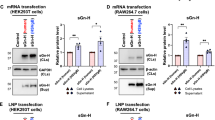
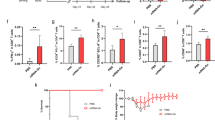
Abstract
Current lipid nanoparticles (LNPs) used in mRNA vaccines typically incorporate polyethylene glycol (PEG) as a stabilizing material; however, safety concerns have prompted the search for non-PEG alternatives. Severe Fever with Thrombocytopenia Syndrome (SFTS) is a life-threatening zoonotic disease with a case fatality rate of up to 30%, and its incidence is rising in East Asia, including China, South Korea, and Japan. Despite its growing public health impact, no licensed vaccine or specific antiviral therapy is available, highlighting the urgent need for vaccine development. Human serum albumin (HSA) is a promising stabilizer, yet an HSA-based LNP mRNA vaccine has not been reported. Here, we developed SFTSV HSAmvac, an HSA-LNP mRNA vaccine encoding a modified Gn protein of SFTSV. Immunization of BALB/c and C57BL/6 mice elicited robust humoral and cellular immune responses, and vaccination of IFNAR–/– mice provided complete protection against a lethal challenge, demonstrating the potential of HSA-LNP as a safe and effective mRNA vaccine delivery platform.
Similar content being viewed by others
Data availability
The SFTS Gn sequence generated in this study has been deposited in GenBank under accession number PX394566. All other data supporting the findings of this study are included within the article and its Supplementary Information. Additional relevant data are available from the corresponding author upon reasonable request.
References
Yu, X. J. et al. Fever with thrombocytopenia associated with a novel bunyavirus in China. N. Engl. J. Med 364, 1523–1532 (2011).
Ding, Y. P. et al. Prognostic value of clinical and immunological markers in acute phase of SFTS virus infection. Clin. Microbiol. Infect. 20, O870–O878 (2014).
Casel, M. A., Park, S. J. & Choi, Y. K. Severe fever with thrombocytopenia syndrome virus: emerging novel phlebovirus and their control strategy. Exp. Mol. Med. 53, 713–722 (2021).
Tran Quang, K. et al. Study on the co-infection of children with severe community-acquired pneumonia. Pediatr. Int. 64, e14853. https://doi.org/10.1111/ped.14853 (2022).
Rattanakomol, P. et al. Severe fever with thrombocytopenia syndrome virus infection, Thailand, 2019-2020. Emerg. Infect. Dis. 28, 2572–2574 (2022).
Charoensakulchai, S., Matsuno, K., Nakayama, E. E., Shioda, T. & Imad, H. A. Epidemiological characteristics of severe fever with thrombocytopenia syndrome. Am. J. Trop. Med. Hyg. 112, 956–962 (2025).
Zhang, X. et al. Rapid spread of severe fever with thrombocytopenia syndrome virus by parthenogenetic Asian longhorned ticks. Emerg. Infect. Dis. 28, 363–372 (2022).
Kaneko, C. et al. Seroprevalence of severe fever with thrombocytopenia syndrome virus in medium-sized wild mammals in Miyazaki, Japan. Ticks Tick. Borne Dis. 14, 102115. https://doi.org/10.1016/j.ttbdis.2022.102115 (2023).
Lee, S. H. et al. Molecular detection and phylogenetic analysis of severe fever with thrombocytopenia syndrome virus in shelter dogs and cats in the Republic of Korea. Ticks Tick. Borne Dis. 8, 626–630 (2017).
WHO. Annual Review of Diseases Prioritized under the Research and Development Blueprint, Informal Consultation (WHO, Geneva, Switzerland, 2018).
National Institute of Allergy and Infectious Diseases (NIAID) Emerging Infectious Diseases/Pathogens. National Institute of Allergy and Infectious Diseases [(accessed on 19 November 2023)] (2018).
Pardi, N., Hogan, M. J., Porter, F. W. & Weissman, D. mRNA vaccines - a new era in vaccinology. Nat. Rev. Drug Discov. 17, 261–279 (2018).
Kim, D., Lai, C. J., Cha, I. & Jung, J. U. Current Progress of Severe Fever with Thrombocytopenia Syndrome Virus (SFTSV) Vaccine Development. Viruses https://doi.org/10.3390/v16010128 (2024).
Li, A. Q. et al. Immunogenicity and protective efficacy of an inactivated SFTS vaccine candidate in mice. Biosaf. Health 4, 45–52 (2022).
Yoshikawa, T. Vaccine development for severe fever with thrombocytopenia syndrome. Viruses https://doi.org/10.3390/v13040627 (2021).
Huang, Q. R. et al. A single-dose mRNA vaccine provides a long-term protection for hACE2 transgenic mice from SARS-CoV-2. Nat. Commun. 12, 2021 (2021).
Shimoyama, T. et al. Potent immunogenicity and neutralization of recombinant adeno-associated virus expressing the glycoprotein of severe fever with thrombocytopenia virus. J. Vet. Med. Sci. 86, 228–238 (2024).
Zhang, L. et al. Characterization of severe fever with thrombocytopenia syndrome in rural regions of Zhejiang, China. PLoS ONE 9, e111127 https://doi.org/10.1371/journal.pone.0111127 (2014).
Xia, T. et al. Glucosylceramide is essential for Heartland and Dabie bandavirus glycoprotein-induced membrane fusion. PLoS Pathog. 19, e1011232 https://doi.org/10.1371/journal.ppat.1011232 (2023).
Halldorsson, S. et al. Shielding and activation of a viral membrane fusion protein. Nat. Commun. 9, 349 https://doi.org/10.1038/s41467-017-02789-2 (2018).
Cifuentes-Muñoz, N., Salazar-Quiroz, N. & Tischler, N. D. Hantavirus Gn and Gc envelope glycoproteins: key structural units for virus cell entry and virus assembly. Viruses 6, 1801–1822 (2014).
Kim, D. et al. Self-assembling Gn head ferritin nanoparticle vaccine provides full protection from lethal challenge of in aged ferrets. mBio 14, e0186823 https://doi.org/10.1128/mbio.01868-23 (2023).
Kim, D. et al. SFTSV Gn-Head mRNA vaccine confers efficient protection against lethal viral challenge. J. Med. Virol. 95, e29203 https://doi.org/10.1002/jmv.29203 (2023).
Kim, J. Y. et al. mRNA vaccine encoding Gn provides protection against severe fever with thrombocytopenia syndrome virus in mice. NPJ Vaccines 8, 167 (2023).
Kim, K. H. et al. An anti-Gn glycoprotein antibody from a convalescent patient potently inhibits the infection of severe fever with thrombocytopenia syndrome virus. PLoS Pathog. 15, e1007375 (2019).
Liu, Q., He, B., Huang, S. Y., Wei, F. & Zhu, X. Q. Severe fever with thrombocytopenia syndrome, an emerging tick-borne zoonosis. Lancet Infect. Dis. 14, 763–772 (2014).
Albertsen, C. H. et al. The role of lipid components in lipid nanoparticles for vaccines and gene therapy. Adv. Drug Del. Rev. 188, 114416 https://doi.org/10.1016/j.addr.2022.114416 (2022).
Reichmuth, A. M., Oberli, M. A., Jaklenec, A., Langer, R. & Blankschtein, D. mRNA vaccine delivery using lipid nanoparticles. Ther. Deliv. 7, 319–334 (2016).
Tenchov, R., Bird, R., Curtze, A. E. & Zhou, Q. Q. Lipid nanoparticles-from liposomes to mRNA vaccine delivery, a landscape of research diversity and advancement. ACS Nano 15, 16982–17015 (2021).
Sellaturay, P., Nasser, S. & Ewan, P. Polyethylene glycol-induced systemic allergic reactions (anaphylaxis). J. Allergy Clin. Immunol. Pract. 9, 670–675 (2021).
Li, C. Y., Zhang, D. G., Pan, Y. J. & Chen, B. Q. Human serum albumin based nanodrug delivery systems: recent advances and future perspective. Polymers 15, 3354 https://doi.org/10.3390/polym15163354 (2023).
Chung, H. J., Kim, H. J. & Hong, S. T. Tumor-specific delivery of a paclitaxel-loading HSA-haemin nanoparticle for cancer treatment. Nanomedicine 23, 102089. https://doi.org/10.1016/j.nano.2019.102089 (2020).
Zhai, C. K. et al. Oral delivery of a host-directed antiviral, niclosamide, as a cholate-coated nanoformulation. Int. J. Antimicrob. Agents 62, 106973 https://doi.org/10.1016/j.ijantimicag.2023.106973 (2023).
Zhai, C. et al. Noncovalent nanoformulation of a drug candidate with limited systemic delivery for systemic oral delivery as much as a typical common oral drug. SSRN Electron. J. https://doi.org/10.2139/ssrn.4189035 (2022).
Guo, X. et al. Human antibody neutralizes severe fever with thrombocytopenia syndrome virus, an emerging hemorrhagic fever virus. Clin. Vaccine Immunol. 20, 1426–1432 (2013).
Attallah, C., Etcheverrigaray, M., Kratje, R. & Oggero, M. A highly efficient modified human serum albumin signal peptide to secrete proteins in cells derived from different mammalian species. Protein Expr. Purif. 132, 27–33 (2017).
Zhang, Y. et al. Efficient signal sequence of mRNA vaccines enhances the antigen expression to expand the immune protection against viral infection. J. Nanobiotechnol. 22, 295 (2024).
Schlake, T., Thess, A., Fotin-Mleczek, M. & Kallen, K. J. Developing mRNA-vaccine technologies. RNA Biol. 9, 1319–1330 (2012).
Kim, S. C. et al. Modifications of mRNA vaccine structural elements for improving mRNA stability and translation efficiency. Mol. Cell Toxicol. 18, 1–8 (2022).
Kirshina, A. et al. Effects of combinations of untranslated-region sequences on translation of mRNA. Biomolecules https://doi.org/10.3390/biom13111677 (2023).
Sun, J. et al. Animal model of severe fever with thrombocytopenia syndrome virus infection. Front. Microbiol. 12, 797189. https://doi.org/10.3389/fmicb.2021.797189 (2021).
Park, S. C. et al. Pathogenicity of severe fever with thrombocytopenia syndrome virus in mice regulated in type I interferon signaling: Severe fever with thrombocytopenia and type I interferon. Lab Anim. Res. 36, 38 (2020).
Shroff, H. COVID-19 vaccine-induced liver injury. Curr. Opin. Gastroenterol. 40, 119–125 (2024).
Bopp, N. E. et al. Baseline mapping of severe fever with thrombocytopenia syndrome virology, epidemiology and vaccine research and development. NPJ Vaccines 5, 111 (2020).
Lu, S. et al. Liver function derangement in patients with severe fever and thrombocytopenia syndrome. J. Clin. Transl. Hepatol. 10, 825–834 (2022).
Teijaro, J. R. Cytokine storms in infectious diseases. Semin Immunopathol. 39, 501–503 (2017).
Gu, Y. et al. The mechanism behind influenza virus cytokine storm. Viruses https://doi.org/10.3390/v13071362 (2021).
England, J. T. et al. Weathering the COVID-19 storm: Lessons from hematologic cytokine syndromes. Blood Rev. 45, 100707. https://doi.org/10.1016/j.blre.2020.100707 (2021).
Sun, Y. et al. Host cytokine storm is associated with disease severity of severe fever with thrombocytopenia syndrome. J. Infect. Dis. 206, 1085–1094 (2012).
Kwon, J. S. et al. Kinetics of viral load and cytokines in severe fever with thrombocytopenia syndrome. J. Clin. Virol. 101, 57–62 (2018).
Korzun, T. et al. Lipid nanoparticles elicit reactogenicity and sickness behavior in mice via toll-like receptor 4 and myeloid differentiation protein 88 axis. ACS Nano 18, 24842–24859 (2024).
Matsuno, K. et al. Animal models of emerging tick-borne phleboviruses: determining target cells in a lethal model of SFTSV infection. Front. Microbiol. 8, 104 (2017).
Tani, H. et al. Characterization of glycoprotein-mediated entry of severe fever with thrombocytopenia syndrome virus. J. Virol. 90, 5292–5301 (2016).
Liu, Y. et al. The pathogenesis of severe fever with thrombocytopenia syndrome virus infection in alpha/beta interferon knockout mice: insights into the pathologic mechanisms of a new viral hemorrhagic fever. J. Virol. 88, 1781–1786 (2014).
Marín-Lopez, A. et al. Modeling arboviral infection in mice lacking the interferon alpha/beta receptor. Viruses 11, 35 https://doi.org/10.3390/v11010035 (2019).
Jin, C. et al. Pathogenesis of emerging severe fever with thrombocytopenia syndrome virus in C57/BL6 mouse model. Proc. Natl. Acad. Sci. USA 109, 10053–10058 (2012).
Liu, M. M., Lei, X. Y., Yu, H., Zhang, J. Z. & Yu, X. J. Correlation of cytokine level with the severity of severe fever with thrombocytopenia syndrome. Virol. J. 14, 6 (2017).
Takahashi, T. et al. The first identification and retrospective study of severe fever with thrombocytopenia syndrome in Japan. J. Infect. Dis. 209, 816–827 (2014).
Hsu, C. K. et al. Paper-based ELISA for the detection of autoimmune antibodies in body fluid-the case of bullous pemphigoid. Anal. Chem. 86, 4605–4610 (2014).
Elmore, S. A. Enhanced histopathology of the spleen. Toxicol. Pathol. 34, 648–655 (2006).
Acknowledgements
We thank Seo-Bin Oh and Si-On Lee for their secretarial assistance. We also extend our sincere gratitude to the staff at the Center for University-Wide Research Facilities, Jeonbuk National University (JBNU), for their invaluable support with confocal microscopy and flow cytometry analyses. This study was supported by the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (RS-2025-02214485).
Author information
Authors and Affiliations
Contributions
T.S.G., E.L., and E.M. performed experiments, analyzed primary data, and generated figures. S.A. performed the animal experiments. S.T.H. conceptualized and designed the study and planned the specific experiments. T.S.G. analyzed the data and prepared the figures. S.T.H. and T.S.G. wrote the manuscript. S.T.H. and H.J.K. reviewed and revised the manuscript. S.T.H. coordinated the study and secured funding. All authors reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Ganbold, T., Lkhagva, E., Munkhtur, E. et al. Development of a Prophylactic mRNA Vaccine for Severe Fever with Thrombocytopenia Syndrome Using HSA based LNP. npj Vaccines (2026). https://doi.org/10.1038/s41541-026-01385-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41541-026-01385-0