Fig. 1: Schematic diagram depicting the bottlenecks of conventional CL and new catalyst design strategy.
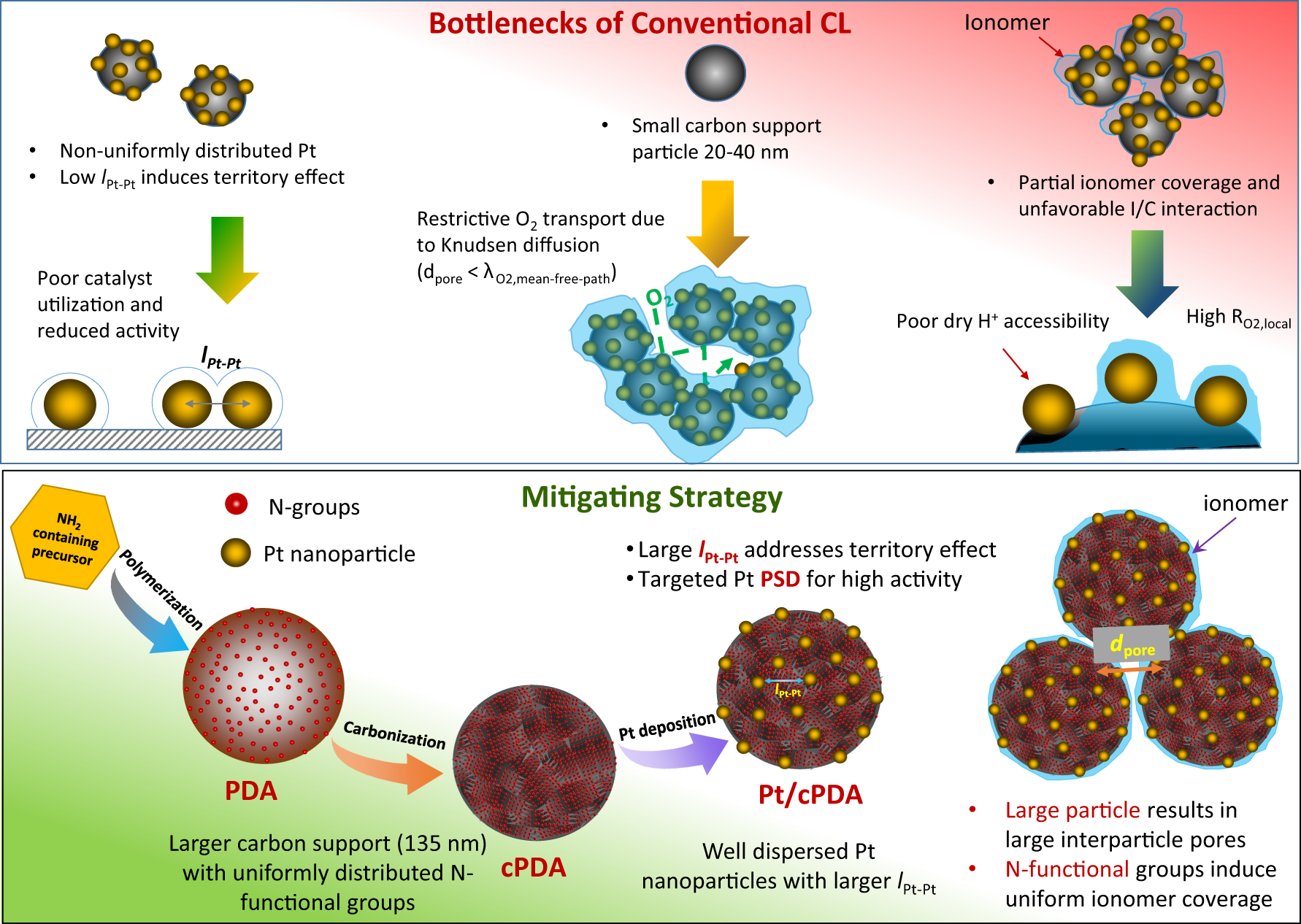
Top Panel: In conventional Pt/C catalysts, Pt nanoparticles are distributed non-uniformly due to the non-uniform surface functional group, which results in low platinum interparticle distance (lPt-Pt) inducing territory effect that leads to reduced mass specific activity and poor catalyst utilization at high current densities. The usage of small carbon particles (~20-40 nm) results in catalyst layers with small pore diameter (dpore), which leads to restrictive gas-phase oxygen transport dominated by Knudsen diffusion. Lack of surface functional groups or non-uniform distribution of surface functional groups result in non-uniform ionomer coverge of the Pt/C catalyst, which further limit the proton accessibility of Pt catalyst under low RH (dry) conditions. Bottom Panel: To mitigate these issues, a NH2-containing small molecule is polymerized to create large spherical particles with well-distributed N-functional group on the particle surface. The particle is carbonized prior to Pt deposition. The uniformly distributed N-functional groups on carbonized particles enables deposition of uniformly distributed Pt and size-controlled nanoparticles with large Pt-Pt interparticle distance (lPt-Pt)which addresses territory effect. Large carbon support results in large pores improving gaseous oxygen transport while the uniformly distributed N-functional group hypothesized to have favorable interaction with the ionomer induces high ionomer coverage, which improves catalyst utilization.
