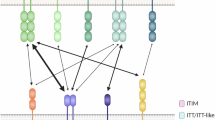
Abstract
Substantial progress in understanding T cell signalling, particularly with respect to T cell co-receptors such as the co-stimulatory receptor CD28, has been made in recent years. This knowledge has been instrumental in the development of innovative immunotherapies for patients with cancer, including immune checkpoint blockade antibodies, adoptive cell therapies, tumour-targeted immunostimulatory antibodies, and immunostimulatory small-molecule drugs that regulate T cell activation. Following the failed clinical trial of a CD28 superagonist antibody in 2006, targeted CD28 agonism has re-emerged as a technologically viable and clinically promising strategy for cancer immunotherapy. In this Review, we explore recent insights into the molecular functions and regulation of CD28. We describe how CD28 is central to the success of current cancer immunotherapies and examine how new questions arising from studies of CD28 as a clinical target have enhanced our understanding of its biological role and may guide the development of future therapeutic strategies in oncology.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Callahan, M. K., Postow, M. A. & Wolchok, J. D. Targeting T cell co-receptors for cancer therapy. Immunity 44, 1069–1078 (2016).
Attanasio, J. & Wherry, E. J. Costimulatory and coinhibitory receptor pathways in infectious disease. Immunity 44, 1052–1068 (2016).
Chen, L. et al. Costimulation of antitumor immunity by the B7 counterreceptor for the T lymphocyte molecules CD28 and CTLA-4. Cell 71, 1093–1102 (1992).
Townsend, S. E. & Allison, J. P. Tumor rejection after direct costimulation of CD8+ T cells by B7-transfected melanoma cells. Science 259, 368–370 (1993). Together with Chen et al. (1992), this important study shows that co-stimulation through CD28 can induce antitumour immunity.
Leach, D. R., Krummel, M. F. & Allison, J. P. Enhancement of antitumor immunity by CTLA-4 blockade. Science 271, 1734–1736 (1996).
Kamphorst, A. O. et al. Rescue of exhausted CD8 T cells by PD-1-targeted therapies is CD28-dependent. Science 355, 1423–1427 (2017).
Hui, E. et al. T cell costimulatory receptor CD28 is a primary target for PD-1-mediated inhibition. Science 355, 1428–1433 (2017). Together with Kamphorst et al. (2017), this paper shows that CD28 is the main target of PD1 inhibition, which led to the re-emergence of CD28 as a therapeutic target in cancer.
Suntharalingam, G. et al. Cytokine storm in a phase 1 trial of the anti-CD28 monoclonal antibody TGN1412. N. Engl. J. Med. 355, 1018–1028 (2006). This paper reports the failed phase I clinical trial of a CD28-superagonist antibody (TGN1412), which set back research into CD28 agonism for cancer therapy.
Hunig, T. The storm has cleared: lessons from the CD28 superagonist TGN1412 trial. Nat. Rev. Immunol. 12, 317–318 (2012). This paper provides a comprehensive description of lessons learned from the TGN1412 clinical trial that provides a basis for changes made to the way in which immunotherapies are clinically tested.
Horvath, C. et al. Storm forecasting: additional lessons from the CD28 superagonist TGN1412 trial. Nat. Rev. Immunol. 12, 740 (2012).
Burke, K. P., Chaudhri, A., Freeman, G. J. & Sharpe, A. H. The B7:CD28 family and friends: unraveling coinhibitory interactions. Immunity 57, 223–244 (2024).
Esensten, J. H., Helou, Y. A., Chopra, G., Weiss, A. & Bluestone, J. A. CD28 costimulation: from mechanism to therapy. Immunity 44, 973–988 (2016).
Edner, N. M., Carlesso, G., Rush, J. S. & Walker, L. S. K. Targeting co-stimulatory molecules in autoimmune disease. Nat. Rev. Drug Discov. 19, 860–883 (2020).
Kohler, G. & Milstein, C. Continuous cultures of fused cells secreting antibody of predefined specificity. Nature 256, 495–497 (1975).
Hansen, J. A., Martin, P. J. & Nowinski, R. C. Monoclonal antibodies identifying a novel T-Cell antigen and Ia antigens of human lymphocytes. Immunogenetics 10, 247–260 (1980).
Clark, E. A., Martin, P. J., Hansen, J. A. & Ledbetter, J. A. Evolution of epitopes on human and nonhuman primate lymphocyte cell surface antigens. Immunogenetics 18, 599–615 (1983).
Testi, R. & Lanier, L. L. Functional expression of CD28 on T cell antigen receptor γ/δ-bearing T lymphocytes. Eur. J. Immunol. 19, 185–188 (1989).
Kozbor, D., Moretta, A., Messner, H. A., Moretta, L. & Croce, C. M. Tp44 molecules involved in antigen-independent T cell activation are expressed on human plasma cells. J. Immunol. 138, 4128–4132 (1987).
Hara, T., Fu, S. M. & Hansen, J. A. Human T cell activation. II. A new activation pathway used by a major T cell population via a disulfide-bonded dimer of a 44 kilodalton polypeptide (9.3 antigen). J. Exp. Med. 161, 1513–1524 (1985).
Moretta, A., Pantaleo, G., Lopez-Botet, M. & Moretta, L. Involvement of T44 molecules in an antigen-independent pathway of T cell activation. Analysis of the correlations to the T cell antigen-receptor complex. J. Exp. Med. 162, 823–838 (1985).
Martin, P. J. et al. A 44 kilodalton cell surface homodimer regulates interleukin 2 production by activated human T lymphocytes. J. Immunol. 136, 3282–3287 (1986).
Weiss, A., Manger, B. & Imboden, J. Synergy between the T3/antigen receptor complex and Tp44 in the activation of human T cells. J. Immunol. 137, 819–825 (1986).
June, C. H., Ledbetter, J. A., Gillespie, M. M., Lindsten, T. & Thompson, C. B. T-cell proliferation involving the CD28 pathway is associated with cyclosporine-resistant interleukin 2 gene expression. Mol. Cell Biol. 7, 4472–4481 (1987).
Lindsten, T., June, C. H., Ledbetter, J. A., Stella, G. & Thompson, C. B. Regulation of lymphokine messenger RNA stability by a surface-mediated T cell activation pathway. Science 244, 339–343 (1989).
Azuma, M. et al. B70 antigen is a second ligand for CTLA-4 and CD28. Nature 366, 76–79 (1993).
Freedman, A. S., Freeman, G., Horowitz, J. C., Daley, J. & Nadler, L. M. B7, a B-cell-restricted antigen that identifies preactivated B cells. J. Immunol. 139, 3260–3267 (1987).
Freeman, G. J. et al. Cloning of B7-2: a CTLA-4 counter-receptor that costimulates human T cell proliferation. Science 262, 909–911 (1993).
Freeman, G. J. et al. Uncovering of functional alternative CTLA-4 counter-receptor in B7-deficient mice. Science 262, 907–909 (1993).
Thompson, C. B. et al. CD28 activation pathway regulates the production of multiple T-cell-derived lymphokines/cytokines. Proc. Natl Acad. Sci. USA 86, 1333–1337 (1989).
Bretscher, P. & Cohn, M. A theory of self-nonself discrimination. Science 169, 1042–1049 (1970).
Brunet, J. F. et al. A new member of the immunoglobulin superfamily-CTLA-4. Nature 328, 267–270 (1987).
Walunas, T. L. et al. CTLA-4 can function as a negative regulator of T cell activation. Immunity 1, 405–413 (1994).
Alegre, M. L., Frauwirth, K. A. & Thompson, C. B. T-cell regulation by CD28 and CTLA-4. Nat. Rev. Immunol. 1, 220–228 (2001).
Acuto, O. & Michel, F. CD28-mediated co-stimulation: a quantitative support for TCR signalling. Nat. Rev. Immunol. 3, 939–951 (2003).
Lazar-Molnar, E., Almo, S. C. & Nathenson, S. G. The interchain disulfide linkage is not a prerequisite but enhances CD28 costimulatory function. Cell Immunol. 244, 125–129 (2006).
Sanchez-Lockhart, M., Kim, M. & Miller, J. Cutting edge: a role for inside-out signaling in TCR regulation of CD28 ligand binding. J. Immunol. 187, 5515–5519 (2011).
Boomer, J. S. & Green, J. M. An enigmatic tail of CD28 signaling. Cold Spring Harb. Perspect. Biol. 2, a002436 (2010).
Leddon, S. A. et al. The CD28 transmembrane domain contains an essential dimerization motif. Front. Immunol. 11, 1519 (2020).
Muller, Y. D. et al. The CD28-transmembrane domain mediates chimeric antigen receptor heterodimerization with CD28. Front. Immunol. 12, 639818 (2021).
Wu, H., Cao, R., Wen, M., Xue, H. & OuYang, B. Structural characterization of a dimerization interface in the CD28 transmembrane domain. Structure 30, 803–812.e5 (2022).
Tian, R. et al. Combinatorial proteomic analysis of intercellular signaling applied to the CD28 T-cell costimulatory receptor. Proc. Natl Acad. Sci. USA 112, E1594–E1603 (2015).
Chen, X. et al. Motif-dependent immune co-receptor interactome profiling by photoaffinity chemical proteomics. Cell Chem. Biol. 29, 1024–1036.e5 (2022). This paper comprehensively delineates signalling proteins recruited to active, tyrosine-phosphorylated signalling motifs in the CD28 intracellular domain.
Friend, L. D. et al. A dose-dependent requirement for the proline motif of CD28 in cellular and humoral immunity revealed by a targeted knockin mutant. J. Exp. Med. 203, 2121–2133 (2006).
Dodson, L. F. et al. Targeted knock-in mice expressing mutations of CD28 reveal an essential pathway for costimulation. Mol. Cell Biol. 29, 3710–3721 (2009).
Porciello, N. et al. A non-conserved amino acid variant regulates differential signalling between human and mouse CD28. Nat. Commun. 9, 1080 (2018).
Dobbins, J. et al. Binding of the cytoplasmic domain of CD28 to the plasma membrane inhibits Lck recruitment and signaling. Sci. Signal. 9, ra75 (2016).
Yang, W. et al. Dynamic regulation of CD28 conformation and signaling by charged lipids and ions. Nat. Struct. Mol. Biol. 24, 1081–1092 (2017).
Yokosuka, T. et al. Spatiotemporal regulation of T cell costimulation by TCR-CD28 microclusters and protein kinase C θ translocation. Immunity 29, 589–601 (2008).
Raab, M. et al. p56Lck and p59Fyn regulate CD28 binding to phosphatidylinositol 3-kinase, growth factor receptor-bound protein GRB-2, and T cell-specific protein-tyrosine kinase ITK: implications for T-cell costimulation. Proc. Natl Acad. Sci. USA 92, 8891–8895 (1995).
August, A. et al. CD28 is associated with and induces the immediate tyrosine phosphorylation and activation of the Tec family kinase ITK/EMT in the human Jurkat leukemic T-cell line. Proc. Natl Acad. Sci. USA 91, 9347–9351 (1994).
Hallumi, E. et al. Itk promotes the integration of TCR and CD28 costimulation through its direct substrates SLP-76 and gads. J. Immunol. 206, 2322–2337 (2021).
Azuma, M., Yssel, H., Phillips, J. H., Spits, H. & Lanier, L. L. Functional expression of B7/BB1 on activated T lymphocytes. J. Exp. Med. 177, 845–850 (1993).
Hwang, I. et al. T cells can use either T cell receptor or CD28 receptors to absorb and internalize cell surface molecules derived from antigen-presenting cells. J. Exp. Med. 191, 1137–1148 (2000).
Zhao, Y. et al. cis-B7:CD28 interactions at invaginated synaptic membranes provide CD28 co-stimulation and promote CD8+ T cell function and anti-tumor immunity. Immunity 56, 1187–1203.e12 (2023). This study shows that CD28 signalling can be initiated by cis-interaction with B7 proteins expressed on the same T cell.
Dagar, G., Kumar, R., Yadav, K. K., Singh, M. & Pandita, T. K. Ubiquitination and deubiquitination: implications on cancer therapy. Biochim. Biophys. Acta Gene Regul. Mech. 1866, 194979 (2023).
Michaelides, I. N. & Collie, G. W. E3 ligases meet their match: fragment-based approaches to discover new E3 ligands and to unravel E3 biology. J. Med. Chem. 66, 3173–3194 (2023).
Lane, I. C. et al. Genetic retargeting of E3 ligases to enhance CAR T cell therapy. Cell Chem. Biol. 31, 338–348.e5 (2024).
Hou, B. et al. The E3 ubiquitin ligases regulate PD-1/PD-L1 protein levels in tumor microenvironment to improve immunotherapy. Front. Immunol. 14, 1123244 (2023).
Berges, C. et al. Proteasome inhibition suppresses essential immune functions of human CD4+ T cells. Immunology 124, 234–246 (2008).
Appleman, L. J., Berezovskaya, A., Grass, I. & Boussiotis, V. A. CD28 costimulation mediates T cell expansion via IL-2-independent and IL-2-dependent regulation of cell cycle progression. J. Immunol. 164, 144–151 (2000).
Liu, J. et al. FBW7-mediated ubiquitination and destruction of PD-1 protein primes sensitivity to anti-PD-1 immunotherapy in non-small cell lung cancer. J. Immunother. Cancer 10, e005116 (2022).
Bekes, M., Langley, D. R. & Crews, C. M. PROTAC targeted protein degraders: the past is prologue. Nat. Rev. Drug Discov. 21, 181–200 (2022).
Haymaker, C. et al. Absence of Grail promotes CD8+ T cell anti-tumour activity. Nat. Commun. 8, 239 (2017).
Venuprasad, K. Cbl-b and Itch: key regulators of peripheral T-cell tolerance. Cancer Res. 70, 3009–3012 (2010).
Nguyen, T. T. T. et al. Cbl-b deficiency prevents functional but not phenotypic T cell anergy. J. Exp. Med. 218, e20202477 (2021).
Chiang, Y. J. et al. Cbl-b regulates the CD28 dependence of T-cell activation. Nature 403, 216–220 (2000).
Zhang, J. et al. Cutting edge: regulation of T cell activation threshold by CD28 costimulation through targeting Cbl-b for ubiquitination. J. Immunol. 169, 2236–2240 (2002).
Stromnes, I. M. et al. Abrogating Cbl-b in effector CD8+ T cells improves the efficacy of adoptive therapy of leukemia in mice. J. Clin. Invest. 120, 3722–3734 (2010). This classic manuscript demonstrates the extraordinary efficacy of adoptively transferred T cells when CBL-B is deleted (but limited memory formation).
Shah, N. N. et al. Clonal expansion of CAR T cells harboring lentivector integration in the CBL gene following anti-CD22 CAR T-cell therapy. Blood Adv. 3, 2317–2322 (2019).
Augustin, R. C., Bao, R. & Luke, J. J. Targeting Cbl-b in cancer immunotherapy. J. Immunother. Cancer 11, e006007 (2023).
Frauwirth, K. A. et al. The CD28 signaling pathway regulates glucose metabolism. Immunity 16, 769–777 (2002). This study links CD28 co-stimulation to T cell metabolic reprogramming.
Beckermann, K. E. et al. CD28 costimulation drives tumor-infiltrating T cell glycolysis to promote inflammation. JCI Insight 5, e138729 (2020).
Menk, A. V. et al. Early TCR signaling induces rapid aerobic glycolysis enabling distinct acute T cell effector functions. Cell Rep. 22, 1509–1521 (2018).
Holling, G. A. et al. CD8+ T cell metabolic flexibility elicited by CD28-ARS2 axis-driven alternative splicing of PKM supports antitumor immunity. Cell Mol. Immunol. 21, 260–274 (2024).
Klein Geltink, R. I. et al. Mitochondrial priming by CD28. Cell 171, 385–397.e11 (2017).
Bailis, W. et al. Distinct modes of mitochondrial metabolism uncouple T cell differentiation and function. Nature 571, 403–407 (2019).
Humblin, E. et al. Sustained CD28 costimulation is required for self-renewal and differentiation of TCF-1+ PD-1+ CD8 T cells. Sci. Immunol. 8, eadg0878 (2023).
Scharping, N. E. et al. Mitochondrial stress induced by continuous stimulation under hypoxia rapidly drives T cell exhaustion. Nat. Immunol. 22, 205–215 (2021).
Popovic, B. et al. Time-dependent regulation of cytokine production by RNA binding proteins defines T cell effector function. Cell Rep. 42, 112419 (2023).
Salerno, F., Turner, M. & Wolkers, M. C. Dynamic post-transcriptional events governing CD8+ T cell homeostasis and effector function. Trends Immunol. 41, 240–254 (2020).
Butte, M. J. et al. CD28 costimulation regulates genome-wide effects on alternative splicing. PLoS ONE 7, e40032 (2012).
Elahi, S. et al. The RNA binding protein Ars2 supports hematopoiesis at multiple levels. Exp. Hematol. 64, 45–58.e9 (2018).
Dolz, M. et al. Forced expression of the non-coding RNA miR-17 approximately 92 restores activation and function in CD28-deficient CD4+ T cells. iScience 25, 105372 (2022).
Gracias, D. T. et al. The microRNA miR-155 controls CD8+ T cell responses by regulating interferon signaling. Nat. Immunol. 14, 593–602 (2013).
Bronevetsky, Y. et al. T cell activation induces proteasomal degradation of Argonaute and rapid remodeling of the microRNA repertoire. J. Exp. Med. 210, 417–432 (2013).
La Rocca, G. et al. In vivo, Argonaute-bound microRNAs exist predominantly in a reservoir of low molecular weight complexes not associated with mRNA. Proc. Natl Acad. Sci. USA 112, 767–772 (2015). This study shows that microRNAs exist in inactive ribonucleoprotein complexes in resting T cells and that CD28 signalling can activate microRNA function by stimulating RISC assembly.
Olejniczak, S. H. et al. Coordinated regulation of cap-dependent translation and microRNA function by convergent signaling pathways. Mol. Cell Biol. 36, 2360–2373 (2016).
Tang, W. W., Bauer, K. M., Barba, C., Ekiz, H. A. & O’Connell, R. miR-aculous new avenues for cancer immunotherapy. Front. Immunol. 13, 929677 (2022).
DuPage, M. et al. The chromatin-modifying enzyme Ezh2 is critical for the maintenance of regulatory T cell identity after activation. Immunity 42, 227–238 (2015).
Arvey, A. et al. Inflammation-induced repression of chromatin bound by the transcription factor Foxp3 in regulatory T cells. Nat. Immunol. 15, 580–587 (2014).
Tumes, D. J. et al. The polycomb protein Ezh2 regulates differentiation and plasticity of CD4+ T helper type 1 and type 2 cells. Immunity 39, 819–832 (2013).
Kakaradov, B. et al. Early transcriptional and epigenetic regulation of CD8+ T cell differentiation revealed by single-cell RNA sequencing. Nat. Immunol. 18, 422–432 (2017). This study links early T cell signalling events to the subsequent differentiation of effector T cells via epigenetic remodelling.
Gray, S. M., Amezquita, R. A., Guan, T., Kleinstein, S. H. & Kaech, S. M. Polycomb repressive complex 2-mediated chromatin repression guides effector CD8+ T cell terminal differentiation and loss of multipotency. Immunity 46, 596–608 (2017).
Zhao, E. et al. Cancer mediates effector T cell dysfunction by targeting microRNAs and EZH2 via glycolysis restriction. Nat. Immunol. 17, 95–103 (2016).
He, S. et al. Ezh2 phosphorylation state determines its capacity to maintain CD8+ T memory precursors for antitumor immunity. Nat. Commun. 8, 2125 (2017).
Long, X. et al. Histone methyltransferase Nsd2 is required for follicular helper T cell differentiation. J. Exp. Med. 217, e20190832 (2020).
Thomas, R. M., Gao, L. & Wells, A. D. Signals from CD28 induce stable epigenetic modification of the IL-2 promoter. J. Immunol. 174, 4639–4646 (2005).
Liu, Y., Chen, Y. & Richardson, B. Decreased DNA methyltransferase levels contribute to abnormal gene expression in “senescent” CD4+CD28− T cells. Clin. Immunol. 132, 257–265 (2009).
Connolly, K. A. et al. A reservoir of stem-like CD8+ T cells in the tumor-draining lymph node preserves the ongoing antitumor immune response. Sci. Immunol. 6, eabg7836 (2021).
Prokhnevska, N. et al. CD8+ T cell activation in cancer comprises an initial activation phase in lymph nodes followed by effector differentiation within the tumor. Immunity 56, 107–124.e5 (2023).
Duraiswamy, J. et al. Myeloid antigen-presenting cell niches sustain antitumor T cells and license PD-1 blockade via CD28 costimulation. Cancer Cell 39, 1623–1642.e20 (2021).
Giles, J. R., Globig, A. M., Kaech, S. M. & Wherry, E. J. CD8+ T cells in the cancer-immunity cycle. Immunity 56, 2231–2253 (2023).
Ugurel, S. et al. Down-regulation of HLA class II and costimulatory CD86/B7-2 on circulating monocytes from melanoma patients. Cancer Immunol. Immunother. 53, 551–559 (2004).
Tirapu, I. et al. Low surface expression of B7-1 (CD80) is an immunoescape mechanism of colon carcinoma. Cancer Res. 66, 2442–2450 (2006).
Ta, H. M. et al. LRIG1 engages ligand VISTA and impairs tumor-specific CD8+ T cell responses. Sci. Immunol. 9, eadi7418 (2024).
Vallejo, A. N. CD28 extinction in human T cells: altered functions and the program of T-cell senescence. Immunol. Rev. 205, 158–169 (2005). A review of the association between CD28 loss and T cell senescence.
Filaci, G. et al. CD8+CD28− T regulatory lymphocytes inhibiting T cell proliferative and cytotoxic functions infiltrate human cancers. J. Immunol. 179, 4323–4334 (2007).
Huang, Y. et al. Loss of CD28 expression associates with severe T-cell exhaustion in acute myeloid leukemia. Front. Immunol. 14, 1139517 (2023).
Sanchez-Lockhart, M., Graf, B. & Miller, J. Signals and sequences that control CD28 localization to the central region of the immunological synapse. J. Immunol. 181, 7639–7648 (2008).
Siokis, A., Robert, P. A., Demetriou, P., Dustin, M. L. & Meyer-Hermann, M. F-actin-driven CD28-CD80 localization in the immune synapse. Cell Rep. 24, 1151–1162 (2018).
Waite, J. C. et al. Tumor-targeted CD28 bispecific antibodies enhance the antitumor efficacy of PD-1 immunotherapy. Sci. Transl. Med. 12, eaba2325 (2020). This landmark study shows that CD28-targeted bispecific antibodies can increase the efficacy of ICB therapy.
Ishida, Y., Agata, Y., Shibahara, K. & Honjo, T. Induced expression of PD-1, a novel member of the immunoglobulin gene superfamily, upon programmed cell death. EMBO J. 11, 3887–3895 (1992).
Gibson, H. M. et al. Induction of the CTLA-4 gene in human lymphocytes is dependent on NFAT binding the proximal promoter. J. Immunol. 179, 3831–3840 (2007).
Freeman, G. J. et al. Engagement of the PD-1 immunoinhibitory receptor by a novel B7 family member leads to negative regulation of lymphocyte activation. J. Exp. Med. 192, 1027–1034 (2000).
Chan, W. et al. TCR ligand potency differentially impacts PD-1 inhibitory effects on diverse signaling pathways. J. Exp. Med. 220, e20231242 (2023).
Fife, B. T. et al. Interactions between PD-1 and PD-L1 promote tolerance by blocking the TCR-induced stop signal. Nat. Immunol. 10, 1185–1192 (2009).
Gill, A. L. et al. PD-1 blockade increases the self-renewal of stem-like CD8 T cells to compensate for their accelerated differentiation into effectors. Sci. Immunol. 8, eadg0539 (2023).
Yost, K. E. et al. Clonal replacement of tumor-specific T cells following PD-1 blockade. Nat. Med. 25, 1251–1259 (2019).
van der Leun, A. M., Thommen, D. S. & Schumacher, T. N. CD8+ T cell states in human cancer: insights from single-cell analysis. Nat. Rev. Cancer 20, 218–232 (2020).
Philip, M. et al. Chromatin states define tumour-specific T cell dysfunction and reprogramming. Nature 545, 452–456 (2017).
Im, S. J. et al. Defining CD8+ T cells that provide the proliferative burst after PD-1 therapy. Nature 537, 417–421 (2016).
Lesch, S. et al. PD-1-CD28 fusion protein strengthens mesothelin-specific TRuC T cells in preclinical solid tumor models. Cell Oncol. 46, 227–235 (2023).
Zhao, Y. et al. Tumor infiltrating lymphocyte (TIL) therapy for solid tumor treatment: progressions and challenges. Cancers 14, 4160 (2022).
Chen, R. et al. CAR-T treatment for cancer: prospects and challenges. Front. Oncol. 13, 1288383 (2023).
Baker, D. J., Arany, Z., Baur, J. A., Epstein, J. A. & June, C. H. CAR T therapy beyond cancer: the evolution of a living drug. Nature 619, 707–715 (2023).
Dwyer, C. J. et al. Ex vivo blockade of PI3K gamma or delta signaling enhances the antitumor potency of adoptively transferred CD8+ T cells. Eur. J. Immunol. 50, 1386–1399 (2020).
Funk, C. R. et al. PI3Kδ/γ inhibition promotes human CART cell epigenetic and metabolic reprogramming to enhance antitumor cytotoxicity. Blood 139, 523–537 (2022).
Weber, E. W. et al. Transient rest restores functionality in exhausted CAR-T cells through epigenetic remodeling. Science 372, eaba1786 (2021). This study shows mechanistically that transiently limiting T cell signalling could enhance the antitumour effects of CAR T cells.
Wang, Y. et al. Low-dose decitabine priming endows CAR T cells with enhanced and persistent antitumour potential via epigenetic reprogramming. Nat. Commun. 12, 409 (2021).
Jaccard, A. et al. Reductive carboxylation epigenetically instructs T cell differentiation. Nature 621, 849–856 (2023).
Maher, J., Brentjens, R. J., Gunset, G., Riviere, I. & Sadelain, M. Human T-lymphocyte cytotoxicity and proliferation directed by a single chimeric TCRζ/CD28 receptor. Nat. Biotechnol. 20, 70–75 (2002). This seminal paper describes the addition of a CD28 co-stimulatory domain to CAR constructs, which enhanced their antitumour activity compared with first-generation CAR T cells that only contained a CD3ζ signalling domain.
Gong, M. C. et al. Cancer patient T cells genetically targeted to prostate-specific membrane antigen specifically lyse prostate cancer cells and release cytokines in response to prostate-specific membrane antigen. Neoplasia 1, 123–127 (1999).
Honikel, M. M. & Olejniczak, S. H. Co-stimulatory receptor signaling in CAR-T cells. Biomolecules 12, 1303 (2022).
Cappell, K. M. & Kochenderfer, J. N. A comparison of chimeric antigen receptors containing CD28 versus 4-1BB costimulatory domains. Nat. Rev. Clin. Oncol. 18, 715–727 (2021).
Melenhorst, J. J. et al. Decade-long leukaemia remissions with persistence of CD4+ CAR T cells. Nature 602, 503–509 (2022).
Salter, A. I. et al. Phosphoproteomic analysis of chimeric antigen receptor signaling reveals kinetic and quantitative differences that affect cell function. Sci. Signal. 11, eaat6753 (2018).
Boucher, J. C. et al. CD28 costimulatory domain-targeted mutations enhance chimeric antigen receptor T-cell function. Cancer Immunol. Res. 9, 62–74 (2021).
Guedan, S. et al. Single residue in CD28-costimulated CAR-T cells limits long-term persistence and antitumor durability. J. Clin. Invest. 130, 3087–3097 (2020).
Li, G. et al. 4-1BB enhancement of CAR T function requires NF-κB and TRAFs. JCI Insight 3, e121322 (2018).
Philipson, B. I. et al. 4-1BB costimulation promotes CAR T cell survival through noncanonical NF-κB signaling. Sci. Signal. 13, eaay8248 (2020).
Agarwal, S. et al. Deletion of the inhibitory co-receptor CTLA-4 enhances and invigorates chimeric antigen receptor T cells. Immunity 56, 2388–2407.e9 (2023).
Lieberman, M. M. et al. Endogenous CD28 drives CAR T cell responses in multiple myeloma. Preprint at bioRxiv https://doi.org/10.1101/2024.03.21.586084 (2024).
Wijewarnasuriya, D., Bebernitz, C., Lopez, A. V., Rafiq, S. & Brentjens, R. J. Excessive costimulation leads to dysfunction of adoptively transferred T cells. Cancer Immunol. Res. 8, 732–742 (2020).
Mansilla-Soto, J. et al. HLA-independent T cell receptors for targeting tumors with low antigen density. Nat. Med. 28, 345–352 (2022).
Dobrin, A. et al. Synthetic dual co-stimulation increases the potency of HIT and TCR-targeted cell therapies. Nat. Cancer 5, 760–773 (2024).
Helsen, C. W. et al. The chimeric TAC receptor co-opts the T cell receptor yielding robust anti-tumor activity without toxicity. Nat. Commun. 9, 3049 (2018).
Kalaitsidou, M. et al. Signaling via a CD28/CD40 chimeric costimulatory antigen receptor (CoStAR), targeting folate receptor alpha, enhances T cell activity and augments tumor reactivity of tumor infiltrating lymphocytes. Front. Immunol. 14, 1256491 (2023).
Liu, H. et al. CD19-specific CAR T cells that express a PD-1/CD28 chimeric switch-receptor are effective in patients with PD-L1-positive B-cell lymphoma. Clin. Cancer Res. 27, 473–484 (2021).
Nisonoff, A. & Rivers, M. M. Recombination of a mixture of univalent antibody fragments of different specificity. Arch. Biochem. Biophys. 93, 460–462 (1961).
Lotze, M. T., Roberts, K., Custer, M. C., Segal, D. A. & Rosenberg, S. A. Specific binding and lysis of human melanoma by IL-2-activated cells coated with anti-T3 or anti-Fc receptor cross-linked to antimelanoma antibody: a possible approach to the immunotherapy of human tumors. J. Surg. Res. 42, 580–589 (1987).
Offner, S., Hofmeister, R., Romaniuk, A., Kufer, P. & Baeuerle, P. A. Induction of regular cytolytic T cell synapses by bispecific single-chain antibody constructs on MHC class I-negative tumor cells. Mol. Immunol. 43, 763–771 (2006).
Nagorsen, D. & Baeuerle, P. A. Immunomodulatory therapy of cancer with T cell-engaging BiTE antibody blinatumomab. Exp. Cell Res. 317, 1255–1260 (2011).
Staerz, U. D. & Bevan, M. J. Hybrid hybridoma producing a bispecific monoclonal antibody that can focus effector T-cell activity. Proc. Natl Acad. Sci. USA 83, 1453–1457 (1986).
Spiess, C., Zhai, Q. & Carter, P. J. Alternative molecular formats and therapeutic applications for bispecific antibodies. Mol. Immunol. 67, 95–106 (2015).
Klein, C., Brinkmann, U., Reichert, J. M. & Kontermann, R. E. The present and future of bispecific antibodies for cancer therapy. Nat. Rev. Drug Discov. 23, 301–319 (2024).
Ridgway, J. B., Presta, L. G. & Carter, P. ‘Knobs-into-holes’ engineering of antibody CH3 domains for heavy chain heterodimerization. Protein Eng. 9, 617–621 (1996).
Gunasekaran, K. et al. Enhancing antibody Fc heterodimer formation through electrostatic steering effects: applications to bispecific molecules and monovalent IgG. J. Biol. Chem. 285, 19637–19646 (2010).
Smith, E. J. et al. A novel, native-format bispecific antibody triggering T-cell killing of B-cells is robustly active in mouse tumor models and cynomolgus monkeys. Sci. Rep. 5, 17943 (2015).
Heiss, M. M. et al. The trifunctional antibody catumaxomab for the treatment of malignant ascites due to epithelial cancer: results of a prospective randomized phase II/III trial. Int. J. Cancer 127, 2209–2221 (2010).
Middelburg, J. et al. Overcoming challenges for CD3-bispecific antibody therapy in solid tumors. Cancers 13, 287 (2021).
Muller, P. Y., Milton, M., Lloyd, P., Sims, J. & Brennan, F. R. The minimum anticipated biological effect level (MABEL) for selection of first human dose in clinical trials with monoclonal antibodies. Curr. Opin. Biotechnol. 20, 722–729 (2009).
Skokos, D. et al. A class of costimulatory CD28-bispecific antibodies that enhance the antitumor activity of CD3-bispecific antibodies. Sci. Transl. Med. 12, eaaw7888 (2020).
Zhukovsky, E. A., Morse, R. J. & Maus, M. V. Bispecific antibodies and CARs: generalized immunotherapeutics harnessing T cell redirection. Curr. Opin. Immunol. 40, 24–35 (2016).
Morris, E. C., Neelapu, S. S., Giavridis, T. & Sadelain, M. Cytokine release syndrome and associated neurotoxicity in cancer immunotherapy. Nat. Rev. Immunol. 22, 85–96 (2022).
Amini, L. et al. Preparing for CAR T cell therapy: patient selection, bridging therapies and lymphodepletion. Nat. Rev. Clin. Oncol. 19, 342–355 (2022).
Haber, L. et al. Generation of T-cell-redirecting bispecific antibodies with differentiated profiles of cytokine release and biodistribution by CD3 affinity tuning. Sci. Rep. 11, 14397 (2021).
Ball, K., Dovedi, S. J., Vajjah, P. & Phipps, A. Strategies for clinical dose optimization of T cell-engaging therapies in oncology. MAbs 15, 2181016 (2023).
Laszlo, G. S., Gudgeon, C. J., Harrington, K. H. & Walter, R. B. T-cell ligands modulate the cytolytic activity of the CD33/CD3 BiTE antibody construct, AMG 330. Blood Cancer J. 5, e340 (2015).
Arndt, C. et al. Costimulation improves the killing capability of T cells redirected to tumor cells expressing low levels of CD33: description of a novel modular targeting system. Leukemia 28, 59–69 (2014).
Yano, H. et al. Ipilimumab augments antitumor activity of bispecific antibody-armed T cells. J. Transl. Med. 12, 191 (2014).
Wei, J. et al. CD22-targeted CD28 bispecific antibody enhances antitumor efficacy of odronextamab in refractory diffuse large B cell lymphoma models. Sci. Transl. Med. 14, eabn1082 (2022).
Sam, J. et al. CD19-CD28: an affinity-optimized CD28 agonist for combination with glofitamab (CD20-TCB) as off-the-shelf immunotherapy. Blood 143, 2152–2165 (2024).
Peper-Gabriel, J. K. et al. The PD-L1/4-1BB bispecific antibody-anticalin fusion protein PRS-344/S095012 elicits strong T-cell stimulation in a tumor-localized manner. Clin. Cancer Res. 28, 3387–3399 (2022).
Ramaswamy, M. et al. Immunomodulation of T- and NK-cell responses by a bispecific antibody targeting CD28 homolog and PD-L1. Cancer Immunol. Res. 10, 200–214 (2022).
Wang, X. B. et al. A new recombinant single chain trispecific antibody recruits T lymphocytes to kill CEA (carcinoma embryonic antigen) positive tumor cells in vitro efficiently. J. Biochem. 135, 555–565 (2004).
Liu, J. et al. A new format of single chain tri-specific antibody with diminished molecular size efficiently induces ovarian tumor cell killing. Biotechnol. Lett. 27, 1821–1827 (2005).
Wu, L. et al. Trispecific antibodies enhance the therapeutic efficacy of tumor-directed T cells through T cell receptor co-stimulation. Nat. Cancer 1, 86–98 (2020). This study describes a novel trispecific antibody that targets a tumour antigen plus TCR and CD28 to improve antitumour activity.
Chen, L. et al. A trispecific antibody induces potent tumor-directed T-cell activation and antitumor activity by CD3/CD28 co-engagement. Immunotherapy 16, 143–159 (2024).
Eastwood, D. et al. Monoclonal antibody TGN1412 trial failure explained by species differences in CD28 expression on CD4+ effector memory T-cells. Br. J. Pharmacol. 161, 512–526 (2010).
Rodig, S. J. et al. MHC proteins confer differential sensitivity to CTLA-4 and PD-1 blockade in untreated metastatic melanoma. Sci. Transl. Med. 10, eaar3342 (2018).
Roemer, M. G. M. et al. Major histocompatibility complex class II and programmed death ligand 1 expression predict outcome after programmed death 1 blockade in classic Hodgkin lymphoma. J. Clin. Oncol. 36, 942–950 (2018).
Alspach, E. et al. MHC-II neoantigens shape tumour immunity and response to immunotherapy. Nature 574, 696–701 (2019).
Angelova, M. et al. Evolution of metastases in space and time under immune selection. Cell 175, 751–765.e16 (2018).
Dong, B. et al. NK receptor signaling lowers TCR activation threshold, enhancing selective recognition of cancer cells by TAA-specific CTLs. Cancer Immunol. Res. https://doi.org/10.1158/2326-6066.CIR-24-0061 (2024).
Broz, M. L. et al. Dissecting the tumor myeloid compartment reveals rare activating antigen-presenting cells critical for T cell immunity. Cancer Cell 26, 638–652 (2014).
Cueto, F. J. et al. DNGR-1 limits Flt3L-mediated antitumor immunity by restraining tumor-infiltrating type I conventional dendritic cells. J. Immunother. Cancer 9, e002054 (2021).
Spranger, S., Dai, D., Horton, B. & Gajewski, T. F. Tumor-residing Batf3 dendritic cells are required for effector T cell trafficking and adoptive T cell therapy. Cancer Cell 31, 711–723.e4 (2017). This study reports the necessity for a rare dendritic cell subset to sustain T cell effector function and survival in the tumour microenvironment, demonstrating the need for CD28 ligands.
Duong, E. et al. Type I interferon activates MHC class I-dressed CD11b+ conventional dendritic cells to promote protective anti-tumor CD8+ T cell immunity. Immunity 55, 308–323.e9 (2022).
Mailliard, R. B. et al. Complementary dendritic cell-activating function of CD8+ and CD4+ T cells: helper role of CD8+ T cells in the development of T helper type 1 responses. J. Exp. Med. 195, 473–483 (2002).
Semino, C., Angelini, G., Poggi, A. & Rubartelli, A. NK/iDC interaction results in IL-18 secretion by DCs at the synaptic cleft followed by NK cell activation and release of the DC maturation factor HMGB1. Blood 106, 609–616 (2005). This paper shows that NK cells have a crucial role in driving the maturation of dendritic cells, which supports T cell function, by upregulating the CD28 ligands CD80 and CD86.
Castellani, P., Angelini, G., Delfino, L., Matucci, A. & Rubartelli, A. The thiol redox state of lymphoid organs is modified by immunization: role of different immune cell populations. Eur. J. Immunol. 38, 2419–2425 (2008).
Angelini, G. et al. Antigen-presenting dendritic cells provide the reducing extracellular microenvironment required for T lymphocyte activation. Proc. Natl Acad. Sci. USA 99, 1491–1496 (2002).
Shahinian, A. et al. Differential T cell costimulatory requirements in CD28-deficient mice. Science 261, 609–612 (1993).
Drake, C. G. et al. A phase I/II study of REGN5678 (anti-PSMAxCD28, a costimulatory bispecific antibody) with cemiplimab (anti-PD-1) in patients with metastatic castration-resistant prostate cancer. J. Clin. Oncol. https://doi.org/10.1200/JCO.2020.38.15_suppl.TPS5592 (2020).
Beziat, V. et al. Humans with inherited T cell CD28 deficiency are susceptible to skin papillomaviruses but are otherwise healthy. Cell 184, 3812–3828.e30 (2021).
Segal, N. H. et al. A phase 1/2 study of REGN7075 in combination with cemiplimab (cemi) in patients (pts) with advanced solid tumors: efficacy and safety results. J. Clin. Oncol. https://doi.org/10.1200/JCO.2024.42.16_suppl.2503 (2024).
Tacke, M., Hanke, G., Hanke, T. & Hunig, T. CD28-mediated induction of proliferation in resting T cells in vitro and in vivo without engagement of the T cell receptor: evidence for functionally distinct forms of CD28. Eur. J. Immunol. 27, 239–247 (1997).
Beyersdorf, N., Hanke, T., Kerkau, T. & Hunig, T. Superagonistic anti-CD28 antibodies: potent activators of regulatory T cells for the therapy of autoimmune diseases. Ann. Rheum. Dis. 64, iv91–iv95 (2005).
Luhder, F. et al. Topological requirements and signaling properties of T cell-activating, anti-CD28 antibody superagonists. J. Exp. Med. 197, 955–966 (2003).
Evans, E. J. et al. Crystal structure of a soluble CD28-Fab complex. Nat. Immunol. 6, 271–279 (2005).
Stebbings, R. et al. “Cytokine storm” in the phase I trial of monoclonal antibody TGN1412: better understanding the causes to improve preclinical testing of immunotherapeutics. J. Immunol. 179, 3325–3331 (2007).
Dharra, R., Kumar Sharma, A. & Datta, S. Emerging aspects of cytokine storm in COVID-19: the role of proinflammatory cytokines and therapeutic prospects. Cytokine 169, 156287 (2023).
Patrick, M. & Weng, N. P. Expression and regulation of telomerase in human T cell differentiation, activation, aging and diseases. Cell Immunol. 345, 103989 (2019).
Barbe-Tuana, F., Funchal, G., Schmitz, C. R. R., Maurmann, R. M. & Bauer, M. E. The interplay between immunosenescence and age-related diseases. Semin. Immunopathol. 42, 545–557 (2020).
Beider, K. et al. Molecular and functional signatures associated with CAR T cell exhaustion and impaired clinical response in patients with B cell malignancies. Cells 11, 1140 (2022).
Naismith, E. et al. Peripheral antibody concentrations are associated with highly differentiated T cells and inflammatory processes in the human bone marrow. Immun. Ageing 16, 21 (2019).
Ko, E. J. et al. Phenotype and molecular signature of CD8+ T cell subsets in T cell- mediated rejections after kidney transplantation. PLoS ONE 15, e0234323 (2020).
Mai, H. L. et al. Kidney allograft rejection is associated with an imbalance of B cells, regulatory T cells and differentiated CD28−CD8+ T cells: analysis of a cohort of 1095 graft biopsies. Front. Immunol. 14, 1151127 (2023).
Vallejo, A. N., Nestel, A. R., Schirmer, M., Weyand, C. M. & Goronzy, J. J. Aging-related deficiency of CD28 expression in CD4+ T cells is associated with the loss of gene-specific nuclear factor binding activity. J. Biol. Chem. 273, 8119–8129 (1998).
Bryl, E. et al. Modulation of CD28 expression with anti-tumor necrosis factor alpha therapy in rheumatoid arthritis. Arthritis Rheum. 52, 2996–3003 (2005).
Teteloshvili, N. et al. Involvement of microRNAs in the aging-related decline of CD28 expression by human T cells. Front. Immunol. 9, 1400 (2018).
Pawelec, G. et al. Human immunosenescence: is it infectious? Immunol. Rev. 205, 257–268 (2005).
Chen, S. et al. Associations of cytomegalovirus infection with all-cause and cardiovascular mortality in multiple observational cohort studies of older adults. J. Infect. Dis. 223, 238–246 (2021).
Leng, S. X. et al. Association of detectable cytomegalovirus (CMV) DNA in monocytes rather than positive CMV IgG serology with elevated neopterin levels in community-dwelling older adults. Exp. Gerontol. 46, 679–684 (2011).
Jackson, S. E. et al. Latent cytomegalovirus (CMV) infection does not detrimentally alter T cell responses in the healthy old, but increased latent CMV carriage is related to expanded CMV-specific T cells. Front. Immunol. 8, 733 (2017).
Lim, E. Y., Jackson, S. E. & Wills, M. R. The CD4+ T cell response to human cytomegalovirus in healthy and immunocompromised people. Front. Cell Infect. Microbiol. 10, 202 (2020).
Bauer, M. E. Accelerated immunosenescence in rheumatoid arthritis: impact on clinical progression. Immun. Ageing 17, 6 (2020).
Dvergsten, J. A. et al. Premature cell senescence and T cell receptor-independent activation of CD8+ T cells in juvenile idiopathic arthritis. Arthritis Rheum. 65, 2201–2210 (2013).
Vallejo, A. N. et al. Molecular basis for the loss of CD28 expression in senescent T cells. J. Biol. Chem. 277, 46940–46949 (2002). This paper provides a detailed demonstration that CD28 loss could be accounted for by molecular alteration in signalling pathways in T cells.
Lewis, D. E., Merched-Sauvage, M., Goronzy, J. J., Weyand, C. M. & Vallejo, A. N. Tumor necrosis factor-α and CD80 modulate CD28 expression through a similar mechanism of T-cell receptor-independent inhibition of transcription. J. Biol. Chem. 279, 29130–29138 (2004).
Hrusch, C. L. et al. T-cell phenotypes are associated with serum IgE levels in Amish and Hutterite children. J. Allergy Clin. Immunol. 144, 1391–1401.e10 (2019).
Michel, J. J., Griffin, P. & Vallejo, A. N. Functionally diverse NK-like T cells are effectors and predictors of successful aging. Front. Immunol. 7, 530 (2016).
Liaskou, E. et al. Genetic variation at the CD28 locus and its impact on expansion of pro-inflammatory CD28 negative T cells in healthy individuals. Sci. Rep. 7, 7652 (2017).
Acknowledgements
The authors thank the careful review by their colleagues L. Appleman, A. Ross, A. de Vallejo, M. Flajnik, E. Geoghegan and C. B. Thompson. This work was supported, in part, by grants from the National Institute for Allergy and Infectious Diseases (NIAID) (R01 AI155499 to S.H.O.) and by the National Cancer Institute (NCI) Roswell Park Cancer Center Support Grant (P30 CA016056), as well as that to the University of Pittsburgh Hillman Cancer Center (P30 CA047904-35).
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding authors
Ethics declarations
Competing interests
M.T.L. is the chairman of the strategic advisory board and has ownership interest in iRepertoire, Inc; he is a member of the scientific advisory boards of KSQ, Inc.; BioCytics; Zai Laboratories; Appel Sauce, Studios; and the Ludwig Institute. S.H.O. declares no competing interests. D.S. is a full-time employee and shareholder of Regeneron Pharmaceuticals, Inc.
Peer review
Peer review information
Nature Reviews Immunology thanks Tadashi Yokosuka and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Adoptive cell therapies
-
Immunotherapeutic strategy in which immune cells, often CAR T cells or TILs, that have been expanded and prepared ex vivo are infused into a patient.
- Central supramolecular activation cluster
-
The centre of an immune synapse between an immune cell and a target cell, comprising recruited receptor molecules such as the T cell receptor and CD28. Phosphatases that constrain immune activation are typically found outside this central area.
- Chimeric antigen receptor
-
(CAR). A synthetic receptor genetically engineered into immune cells to redirect their specificity towards a desired cell-surface antigen in an MHC-independent manner; clinically available CARs contain either a CD28 or 4-1BB co-stimulatory domain.
- Combination cancer immunotherapy
-
A strategy to overcome resistance to existing immunotherapies such as immune checkpoint blockade (ICB). One approach involves bispecific and/or trispecific antibodies targeting CD3 and/or co-stimulatory molecules such as CD28 together with ICB or other standard treatments. Combinations of different antibody formats and/or cell therapies are also being explored.
- Cytokine release syndrome
-
(CRS). Toxicity associated with immune overactivation. This was first observed in the setting of bacteria-induced toxic shock syndrome, following high-dose IL-2 administration and following the delivery of CD28-superagonist antibody; now, it is also commonly observed following CAR T cell administration to patients with haematological cancer and in the setting of severe COVID-19.
- Immune checkpoint blockade
-
(ICB). The therapeutic use of antagonistic antibodies to cell-surface proteins that limit T cell activation (immune checkpoints, such as PD1 and PD-L1), thereby promoting CD28 signalling and antitumour immunity.
- Immune effector cell-associated neurotoxicity syndrome
-
(ICANS). A clinical syndrome characterized by a range of neuropsychiatric symptoms that occurs following certain types of immunotherapy that induce a strong T cell response; it is probably caused by disruption to the blood–brain barrier as a result of inflammation induced by cytokine release syndrome.
- Immune synapse
-
The physical interface between an antigen-presenting cell or target cell and a T cell.
- Senescence
-
A cell state in which mature cells are limited in their proliferation and functions, associated with loss of CD28 expression in T cells.
- T cell exhaustion
-
The progressive loss of effector function and memory potential of T cells owing to persistent T cell receptor signalling.
- T cell-activating immunotherapies
-
Tumour-infiltrating lymphocytes can be activated by antibodies or by small-molecule inhibitors of E3 ligases that limit full T cell activation. Cytokines such as IL-2 or IL-15 can drive T cell proliferation.
- Tumour microenvironment
-
(TME). The TME has a ‘tissue-like’ organization comprising, in addition to the tumour itself, tumour neovasculature, stromal support elements, and recruited myeloid and lymphoid cells. It often contains immunosuppressive elements including prostaglandins, immunosuppressive cytokines and metabolic alterations such as hypoxia and an acidic milieu.
- Tumour-associated antigen
-
(TAA). Typically, a protein or glycoprotein that is found at greater abundance on tumour cells than on normal cells (for example, Her2/Neu, mesothelin and CEA).
- Tumour-infiltrating lymphocytes
-
(TILs). Lymphocytes found within tumours that are generally associated with improved prognosis in patients with cancer. Typically, tumours have one of three TIL phenotypes: inflamed (presence of TILs), excluded (TILs at a stromal barrier) or desert (absence of TILs).
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lotze, M.T., Olejniczak, S.H. & Skokos, D. CD28 co-stimulation: novel insights and applications in cancer immunotherapy. Nat Rev Immunol 24, 878–895 (2024). https://doi.org/10.1038/s41577-024-01061-1
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41577-024-01061-1
This article is cited by
-
Integrating machine learning and multi-omics analysis to explore Treg-associated programmed cell death features in clear cell renal cell carcinoma
Cancer Cell International (2026)
-
NKG2D/CD28 chimeric receptor boosts cytotoxicity and durability of CAR-T cells for solid and hematological tumors
Experimental Hematology & Oncology (2025)
-
CD40 transcriptomic expression patterns across malignancies: implications for clinical trials of CD40 agonists
Cancer Immunology, Immunotherapy (2025)