rare disease genomic testing in the UK and Ireland: promoting timely and equitable access. J Med Genet. 2024;61:1103–12.
Goranitis I, Hayeems RZ, Smith HS, Buchanan J, Weymann D, Regier DA, et al. Determining the value of genomics in healthcare. Nat Med. 2025;31:4022–33.
Mordaunt DA, Dalziel K, Goranitis I, Stark Z. Uptake of funded genomic testing for syndromic and non-syndromic intellectual disability in Australia. Eur J Hum Genet. 2023;31:977–9.
Mordaunt DA, Stark Z, Elshaug AG, Schilling C. Genomic testing in Australia: a budget impact analysis using diffusion modeling from a healthcare system perspective. Value Health. 2025. https://doi.org/10.1016/j.jval.2025.09.3067.
Schilling C, Sjaaf F, Goranitis I, Dalziel K, Martyn M, Stark Z, et al. Utilisation of subsidised genetic and genomic testing in a publicly funded healthcare system 2014–2023. Eur J Hum Genet. 2025;33:1044–50.
Mackley MP, Agrawal PB, Ali SS, Archibald AD, Dawson-McClaren B, Ellard H, et al. Genomic sequencing technologies for rare disease in mainstream healthcare: the current state of implementation. Eur J Hum Genet. 2025;33:1402–13.
Dawson-McClaren B, Martyn M, Ince J, Jan A, Brown NJ, Fahey MC, et al. Opportunities and challenges for paediatricians requesting funded genomic tests for children. Eur J Hum Genet. 2025;33:1173–79.
Stark Z, Boughtwood T, Haas M, Braithwaite J, Gaff CL, Goranitis I, et al. Australian genomics: outcomes of a 5-year national program to accelerate the integration of genomics in healthcare. Am J Hum Genet. 2023;110:419–26.
Mackley MP, Weisz E, Hayeems RZ, Gaff C, Dawson-McClaren B. Non-geneticist champions are essential to the mainstreaming of genomic medicine. Eur J Hum Genet. 2025;33:975–7.
Nisselle A, Terrill B, Janinski M, Martyn M, Jordan H, Kaunein N, et al. Ensuring best practice in genomics education: a theory- and empirically informed evaluation framework. Am J Hum Genet. 2024;111:1497–507.
Nisselle A, King EA, McClaren B, Janinski M, Metcalfe S, Gaff C. Measuring physician practice, preparedness and preferences for genomic medicine: a national survey. BMJ Open. 2021;11:e044408.
Macken WL, Falabella M, McKittrick C, Pizzamiglio C, Ellmers R, Eggleton K, et al. Specialist multidisciplinary input maximises rare disease diagnoses from whole genome sequencing. Nat Commun. 2022;13:6324.
Schon KR, Horvath R, Wei W, Calabrese C, Ibanez K, Ratnaike T, et al. Use of whole genome sequencing to determine the genetic basis of suspected mitochondrial disorders: cohort study. BMJ. 2021;375:e066288.
Davis RL, Kumar KR, Puttick C, Liang C, Ahmad KE, Edema-Hildebrand F, et al. Use of whole-genome sequencing for mitochondrial disease diagnosis. Neurology. 2022;99:e730–e42.
Rius R, Compton AG, Baker NL, Balasubramaniam S, Best S, Bhattacharya K, et al. The Australian Genomics Mitochondrial Flagship: a national program delivering mitochondrial diagnoses. Genet Med. 2025;27:101271.
Riley LG, Cowley MJ, Gayevskiy V, Minoche AE, Puttick C, Thorburn DR, et al. The diagnostic utility of genome sequencing in a pediatric cohort with suspected mitochondrial disease. Genet Med. 2020;22:1254–61.
Heath O, Feichtinger RG, Achleitner MT, Hofbauer P, Mayr D, Merkevicius K, et al. Mitochondrial disorder diagnosis and management - what the pediatric neurologist wants to know. Eur J Paediatr Neurol. 2025;54:75–88.
Kaschta D, Post C, Gaass F, Al-Tawil M, Arriens V, Balachandran S, et al. Evaluating genome sequencing strategies: trio, singleton, and standard testing in rare disease diagnosis. Genome Med. 2025;17:100.
Grady JP, Pickett SJ, Ng YS, Alston CL, Blakely EL, Hardy SA, et al. mtDNA heteroplasmy level and copy number indicate disease burden in m.3243A>G mitochondrial disease. EMBO Mol Med. 2018;10:e8262.
Wojcik MH, Lemire G, Berger E, Zaki MS, Wissmann M, Win W, et al. Genome sequencing for diagnosing rare diseases. N Engl J Med. 2024;390:1985–97.
Nurchis MC, Radio FC, Salmasi L, Heidar Alizadeh A, Raspolini GM, Altamura G, et al. Cost-effectiveness of whole-genome vs whole-exome sequencing among children with suspected genetic disorders. JAMA Netw Open. 2024;7:e2353514.
Welland MJ, Ahlquist KD, De Fazio P, Austin-Tse C, Pais L, Wedd L, et al. Scalable automated reanalysis of genomic data in research and clinical rare disease cohorts. Preprint at. https://doi.org/10.1101/2025.05.19.25327921 2025.
Rath S, Sharma R, Gupta R, Ast T, Chan C, Durham TJ, et al. MitoCarta3.0: an updated mitochondrial proteome now with sub-organelle localization and pathway annotations. Nucleic Acids Res. 2021;49:D1541–D7.
Zhao T, Hock DH, Pitt J, Thorburn DR, Stroud DA, Christodoulou J. Review: utility of mass spectrometry in rare disease research and diagnosis. npj Genom Med. 2025;10:29.
Kremer LS, Bader DM, Mertes C, Kopajtich R, Pichler G, Iuso A, et al. Genetic diagnosis of Mendelian disorders via RNA sequencing. Nat Commun. 2017;8:15824.
Hock DH, Compton AG, Amarasekera SSC, Frazier AE, Helman G, Lake NJ, et al. Mainstreaming proteomics into rare disease diagnostics. Pathology. 2022;54:S17.
Acknowledgements
The authors would like to acknowledge Victorian Clinical Genetics Services for their contribution to this research.
Funding
MB is supported by a Melbourne Academic Centre for Health Clinician—Researcher Scholarship and a Mito Foundation PhD Top-Up Scholarship. The research conducted at the Murdoch Children’s Research Institute (MCRI) was supported by the Victorian Government’s Operational Infrastructure Support Program. The Chair in Genomic Medicine awarded to JC is generously supported by The Royal Children’s Hospital Foundation. Open Access funding enabled and organized by CAUL and its Member Institutions.
Author information
Authors and Affiliations
Contributions
Conceptualisation—MB and ZS; Data curation—MB and SL; Formal analysis—MB and SC; Investigation/Methodology—AGC, DRT, JC, MB, NB, SC, SCL, SL, and ZS; Supervision—AGC, DRT, JC, and ZS; Writing-original draft— MB; Writing-review and editing—all authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics
This study was conducted in accordance with the revised Declaration of Helsinki and following the Australian National Health and Medical Research Council statement of ethical conduct in research involving humans. This study has Human Research Ethics Committee approval (QA/104560/RCHM-2024).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ball, M., Baker, N., Lim, S.C. et al. Mainstreaming genomic testing for mitochondrial disease in Australia. Eur J Hum Genet (2026). https://doi.org/10.1038/s41431-026-02053-6
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41431-026-02053-6