Fig. 1: SIMPLEx-mediated expression of biologically active HsST6Gal1.
From: A universal glycoenzyme biosynthesis pipeline that enables efficient cell-free remodeling of glycans
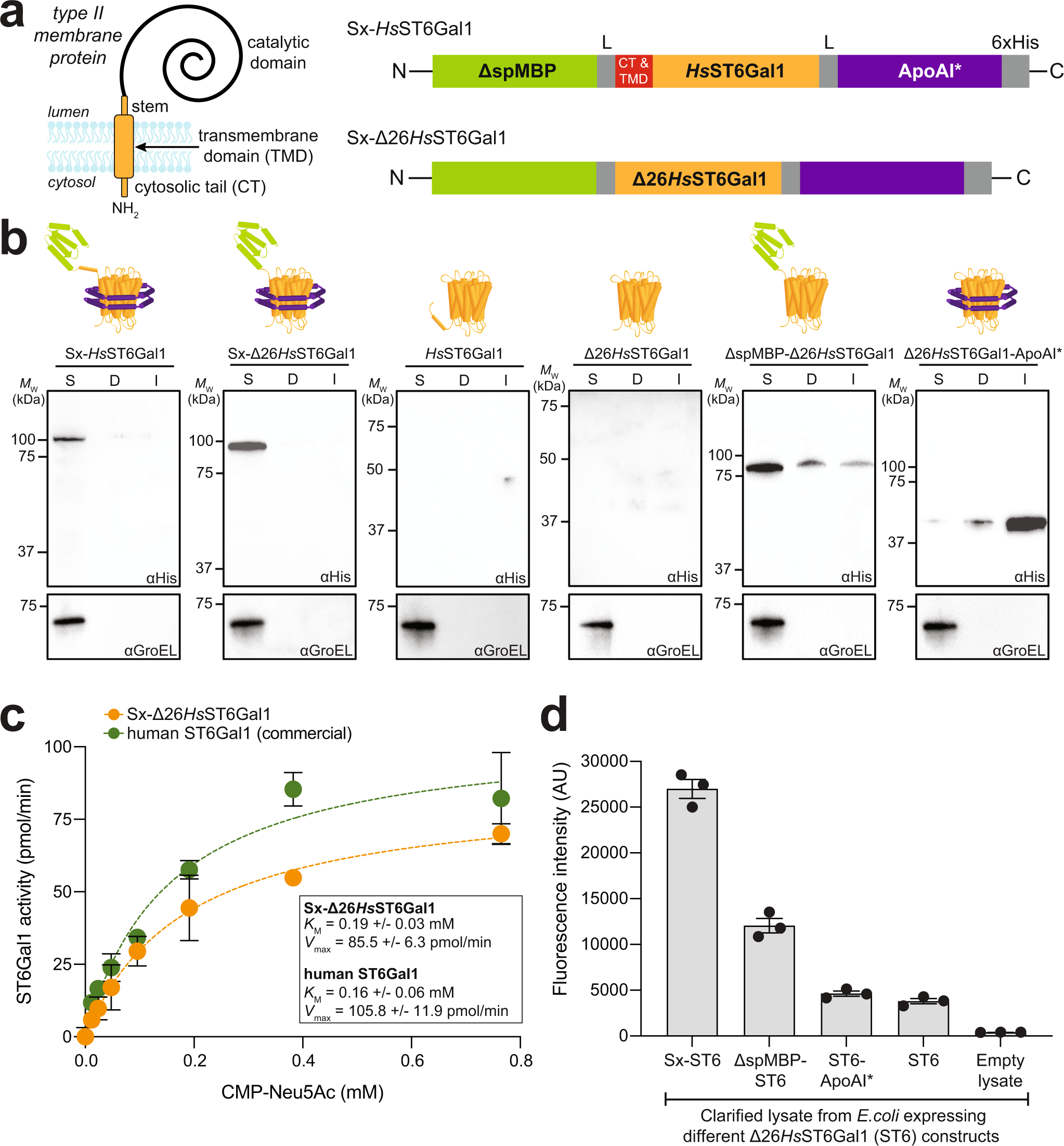
a Membrane topology of type II transmembrane proteins and molecular architecture of SIMPLEx constructs used in this study. Each construct consisted of N-terminal ΔspMBP and C-terminal ApoAI* that flanked HsST6Gal1. Intervening flexible linker (L) connects ΔspMBP and ApoAI* to the GT domains while the 6xHis tag was placed at the C-terminus to facilitate detection and purification. HsST6Gal1 domain variants studied here were wild-type (wt) HsST6Gal1 (top) and truncated Δ26HsST6Gal1 (bottom), in which the cytoplasmic tail (CT) and transmembrane domain (TMD) were removed. b Immunoblot analysis of the soluble (S), detergent-solubilized (D), and insoluble (I) fractions prepared from E. coli SHuffle T7 Express lysY cells carrying plasmid pET28a(+) encoding each of the indicated constructs. An equivalent amount of total protein was loaded in each lane. Blots were probed with anti-polyhistidine antibody (αHis). Control blots were generated by probing with anti-GroEL antibody. Results are representative of three biological replicates. Molecular weight (Mw) markers are shown at left. c Kinetic analysis of purified Sx-Δ26HsST6Gal1 and commercial human ST6Gal1 performed using asialofetuin as acceptor substrate and CMP-Neu5Ac as donor substrate. A standard phosphate curve was generated to convert the initial raw absorbance reading to the enzymatically released inorganic phosphate from CMP-Neu5Ac. Values for Vmax and Km values were determined using Prism 9. Data are the mean of three biological replicates ± SEM. d Functional characterization of sialyltransferase-mediated chemoenzymatic remodeling of protein-linked glycans using bioorthogonal click chemistry-based assay. Fluorescence (501/523 nm ex/em) measured in clarified lysates prepared from E. coli cells expressing: Sx-Δ26HsST6Gal1 (Sx-ST6), ΔspMBP-Δ26HsST6Gal1 (ΔspMBP-ST6), Δ26HsST6Gal1-ApoAI (ST6-ApoAI), or Δ26HsST6Gal1 (ST6), as indicated. Lysates from E. coli cells carrying empty pET28a(+) plasmid were used as a negative control (empty lysate). Fluorescence data, corresponding to the extent of chemoenzymatic modification, are the mean of three biological replicates (starting from freshly transformed cells) ± SEM. Source data are provided as a Source Data file.
