Abstract
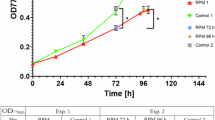
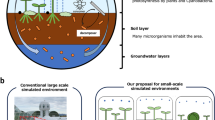
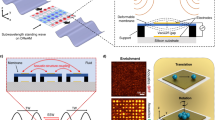
Space missions require sustainable life support systems capable of producing oxygen and biomass under microgravity. We report the use of acoustic levitation to trap and manipulate the filamentous cyanobacterium Limnospira indica PCC 8005 during parabolic flights. Within a millimeter-scale fluidic chamber, this helical microorganism rapidly assembles into thin layers under a standing ultrasonic wave. Stable trapping in microgravity requires substantially less acoustic power (0.42 mW) than on Earth (1.4 mW), highlighting the potential for energy-efficient bioprocessing in space. Monte Carlo simulations and light attenuation modeling show that layered structuring enhances light penetration, potentially overcoming the “compensation point" limitation in bulk cultures. These findings open new perspectives for photobioreactors using acoustic manipulation to boost photosynthetic efficiency and reduce energy demands for oxygen and biomass production in space.
Similar content being viewed by others
Data availability
All relevant data and codes are available upon demand.
References
Gentry, G. J. & Cover, J. International space station (iss) environmental control and life support (ecls) system overview of events: 2010–2014. In 45th International Conference on Environmental Systems (Bellevue, Washington, 2015). https://ntrs.nasa.gov/api/citations/20150022318/downloads/20150022318.pdf.
Witt, J., Hovland, S. & Bockstahler, K. Acls - Europe’s closed loop air revitalisation system. In 40th International Conference on Environmental Systems (2010).
Bockstahler, K., Lucas, J. & Witt, J. Design status of the advanced closed loop system acls for accommodation on the ISS. In 43rd International Conference on Environmental Systems (Vail, Colorado, 2013).
Hendrickx, L. et al. Microbial ecology of the closed artificial ecosystem Melissa (micro-ecological life support system alternative): reinventing and compartmentalizing the earth’s food and oxygen regeneration system for long-haul space exploration missions. Res. Microbiol. 157, 77–86 (2006).
Farges, B., Poughon, L., Creuly, C., Cornet, J.-F. & Lasseur, C. Dynamic aspects and controllability of the Melissa project: a bioregenerative system to provide life support in space. Appl. Biochem. Biotechnol. 151, 686–699 (2008).
Lasseur, C. Melissa: The european project of a closed life support system. In Gravitational and Space Biology (2008).
Walker, J. & Granjou, C. MELiSSA the minimal biosphere: Human life, waste and refuge in deep space. Futures 92, 59–69 (2017).
Poughon, L. et al. Limnospira indica pcc8005 growth in photobioreactor: model and simulation of the ISS and ground experiments. Life Sci. Space Res. 25, 53–65 (2020).
Ciferri, O. Spirulina, the edible microorganism. Microbiol. Rev. 47, 551–578 (1983).
Soreanu, G. et al. Investigation of a biosystem based on Arthrospira platensis for air revitalisation in spacecrafts: Performance evaluation through response surface methodology. Chemosphere 264, 128465 (2021).
Garcia-Gragera, D. et al. Dynamics of long-term continuous culture of limnospira indica in an air-lift photobioreactor. Microb. Biotechnol. 15, 931–948 (2022).
Cogne, G., Cornet, J.-F. & Gros, J.-B. Design, operation, and modeling of a membrane photobioreactor to study the growth of the cyanobacterium arthrospira platensis in space conditions. Biotechnol. Prog. 21, 741–750 (2005).
Badri, H. et al. Temporal gene expression of the cyanobacterium arthrospira in response to gamma rays. PLoS One 10, 1–29 (2015).
Habib, M., Parvin, M., Huntington, T. & Hasan, M. A review on culture, production, and use of spirulina as food for humans and feeds for domestic animals and fish. FAO Fisheries and Aquaculture Circular No. 1034, FAO, Rome, Italy (2008).
Cornet, J. F., Dussap, C. G. & Dubertret, G. A structured model for simulation of cultures of the cyanobacterium spirulina platensis in photobioreactors: I. coupling between light transfer and growth kinetics. Biotechnol. Bioeng. 40, 817–825 (1992).
Cornet, J.-F., Dussap, C. G., Gros, J.-B. Kinetics and energetics of photosynthetic micro-organisms in photobioreactors. Springer Berlin Heidelberg: Berlin, Heidelberg, 1998). 153–224.
Ma, S. et al. Comprehensive modeling and predicting light transmission in microalgal biofilm. J. Environ. Manage. 326, 116757 (2023).
Bruus, H. Acoustofluidics 7: the acoustic radiation force on small particles. Lab Chip 12, 1014–1021 (2012).
Lenshof, A., Evander, M., Laurell, T. & Nilsson, J. Acoustofluidics 5: building microfluidic acoustic resonators. Lab Chip 12, 684–695 (2012).
Barnkob, R., Augustsson, P., Laurell, T. & Bruus, H. Acoustic radiation- and streaming-induced microparticle velocities determined by microparticle image velocimetry in an ultrasound symmetry plane. Phys. Rev. E 86, 056307 (2012).
Evander, M. & Nilsson, J. Acoustofluidics 20: applications in acoustic trapping. Lab Chip 12, 4667–4676 (2012).
Jeger-Madiot, N. et al. Self-organization and culture of mesenchymal stem cell spheroids in acoustic levitation. Sci. Rep. 11, 8355 (2021).
Rabiet, L. et al. Acoustic levitation as a tool for cell-driven self-organization of human cell spheroids during long-term 3d culture. Biotechnol. Bioeng. 121, 1421–1433 (2024).
Bellebon, L., Levant, M., Hoyos, M. & Aider, J.-L. Accelerating particle aggregation in acoustic levitation by collective effects: Application to cost-effective ultrasonic harvesting of microalgae. Phys. Rev. Appl. 23, 014006 (2025).
Yosioka, K. & Kawasima, Y. Acoustic radiation pressure on a compressible sphere. Acustica 5, 167–173 (1955).
Doinikov, A. A. Acoustic radiation pressure on a rigid sphere in a viscous fluid. Proc. R. Soc. Lond. A 447, 447–466 (1994).
Baresch, D., Thomas, J.-L. & Marchiano, R. Three-dimensional acoustic radiation force on an arbitrarily located elastic sphere. J. Acoust. Soc. Am. 133, 25–36 (2013).
Silva, G. T. Acoustic radiation force and torque on an absorbing compressible particle in an inviscid fluid. J. Acoust. Soc. Am. 136, 2405–2413 (2014).
Wei, W., Thiessen, D. B. & Marston, P. L. Acoustic radiation force on a compressible cylinder in a standing wave. J. Acoust. Soc. Am. 116, 201–208 (2004).
Marston, P. L., Wei, W. & Thiessen, D. B. Acoustic radiation force on elliptical cylinders and spheroidal objects in low frequency standing waves. In AIP Conference Proceedings, vol. 838, 495–499 (2006).
Mitri, F. Acoustic radiation force on a rigid elliptical cylinder in plane (quasi) standing waves. J. Appl. Phys. 118, 214903 (2015).
Dumy, G. et al. Acoustic manipulation of dense nanorods in microgravity. Microgravity Sci. Technol. 32, 1159–1174 (2020).
Leao-Neto, J. P., Hoyos, M., Aider, J.-L. & Silva, G. T. Acoustic radiation force and torque on spheroidal particles in an ideal cylindrical chamber. J. Acoust. Soc. Am. 149, 285–295 (2021).
Böhm, C., Jenke, D. & Gerlach, J. Quantification of a novel h-shaped ultrasonic resonator for separation of biomaterials under terrestrial gravity and microgravity conditions. Biotechnol. Bioeng. 82, 74–85 (2003).
Dron, O. & Aider, J.-L. Varying the agglomeration position of particles in a micro-channel using acoustic radiation force beyond the resonance condition. Ultrasonics 53, 1280–1287 (2013).
Dumy, G., Hoyos, M. & Aider, J.-L. Observation of selective optical manipulation of particles in acoustic levitation. J. Acoust. Soc. Am. 146, 4557–4568 (2019).
Whitworth, G. & Coakley, W. Particle column formation in a stationary ultrasonic field. J. Acoust. Soc. Am. 91, 79–85 (1992).
Barnkob, R., Augustsson, P., Laurell, T. & Bruus, H. Measuring the local pressure amplitude in microchannel acoustophoresis. Lab Chip 10, 563–570 (2010).
Koklu, M., Sabuncu, A. C. & Beskok, A. Acoustophoresis in shallow microchannels. J. Colloid Interface Sci. 351, 407–414 (2010).
Hawkes, J., Cefai, J., Barrow, D., Coakley, W. & Briarty, L. Ultrasonic manipulation of particles in microgravity. J. Phys. D Appl. Phys. 31, 1673–1680 (1998).
Hill, M. The selection of layer thicknesses to control acoustic radiation force profiles in layered resonators. J. Acoust. Soc. Am. 114, 2654–2661 (2003).
Hill, M., Townsend, R. J. & Harris, N. R. Modelling for the robust design of layered resonators for ultrasonic particle manipulation. Ultrasonics 48, 521–528 (2008).
Jeger-Madiot, N. et al. Controlling the force and the position of acoustic traps with a tunable acoustofluidic chip: Application to spheroid manipulations. J. Acoust. Soc. Am. 151, 4165–4179 (2022).
Lecoq, P.-E. et al. Influence of microgravity on spontaneous calcium activity of primary hippocampal neurons grown in microfluidic chips. npj Microgravity 10, 15 (2024).
Schindelin, J. et al. Fiji: an open-source platform for biological-image analysis. Nat. Methods 9, 676–682 (2012).
Laurell, T. & Lenshof, A. Microscale Acoustofluidics (Royal Society of Chemistry, 2014).
Leclercq, D. et al. Controlling cyanobacteria with ultrasound. In Proceedings of the 43rd International Congress on Noise Control Engineering (INTER-NOISE 2014), 4457–4466 (Melbourne, Australia, 2014).
Marti, D., Aasbjerg, R. N., Andersen, P. E. & Hansen, A. K. Mcmatlab: an open-source, user-friendly, MATLAB-integrated three-dimensional Monte Carlo light transport solver with heat diffusion and tissue damage. J. Biomed. Opt. 23, 121622 (2018).
Cornet, J.-F. Procédés limités par le transfert de rayonnement en milieu hétérogène https://hal.science/tel-01281239 (2007).
Ellena, G. et al. Development and implementation of a simulated microgravity setup for edible cyanobacteria. npj Microgr.10, 1–14 (2024).
Silva, G. T. & Drinkwater, B. W. Acoustic radiation force exerted on a small spheroidal rigid particle by a beam of arbitrary wavefront: examples of traveling and standing plane waves. J. Acoust. Soc. Am. 144, EL453–EL459 (2018).
Wang, W., Castro, L. A., Hoyos, M. & Mallouk, T. E. Autonomous motion of metallic microrods propelled by ultrasound. ACS nano 6, 6122–6132 (2012).
Zarrouk, C. Contribution à l’étude d’une cyanophycée: Influence de divers facteurs physiques et chimiques sur la croissance et la photosynthèse de Spirulina maxima. Ph.D. thesis, University of Paris (1966).
Acknowledgements
The authors wish to thank ESA and the Carnot IPGG for funding the PhD thesis of B. Dupont. The participation in the parabolic flights campaign was financially supported by the CNES (French Aerospace Agency). The authors thank Corinne and Franck Chauvat (CEA Saclay) for their advises on cyanobacterial culture and insightful discussions. The authors thank G. Gauquelin-Koch (in charge of the Life Science topic at the CNES) and T. Bret-Dibat (in charge of the Matter Science topic at the CNES). The authors also thank T. Paris (Novespace) for his help in optimizing the experimental setup to make it fit the constraints of the Airbus Air Zero-G. The authors finally thank J.-M. Peyrin (Neurosciences Paris Seine, Sorbonne Université) and P.-E. Lecoq (NPS and PMMH) for their help during the preparation of the setup and the flights.
Author information
Authors and Affiliations
Contributions
B.D., X.B.-G., J-L.A. and M.A.: conception and fabrication of the setup. B.D., J.-L.A. and M.A.: conceptualization and methodology. B.D., J.-L.A. and M.A.: samples preparation, data acquisition, analysis and simulations. B.D., S.B.-V., J.-L.A. and M.A.: writing original draft, review and editing. S.B.-V., J.L.A. and M.A.: supervised the project. All authors contributed to the article and approved the submitted version.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Dupont, B., Benoit-Gonin, X., Vincent-Bonnieu, S. et al. Illumination optimization and low-power trapping of Limnospira indica PCC 8005 using bulk acoustic waves in microgravity. npj Microgravity (2026). https://doi.org/10.1038/s41526-025-00553-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41526-025-00553-1