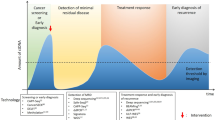
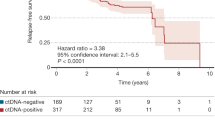
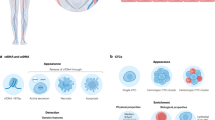
Abstract
Over the past decade, various liquid biopsy techniques have emerged as viable alternatives to the analysis of traditional tissue biopsy samples. Such surrogate ‘biopsies’ offer numerous advantages, including the relative ease of obtaining serial samples and overcoming the issues of interpreting one or more small tissue samples that might not reflect the entire tumour burden. To date, the majority of research in the area of liquid biopsies has focused on blood-based biomarkers, predominantly using plasma-derived circulating tumour DNA (ctDNA). However, ctDNA can also be obtained from various non-blood sources and these might offer unique advantages over plasma ctDNA. In this Review, we discuss advances in the analysis of ctDNA from non-blood sources, focusing on urine, cerebrospinal fluid, and pleural or peritoneal fluid, but also consider other sources of ctDNA. We discuss how these alternative sources can have a distinct yet complementary role to that of blood ctDNA analysis and consider various technical aspects of non-blood ctDNA assay development. We also reflect on the settings in which non-blood ctDNA can offer distinct advantages over plasma ctDNA and explore some of the challenges associated with translating these alternative assays from academia into clinical use.
Key points
-
Plasma-based circulating tumour DNA (ctDNA) assays can provide invaluable information on the status of a patient’s cancer; however, numerous alternative sources of ctDNA are available that might offer unique advantages in certain settings.
-
Non-blood sources of ctDNA include urine, cerebrospinal fluid, pleural or peritoneal fluid, saliva, stool, and seminal fluid, among others.
-
Analysing ctDNA from non-blood sources might provide a more sensitive method than plasma-based ctDNA assays for particular tumour types or anatomical locations.
-
Non-blood ctDNA assays might be complementary to plasma assays when used for the detection of driver alterations or mechanisms of resistance.
-
Challenges to the clinical implementation of non-blood-based ctDNA assays include difficulties relating to standardization of pre-analytical factors, lack of commercially available assays and the invasive procedures required to obtain certain sample types.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Change history
16 December 2022
In the version of this article initially published, the peer reviewer name Y. Nakamura was misspelled and is now amended in the HTML and PDF versions of the article.
References
Siravegna, G. et al. How liquid biopsies can change clinical practice in oncology. Ann. Oncol. 30, 1580–1590 (2019).
De Mattos-Arruda, L. et al. Capturing intra-tumor genetic heterogeneity by de novo mutation profiling of circulating cell-free tumor DNA: A proof-of-principle. Ann. Oncol. 25, 1729–1735 (2014).
Heitzer, E., Haque, I. S., Roberts, C. E. S. & Speicher, M. R. Current and future perspectives of liquid biopsies in genomics-driven oncology. Nat. Rev. Genet. 20, 71–88 (2019).
Gou, Q. et al. Cell-free DNA from bile outperformed plasma as a potential alternative to tissue biopsy in biliary tract cancer. ESMO Open 6, 100275 (2021).
Sethi, S., Benninger, M. S., Lu, M., Havard, S. & Worsham, M. J. Noninvasive molecular detection of head and neck squamous cell carcinoma: an exploratory analysis. Diagn. Mol. Pathol. 18, 81–87 (2009).
Ponti, G. et al. Seminal cell-free DNA assessment as a novel prostate cancer biomarker. Pathol. Oncol. Res. 24, 941–945 (2018).
Imperiale, T. F. et al. Multitarget stool DNA testing for colorectal-cancer screening. N. Engl. J. Med. 370, 1287–1297 (2014).
De Mattos-Arruda, L. et al. Cerebrospinal fluid-derived circulating tumour DNA better represents the genomic alterations of brain tumours than plasma. Nat. Commun. 6, 8839 (2015).
Su, Y.-H. et al. Human urine contains small, 150 to 250 nucleotide-sized, soluble DNA derived from the circulation and may be useful in the detection of colorectal cancer. J. Mol. Diagn. 6, 101–107 (2004).
Bakkum-Gamez, J. N. et al. Detection of endometrial cancer via molecular analysis of DNA collected with vaginal tampons. Gynecol. Oncol. 137, 14–22 (2015).
Schwarzenbach, H., Hoon, D. S. B. & Pantel, K. Cell-free nucleic acids as biomarkers in cancer patients. Nat. Rev. Cancer 11, 426–437 (2011).
Lo, Y. M. D. et al. Maternal plasma DNA sequencing reveals the genome-wide genetic and mutational profile of the fetus. Sci. Transl. Med. 2, 61ra91 (2010).
Thierry, A. R., El Messaoudi, S., Gahan, P. B., Anker, P. & Stroun, M. Origins, structures, and functions of circulating DNA in oncology. Cancer Metastasis Rev. 35, 347–376 (2016).
Underhill, H. R. et al. Fragment length of circulating tumor DNA. PLoS Genet 12, e1006162 (2016).
Mouliere, F. et al. High fragmentation characterizes tumour-derived circulating DNA. PLoS One 6, e23418 (2011).
Higgins, M. J. et al. Detection of tumor PIK3CA status in metastatic breast cancer using peripheral blood. Clin. Cancer Res. 18, 3462–3469 (2012).
Wyatt, A. W. et al. Concordance of circulating tumor DNA and matched metastatic tissue biopsy in prostate cancer. J. Natl Cancer Inst. 109, djx118 (2017).
Tong, L. et al. Tumor-derived DNA from pleural effusion supernatant as a promising alternative to tumor tissue in genomic profiling of advanced lung cancer. Theranostics 9, 5532–5541 (2019).
Ptashkin, R. N. et al. Prevalence of clonal hematopoiesis mutations in tumor-only clinical genomic profiling of solid tumors. JAMA Oncol. 4, 1589–1593 (2018).
Li, Y. S. et al. Unique genetic profiles from cerebrospinal fluid cell-free DNA in leptomeningeal metastases of EGFR-mutant non-small-cell lung cancer: a new medium of liquid biopsy. Ann. Oncol. 29, 945–952 (2018).
Pessoa, L. S., Heringer, M. & Ferrer, V. P. ctDNA as a cancer biomarker: a broad overview. Crit. Rev. Oncol. Hematol. 155, 103109 (2020).
Adashek, J. J., Janku, F. & Kurzrock, R. Signed in blood: circulating tumor DNA in cancer diagnosis, treatment and screening. Cancers 13, 3600 (2021).
Reckamp, K. L. et al. A highly sensitive and quantitative test platform for detection of NSCLC EGFR mutations in urine and plasma. J. Thorac. Oncol. 11, 1690–1700 (2016).
Botezatu, I. et al. Genetic analysis of DNA excreted in urine: a new approach for detecting specific genomic DNA sequences from cells dying in an organism. Clin. Chem. 46, 1078–1084 (2000).
Melkonyan, H. S. et al. Transrenal nucleic acids: from proof of principle to clinical tests. Ann. N. Y. Acad. Sci. 1137, 73–81 (2008).
Green, E. A. et al. Clinical utility of cell-free and circulating tumor DNA in kidney and bladder cancer: a critical review of current literature. Eur. Urol. Oncol. https://doi.org/10.1016/j.euo.2021.04.005 (2021).
Augustus, E. et al. The art of obtaining a high yield of cell-free DNA from urine. PLoS One 15, e0231058 (2020).
Leung, M. S. T., Lin, S. G., Chow, J. & Harky, A. COVID-19 and oncology: service transformation during pandemic. Cancer Med. 9, 7161–7171 (2020).
Zhang, R. et al. Urinary molecular pathology for patients with newly diagnosed urothelial bladder cancer. J. Urol. 206, 873–884 (2021).
Crisafulli, G. et al. Whole exome sequencing analysis of urine trans-renal tumour DNA in metastatic colorectal cancer patients. ESMO Open. 4, e000572 (2019).
Yao, W., Mei, C. & Nan, X. Evaluation and comparison of in vitro degradation kinetics of DNA in serum, urine and saliva: a qualitative study. Gene 590, 142–148 (2016).
Kim, W. T. et al. Urinary cell-free nucleic acid IQGAP3: a new non-invasive diagnostic marker for bladder cancer. Oncotarget 9, 14354–14365 (2018).
Salvi, S. et al. Urine cell-free DNA integrity analysis for early detection of prostate cancer patients. Dis. Markers 2015, 574120 (2015).
Berz, D., Raymond, V. M., Garst, J. H. & Erlander, M. G. Non-invasive urine testing of EGFR activating mutation and T790M resistance mutation in non-small cell lung cancer. Exp. Hematol. Oncol. 5, 24 (2016).
Wang, X. et al. Investigation of transrenal KRAS mutation in late stage NSCLC patients correlates to disease progression. Biomarkers 22, 654–660 (2017).
Guibert, N. et al. Monitoring KRAS mutations in circulating DNA and tumor cells using digital droplet PCR during treatment of KRAS-mutated lung adenocarcinoma. Lung Cancer 100, 1–4 (2016).
Xie, F., Li, P., Gong, J., Tan, H. & Ma, J. Urinary cell-free DNA as a prognostic marker for KRAS-positive advanced-stage NSCLC. Clin. Transl. Oncol. 20, 591–598 (2018).
Shi, L. et al. EGFR mutation status analysis in cerebrospinal fluid and plasma of advanced lung adenocarcinoma with brain metastases. J. Thorac. Oncol. 12, S938–S939 (2017).
Du, Z.-H., Bi, F.-F., Wang, L. & Yang, Q. Next-generation sequencing unravels extensive genetic alteration in recurrent ovarian cancer and unique genetic changes in drug-resistant recurrent ovarian cancer. Mol. Genet. Genom. Med. 6, 638–647 (2018).
Watanabe, J. et al. Comparison of circulating tumor DNA between body fluids in patients with primary central nervous system lymphoma. Leuk. Lymphoma 60, 3587–3589 (2019).
Yu, H., Han, L., Yuan, J. & Sun, Y. Circulating tumor cell free DNA from plasma and urine in the clinical management of colorectal cancer. Cancer Biomark. 27, 29–37 (2019).
Chen, W. et al. Potential use of transrenal DNA for non-invasive monitoring and prognosis of colorectal cancer. Biomarkers 24, 524–529 (2019).
Tian, F., Liao, Y. & Zhang, Y. Variations in transrenal DNA and comparison with plasma DNA as a diagnostic marker for colorectal cancer. Int. J. Biol. Markers 32, 434–440 (2017).
Zuo, Z., Tang, J., Cai, X., Ke, F. & Shi, Z. Probing of breast cancer using a combination of plasma and urinary circulating cell-free DNA. Biosci. Rep. 40, BSR20194306 (2020).
Hann, H.-W. et al. Detection of urine DNA markers for monitoring recurrent hepatocellular carcinoma. Hepatoma Res. 3, 105–111 (2017).
Chang, H. W. et al. Urinary cell-free DNA as a potential tumor marker for bladder cancer. Int. J. Biol. Markers 22, 287–294 (2007).
Brisuda, A. et al. Total amount of cell free DNA in urine of patients harbouring urothelial carcinoma and in urine of controls. Eur. Urol. Suppl. 12, e1221 (2013).
Zancan, M. et al. Free DNA in urine: a new market for bladder cancer? Preliminary data. Int. J. Biol. Markers 20, 134–136 (2005).
Ou, Z. et al. Detection of bladder cancer using urinary cell‐free DNA and cellular DNA. Clin. Transl. Med. 9, 4 (2020).
Christensen, E. et al. Early detection of metastatic relapse and monitoring of therapeutic efficacy by ultra-deep sequencing of plasma cell-free DNA in patients with urothelial bladder carcinoma. J. Clin. Oncol. 37, 1547–1557 (2019).
Birkenkamp-Demtröder, K. et al. Genomic alterations in liquid biopsies from patients with bladder cancer. Eur. Urol. 70, 75–82 (2016).
Togneri, F. S. et al. Genomic complexity of urothelial bladder cancer revealed in urinary cfDNA. Eur. J. Hum. Genet. 24, 1167–1174 (2016).
Christensen, E. et al. Liquid biopsy analysis of FGFR3 and PIK3CA hotspot mutations for disease surveillance in bladder cancer. Eur. Urol. 71, 961–969 (2017).
Springer, S. U. et al. Non-invasive detection of urothelial cancer through the analysis of driver gene mutations and aneuploidy. Elife 7, e32143 (2018).
Dudley, J. C. et al. Detection and surveillance of bladder cancer using urine tumor DNA. Cancer Discov. 9, 500–509 (2019).
Zill, O. A. et al. The landscape of actionable genomic alterations in cell-free circulating tumor DNA from 21,807 advanced cancer patients. Clin. Cancer Res. 24, 3528 (2018).
Smith, C. G. et al. Comprehensive characterization of cell-free tumor DNA in plasma and urine of patients with renal tumors. Genome Med. 12, 23 (2020).
Nuzzo, P. V. et al. Detection of renal cell carcinoma using plasma and urine cell-free DNA methylomes. Nat. Med. 26, 1041–1043 (2020).
Julia, O. et al. MP21-11 diagnostic and prognostic value of urine circulating cell-free DNA in renal cell carcinoma. J. Urol. 201, e296 (2019).
Jahn, J. L., Giovannucci, E. L. & Stampfer, M. J. The high prevalence of undiagnosed prostate cancer at autopsy: implications for epidemiology and treatment of prostate cancer in the prostate-specific antigen-era. Int. J. Cancer 137, 2795–2802 (2015).
Casadio, V. et al. Urine cell-free DNA integrity as a marker for early prostate cancer diagnosis: a pilot study. Biomed. Res. Int. 2013, 270457 (2013).
Silva, R. et al. Evaluating liquid biopsies for methylomic profiling of prostate cancer. Epigenetics 15, 715–727 (2020).
Lu, T. & Li, J. Clinical applications of urinary cell-free DNA in cancer: current insights and promising future. Am. J. Cancer Res. 7, 2318–2332 (2017).
Khatab, S., Spliet, W. & Woerdeman, P. A. Frameless image-guided stereotactic brain biopsies: emphasis on diagnostic yield. Acta Neurochir. 156, 1441–1450 (2014).
Seoane, J., De Mattos-Arruda, L., Le Rhun, E., Bardelli, A. & Weller, M. Cerebrospinal fluid cell-free tumour DNA as a liquid biopsy for primary brain tumours and central nervous system metastases. Ann. Oncol. 30, 211–218 (2019).
De Mattos-Arruda, L. et al. Abstract 930: analysis of cell-free tumor DNA in cerebrospinal fluid to characterize and monitor the genetic alterations of brain tumors. Cancer Res. 75, 930 (2015).
Chen, W. W. et al. Beaming and droplet digital PCR analysis of mutant idh1 mRNA in glioma patient serum and cerebrospinal fluid extracellular vesicles. Mol. Ther. Nucleic Acids 2, e109 (2013).
Moya, I. et al. P05.09 Detection of clinically relevant mutations in the cerebrospinal fluid of patients with central nervous system metastases. Neuro-Oncology 20, iii304 (2018).
Bettegowda, C. et al. Detection of circulating tumor DNA in early- and late-stage human malignancies. Sci. Transl. Med. 6, 224ra24 (2014).
Pan, W., Gu, W., Nagpal, S., Gephart, M. H. & Quake, S. R. Brain tumor mutations detected in cerebral spinal fluid. Clin. Chem. 61, 514–522 (2015).
Escudero, L. et al. Circulating tumour DNA from the cerebrospinal fluid allows the characterisation and monitoring of medulloblastoma. Nat. Commun. 11, 5376 (2020).
Pentsova, E. I. et al. Evaluating cancer of the central nervous system through next-generation sequencing of cerebrospinal fluid. J. Clin. Oncol. 34, 2404–2415 (2016).
Miller, A. M. et al. Tracking tumour evolution in glioma through liquid biopsies of cerebrospinal fluid. Nature 565, 654–658 (2019).
Mouliere, F. et al. Detection of cell-free DNA fragmentation and copy number alterations in cerebrospinal fluid from glioma patients. EMBO Mol. Med. 10, e9323 (2018).
Wesseling, P. & Capper, D. WHO 2016 classification of gliomas. Neuropathol. Appl. Neurobiol. 44, 139–150 (2018).
Fujioka, Y. et al. Molecular diagnosis of diffuse glioma using a chip-based digital PCR system to analyze IDH, TERT, and H3 mutations in the cerebrospinal fluid. J. Neurooncol. 152, 47–54 (2021).
Martínez-Ricarte, F. et al. Molecular diagnosis of diffuse gliomas through sequencing of cell-free circulating tumor DNA from cerebrospinal fluid. Clin. Cancer Res. 24, 2812–2819 (2018).
Panditharatna, E. et al. Clinically relevant and minimally invasive tumor surveillance of pediatric diffuse midline gliomas using patient-derived liquid biopsy. Clin. Cancer Res. 24, 5850–5859 (2018).
Li, D. et al. Standardization of the liquid biopsy for pediatric diffuse midline glioma using ddPCR. Sci. Rep. 11, 5098 (2021).
Liu, A. P. Y. et al. Serial assessment of measurable residual disease in medulloblastoma liquid biopsies. Cancer Cell 39, 1519–1530.e4 (2021).
Rimelen, V. et al. Tumor cell-free DNA detection in CSF for primary CNS lymphoma diagnosis. Acta Neuropathol. Commun. 7, 43 (2019).
Watanabe, J. et al. High detection rate of MYD88 mutations in cerebrospinal fluid from patients with CNS lymphomas. JCO Precis. Oncol. https://doi.org/10.1200/PO.18.00308 (2019).
Grommes, C. et al. Phase 1b trial of an ibrutinib-based combination therapy in recurrent/refractory CNS lymphoma. Blood 133, 436–445 (2019).
Mutter, J. A. et al. Profiling of circulating tumor DNA for noninvasive disease detection, risk stratification, and MRD monitoring in patients with CNS lymphoma. Blood 138, 6 (2021).
Bobillo, S. et al. Cell free circulating tumor DNA in cerebrospinal fluid detects and monitors central nervous system involvement of B-cell lymphomas. Haematologica 106, 513–521 (2021).
Woodcock, D. J. et al. Prostate cancer evolution from multilineage primary to single lineage metastases with implications for liquid biopsy. Nat. Commun. 11, 5070 (2020).
Wong, S. Q. et al. Circulating tumor DNA analysis and functional imaging provide complementary approaches for comprehensive disease monitoring in metastatic melanoma. JCO Precis. Oncol. https://doi.org/10.1200/po.16.00009 (2017).
Ying, S. et al. Unique genomic profiles obtained from cerebrospinal fluid cell-free DNA of non-small cell lung cancer patients with leptomeningeal metastases. Cancer Biol. Ther. 20, 562–570 (2019).
Azad, T. et al. Next generation sequencing of cerebrospinal fluid to improve diagnostic sensitivity, detect spatial heterogeneity, and predict outcomes for advanced lung cancer patients with leptomeningeal carcinomatosis. J. Neurosurg. 132, 110 (2020).
Zhao, Y. et al. Evaluating the cerebrospinal fluid ctDNA detection by next-generation sequencing in the diagnosis of meningeal Carcinomatosis. BMC Neurol. 19, 331 (2019).
Liu, Y. et al. Cell-free DNA from cerebrospinal fluid can be used to detect the EGFR mutation status of lung adenocarcinoma patients with central nervous system metastasis. Transl. Lung Cancer Res. 10, 914–925 (2021).
Zheng, M.-M. et al. Clinical utility of cerebrospinal fluid cell-free DNA as liquid biopsy for leptomeningeal metastases in ALK-rearranged NSCLC. J. Thorac. Oncol. 14, 924–932 (2019).
Yang, H. et al. Sensitive detection of EGFR mutations in cerebrospinal fluid from lung adenocarcinoma patients with brain metastases. J. Mol. Diagn. 16, 558–563 (2014).
Oxnard, G. R. et al. Association between plasma genotyping and outcomes of treatment with osimertinib (AZD9291) in advanced non-small-cell lung cancer. J. Clin. Oncol. 34, 3375–3382 (2016).
Oxnard, G. R. et al. Noninvasive detection of response and resistance in egfrmutant lung cancer using quantitative next-generation genotyping of cell-free plasma DNA. Clin. Cancer Res. 20, 1698–1705 (2014).
Wu, X. et al. Genetic profiling of cerebrospinal fluid cfDNA from NSCLC patients with leptomeningealmetastases reveals EGFR-TKIs resistant mutations independent of extracranial lesions (cases series). Cancer Res. 80 (Suppl. 16), 723 (2020).
Jiang, B. et al. Unique genetic profiles from circulating Cell-Free DNA of cerebrospinal fluid in leptomeningeal metastases of EGFR Mutant NSCLC. J. Thorac. Oncol. 12, S1771 (2017).
Melms, J. et al. Implementation of cell‑free tumor DNA sequencing from the cerebrospinal fluid to guide treatment in a patient with primary leptomeningeal melanoma: a case report. Mol. Clin. Oncol. 9, 58–61 (2018).
Li, Y. et al. Tumor DNA in cerebral spinal fluid reflects clinical course in a patient with melanoma leptomeningeal brain metastases. J. Neurooncol. 128, 93–100 (2016).
Skok, K., Hladnik, G., Grm, A. & Crnjac, A. Malignant pleural effusion and its current management: a review. Medicina 55, 490 (2019).
Sriram, K. B. et al. Pleural fluid cell-free DNA integrity index to identify cytologically negative malignant pleural effusions including mesotheliomas. BMC Cancer 12, 428 (2012).
Durin, L. et al. Liquid biopsy of non-plasma body fluids in non-small cell lung cancer: look closer to the tumor! Cells 9, 2486 (2020).
Zhou, Z. et al. Lung cancer genomic alterations in cell free DNA in pleural effusion compared to plasma. J. Clin. Oncol. 35 (Suppl. 15), e23206 (2017).
Han, M.-R. et al. Clinical implications of circulating tumor DNA from ascites and serial plasma in ovarian cancer. Cancer Res. Treat. 52, 779–788 (2020).
Shi, C. et al. Analysis of mutation detection in cell-free DNA in ascites using comprehensive NGS panel. J. Clin. Oncol. 37 (Suppl. 15), e13029 (2019).
Leick, K. M. et al. Peritoneal cell-free tumor DNA as biomarker for peritoneal surface malignancies. Ann. Surg. Oncol. 27, 5065–5071 (2020).
López-Rojo, I. et al. Liquid biopsy in peritoneal fluid and plasma as a prognostic factor in advanced colorectal and appendiceal tumors after complete cytoreduction and hyperthermic intraperitoneal chemotherapy. Ther. Adv. Med. Oncol. 12, 1758835920981351 (2020).
Van’t Erve, I. et al. Detection of tumor-derived cell-free DNA from colorectal cancer peritoneal metastases in plasma and peritoneal fluid. J. Pathol. Clin. Res. https://doi.org/10.1002/cjp2.207 (2021).
Leick, K. M. et al. Peritoneal cell-free tumor DNA as a biomarker of peritoneal recurrence in resected pancreatic cancers. Ann. Surg. Oncol. 27, S125–S126 (2020).
Kaczor-Urbanowicz, K. E. et al. Saliva diagnostics-current views and directions. Exp. Biol. Med. 242, 459–472 (2017).
Zimmermann, B. G., Park, N. J. & Wong, D. T. Genomic targets in saliva. Ann. N. Y. Acad. Sci. 1098, 184–191 (2007).
Dean, M. et al. Addressing health disparities in Hispanic breast cancer: accurate and inexpensive sequencing of BRCA1 and BRCA2. Gigascience 4, 50 (2015).
Wang, X., Kaczor-Urbanowicz, K. E. & Wong, D. T. W. Salivary biomarkers in cancer detection. Med. Oncol. 34, 7 (2017).
Kaczor-Urbanowicz, K. E. et al. Clinical validity of saliva and novel technology for cancer detection. Biochim. Biophys. Acta Rev. Cancer 1872, 49–59 (2019).
Ahn, S. M. et al. Saliva and plasma quantitative polymerase chain reaction-based detection and surveillance of human papillomavirus-related head and neck cancer. JAMA Otolaryngol. Head Neck Surg. 140, 846–854 (2014).
Hanna, G. J. et al. Salivary HPV DNA informs locoregional disease status in advanced HPV-associated oropharyngeal cancer. Oral. Oncol. 95, 120–126 (2019).
Wang, Y. et al. Detection of somatic mutations and HPV in the saliva and plasma of patients with head and neck squamous cell carcinomas. Sci. Transl. Med. 7, 293ra104 (2015).
Ding, S. et al. Saliva-derived cfDNA is applicable for EGFR mutation detection but not for quantitation analysis in non-small cell lung cancer. Thorac. Cancer 10, 1973–1983 (2019).
Wei, F. et al. Noninvasive saliva-based EGFR gene mutation detection in patients with lung cancer. Am. J. Respir. Crit. Care Med. 190, 1117–1126 (2014).
Pu, D. et al. Evaluation of a novel saliva-based epidermal growth factor receptor mutation detection for lung cancer: a pilot study. Thorac. Cancer 7, 428–436 (2016).
Li, F. et al. EFIRM liquid biopsy (eLB): detection of ultrashort circulating tumor DNA (usctDNA) in plasma and saliva of non-small cell lung cancer (NSCLC) patients. J. Clin. Oncol. 36 (Suppl. 15), e24062 (2018).
Li, F. et al. Ultra-short circulating tumor DNA (usctDNA) in plasma and saliva of non-small cell lung cancer (NSCLC) patients. Cancers 12, 2041 (2020).
Aghagolzadeh, P. & Radpour, R. New trends in molecular and cellular biomarker discovery for colorectal cancer. World J. Gastroenterol. 22, 5678–5693 (2016).
Klaassen, C. H. W. et al. Quantification of human DNA in feces as a diagnostic test for the presence of colorectal cancer. Clin. Chem. 49, 1185–1187 (2003).
Imperiale, T. F., Ransohoff, D. F., Itzkowitz, S. H., Turnbull, B. A. & Ross, M. E. Fecal DNA versus fecal occult blood for colorectal-cancer screening in an average-risk population. N. Engl. J. Med. 351, 2704–2714 (2009).
Prince, M., Lester, L., Chiniwala, R. & Berger, B. Multitarget stool DNA tests increases colorectal cancer screening among previously noncompliant medicare patients. World J. Gastroenterol. 23, 464–471 (2017).
Redwood, D. G. et al. Stool DNA testing for screening detection of colorectal neoplasia in Alaska native people. Mayo Clin. Proc. 91, 61–70 (2016).
Naber, S. K. et al. Cost-effectiveness of a multitarget stool DNA test for colorectal cancer screening of medicare beneficiaries. PLoS One 14, e0220234–e0220234 (2019).
van Lanschot, M. C. J. et al. Molecular stool testing as an alternative for surveillance colonoscopy: a cross-sectional cohort study. BMC Cancer 17, 116 (2017).
Caldas, C. et al. Detection of K-ras mutations in the stool of patients with pancreatic adenocarcinoma and pancreatic ductal hyperplasia. Cancer Res. 54, 3568 LP–3563573 (1994).
Kisiel, J. B. et al. Stool DNA testing for the detection of pancreatic cancer: assessment of methylation marker candidates. Cancer 118, 2623–2631 (2012).
van Dam, L. et al. What influences the decision to participate in colorectal cancer screening with faecal occult blood testing and sigmoidoscopy? Eur. J. Cancer 49, 2321–2330 (2013).
Osborne, J. M. et al. The impact of sample type and procedural attributes on relative acceptability of different colorectal cancer screening regimens. Patient Prefer. Adherence 12, 1825 (2018).
Youssef, O. et al. Gene mutations in stool from gastric and colorectal neoplasia patients by next-generation sequencing. World J. Gastroenterol. 23, 8291–8299 (2017).
Zhang, F. et al. Analysis of the gut microbiota: an emerging source of biomarkers for immune checkpoint blockade therapy in non-small cell lung cancer. Cancers 13, 2514 (2021).
Li, H.-G., Huang, S.-Y., Zhou, H., Liao, A.-H. & Xiong, C.-L. Quick recovery and characterization of cell-free DNA in seminal plasma of normozoospermia and azoospermia: implications for non-invasive genetic utilities. Asian J. Androl. 11, 703–709 (2009).
Ponti, G. et al. Quick assessment of cell-free DNA in seminal fluid and fragment size for early non-invasive prostate cancer diagnosis. Clin. Chim. Acta 497, 76–80 (2019).
Tran, S., Boissier, R., Perrin, J., Karsenty, G. & Lechevallier, E. Review of the different treatments and management for prostate cancer and fertility. Urology 86, 936–941 (2015).
Shen, N. et al. Bile cell‑free DNA as a novel and powerful liquid biopsy for detecting somatic variants in biliary tract cancer. Oncol. Rep. 42, 549–560 (2019).
Han, J.-Y. et al. Liquid biopsy from bile-circulating tumor DNA in patients with biliary tract cancer. Cancers 13, 4581 (2021).
Nair, N. et al. Genomic analysis of uterine lavage fluid detects early endometrial cancers and reveals a prevalent landscape of driver mutations in women without histopathologic evidence of cancer: a prospective cross-sectional study. PLoS Med. 13, e1002206 (2016).
Diehl, F. et al. Circulating mutant DNA to assess tumor dynamics. Nat. Med. 14, 985–990 (2008).
Merker, J. D. et al. Circulating tumor DNA analysis in patients with cancer: American society of clinical oncology and college of American pathologists joint review. J. Clin. Oncol. 36, 1631–1641 (2018).
Kang, Q. et al. Comparative analysis of circulating tumor DNA stability in K3EDTA, Streck, and CellSave blood collection tubes. Clin. Biochem. 49, 1354–1360 (2016).
Woodhouse, R. et al. Clinical and analytical validation of FoundationOne Liquid CDx, a novel 324-Gene cfDNA-based comprehensive genomic profiling assay for cancers of solid tumor origin. PLoS One 15, e0237802 (2020).
Bauml, J. M. et al. Clinical validation of Guardant360 CDx as a blood-based companion diagnostic for sotorasib. Lung Cancer 166, 270–278 (2021).
Abbosh, C., Birkbak, N. J. & Swanton, C. Early stage NSCLC — challenges to implementing ctDNA-based screening and MRD detection. Nat. Rev. Clin. Oncol. 15, 577–586 (2018).
Liu, M. C. et al. Sensitive and specific multi-cancer detection and localization using methylation signatures in cell-free DNA. Ann. Oncol. 31, 745–759 (2020).
Cohen, J. D. et al. Detection and localization of surgically resectable cancers with a multi-analyte blood test. Science 359, 926 (2018).
Cristiano, S. et al. Genome-wide cell-free DNA fragmentation in patients with cancer. Nature 570, 385 (2019).
Aggarwal, C. et al. Clinical implications of plasma-based genotyping with the delivery of personalized therapy in metastatic non-small cell lung cancer. JAMA Oncol. 5, 173–180 (2019).
Li, J. et al. Reliable tumor detection by whole-genome methylation sequencing of cell-free DNA in cerebrospinal fluid of pediatric medulloblastoma. Sci. Adv. 6, eabb5427 (2020).
Acknowledgements
The work of A.T. is supported by an NIHR Academic Clinical Fellowship. The work of D.R. and C.D. is funded by Cancer Research UK (CRUK) via core funding to the CRUK Manchester Institute (grant A27412) and the CRUK Manchester Centre (grant A25254). Support was received by the NIHR Manchester Biomedical Research Centre and the Manchester Experimental Cancer Medicine Centre.
Author information
Authors and Affiliations
Contributions
A.T., M.C. and N.C. researched data for article and wrote the manuscript. All authors contributed to discussions of content and reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
C.D. has acted as a consultant and/or adviser of AstraZeneca, Boehringer Ingelheim, Biocartis, GRAIL and Merck, and has received research funding from Angle, Amgen, Astex Pharmaceuticals, AstraZeneca, Bayer, Bioven, Bristol Myers Squibb, Boehringer Ingelheim, Carrick Therapeutics, Celgene, Clearbridge Biomedics, Epigene Therapeutics, GlaxoSmithKline, Menarini, Merck AG, Neomed Therapeutics, Novartis, Roche, Taiho Oncology and Thermo Fisher Scientific. N.C. has received research funding from AstraZeneca, Avacta, Bayer, Boehringer, Eisai, Merck, Orion, Pfizer, RedX, Roche, Starpharma, Stemline Tarveda, Taiho Oncology and UCB. The other authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Clinical Oncology thanks J. Seoane, Y. Nakamura and the other, anonymous reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tivey, A., Church, M., Rothwell, D. et al. Circulating tumour DNA — looking beyond the blood. Nat Rev Clin Oncol 19, 600–612 (2022). https://doi.org/10.1038/s41571-022-00660-y
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41571-022-00660-y
This article is cited by
-
Detection of primary cancer types via fragment size selection in circulating cell-free extrachromosomal circular DNA
Genome Medicine (2026)
-
Personalized ctDNA analysis for detection of residual disease and recurrence in surgically treated HNSCC patients
npj Precision Oncology (2026)
-
Secreted luciferases as a minimally invasive 3R-compliant tool for accurate monitoring of tumor burden
Nature Protocols (2026)
-
cfGWAS reveal genetic basis of cell-free DNA end motifs
Nature Communications (2026)
-
Unlocking the potential of urine-based liquid biopsy through improved reporting and standardization
Nature Reviews Cancer (2026)