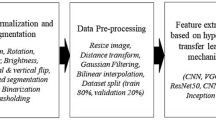
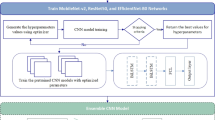
Abstract
Kidney diseases represent a substantial public health concern, with their incidence increasing markedly over the past decade. Addressing this challenge, our research introduces a sophisticated two-stage diagnostic model for enhancing the detection accuracy of various kidney pathologies. The initial stage of the proposed model comprises a novel Modified Specular-Free (MSF) technique designed to improve the visual quality of renal images. This technique adaptively enhances image details by applying targeted enhancements for more discrimination between dark and bright luminance levels. The objective of this technique is to restore critical image information and enrich color representation in diagnostically-significant areas. The enhanced images are then processed through the second stage, which involves classification using the EfficientNet-B2 deep learning architecture. Our model was rigorously compared against a suite of established pre-trained models, including VGG16, ResNet50, VGG19, DenseNet121, DenseNet169, DenseNet201, EfficientNet-B0, EfficientNet-B1, and EfficientNet-B3. Comprehensive testing revealed that our model not only outperforms these benchmarks, but does so with a notable accuracy of 98.27%. The robustness of the model was further ensured through its capability to effectively differentiate between normal renal conditions and various pathologies such as tumors, kidney stones, and cysts. This research not only demonstrates the potential of integrating advanced image enhancement techniques with cutting-edge classification models but also introduces a scalable approach for improving diagnostic accuracies in other complex medical imaging contexts.
Similar content being viewed by others
Data availability
The dataset utilized in this research is publicly accessible at: https://www.kaggle.com/datasets/nazmul0087/ct-kidney-dataset-normal-cyst-tumor-and-stone.
References
Patil, S. & Choudhary, S. Hybrid classification framework for chronic kidney disease prediction model. Imaging Sci. J. 72 (3), 367–381 (2024).
Vineetha, K. R., Maharajan, M. S., Bhagyashree, K. & Sivakumar, N. Classification of adaptive back propagation neural network along with fuzzy logic in chronic kidney disease. e-Prime-Advances Electr. Eng. Electron. Energy. 7, 100463 (2024).
Chen, X. et al. Is there a place for extracorporeal shockwave lithotripsy (ESWL) in the endoscopic era? Urolithiasis 50, 369–374 (2022).
Sudharson, S. & Kokil, P. An ensemble of deep neural networks for kidney ultrasound image classification. Comput. Methods Programs Biomed. 197, 105709 (2020).
Sharma, G. et al. Transfer Learning Empowered Multi-Class Classification of Kidney Diseases: A Deep Learning Approach. In 2024 2nd International Conference on Advancement in Computation & Computer Technologies (InCACCT) (pp. 240–245). IEEE. (2024), May.
Komura, D. & Ishikawa, S. Machine learning methods for histopathological image analysis. Comput. Struct. Biotechnol. J. 16, 34–42 (2018).
Dimitriou, N., Arandjelovic, O. & Caie, P. D. Deep learning for whole slide image analysis: an overview. Front. Med. (Lausanne). 6, 264 (2019).
Serag, A. et al. Translational AI and deep learning in diagnostic pathology. Front. Med. (Lausanne). 6, 185 (2019).
Srinidhi, C. L., Ciga, O. & Martel, A. L. Deep neural network models for computational histopathology: a survey. Med. Image Anal. 67, 101813 (2020).
Patro, K. et al. Application of Kronecker convolutions in deep learning technique for automated detection of kidney stones with coronal CT images. Inf. Sci. 640, 119005 (2023).
Abraham, A. et al. Machine learning prediction of kidney stone composition using electronic health record-derived features. J. Endourol. 36 (2), 243–250 (2022).
El Beze, J. et al. Evaluation and Understanding of automated urinary stone recognition methods. BJU Int. 130, 786–798 (2022).
Abdeltawab, H. A. et al. A deep learning framework for automated classification of histopathological kidney whole-slide images. J. Pathol. Inf. 13, 100093 (2022).
Hayat, M., Aramvith, S. & Achakulvisut, T. SEGSRNet for stereo-endoscopic image super-resolution and surgical instrument segmentation. In 2024 46th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC) (pp. 1–4). IEEE. (2024), July.
Ahmad, N. et al. Automatic segmentation of large-scale CT image datasets for detailed body composition analysis. BMC Bioinform. 24 (1), 346 (2023).
Black, K. M. et al. Deep learning computer vision algorithm for detecting kidney stone composition. BJU Int. 125 (6), 920–924 (2020).
Fitri, L. et al. Automated classification of urinary stones based on microcomputed tomography images using convolutional neural network. Physica Med. 78, 201–208 (2020).
Kazemi, Y. & Seyed Abolghasem Mirroshandel A novel method for predicting kidney stone type using ensemble learning. Artif. Intell. Med. 84, 117–126 (2018).
Chaitanya, S. M. K. & Rajesh Kumar, P. Detection of chronic kidney disease by using artificial neural networks and gravitational search algorithm. Innovations in Electronics and Communication Engineering: Proceedings of the 6th ICIECE 2017. Springer Singapore, (2019).
Bhaskar, N. & Suchetha, M. An approach for analysis and prediction of CKD using deep learning architecture. 2019 International Conference on Communication and Electronics Systems (ICCES). IEEE, (2019).
Hallscheidt, P. J. et al. Preoperative staging of renal cell carcinoma with inferior Vena Cava thrombus using multidetector CT and MRI: prospective study with histopathological correlation. J. Comput. Assist. Tomogr. 29 (1), 64–68 (2005).
Liu, J., Wang, S., Linguraru, M. G., Yao, J. & Summers, R. M. Computer-aided detection of exophytic renal lesions on non-contrast CT images. Med. Image. Anal. 19 (1), 15–29 (2015).
Feng, Z. et al. Machine learning-based quantitative texture analysis of CT images of small renal masses: differentiation of Angiomyolipoma without visible fat from renal cell carcinoma. Eur. Radiol. 28, 1625–1633 (2018).
Kocak, B. et al. Textural differences between renal cell carcinoma subtypes: machine learning-based quantitative computed tomography texture analysis with independent external validation. Eur. J. Radiol. 107, 149–157 (2018).
Muhamed Ali, A. et al. A Machine Learning Approach for the Classification of Kidney cancer Subtypes Using miRNA Genome Data. Appl. Sci. 8, 2422. https://doi.org/10.3390/app8122422 (2018).
Zhou, L. et al. A deep learning-based radiomics model for differentiating benign and malignant renal tumors. Transl Oncol. 12, 292–300 (2019).
Sun, X. Y. et al. Radiologic-radiomic machine learning models for differentiation of benign and malignant solid renal masses: comparison with expert-level radiologists. Am. J. Roentgenol. 214, W44–W54 (2020).
Tabibu, S., Vinod, P. & Jawahar, C. Pan-Renal cell carcinoma classification and survival prediction from histopathology images using deep learning. Sci. Rep. 9, 1–9 (2019).
Zabihollahy, F., Schieda, N., Krishna, S. & Ukwatta, E. Automated Classification of Solid Renal Masses on contrast-enhanced Computed Tomography Images Using Convolutional Neural Network with Decision Fusion. Eur. Radiol. 30, 5183–5190. https://doi.org/10.1007/s00330-020-06787-9 (2020).
Vendrami, C. L., McCarthy, R. J., Villavicencio, C. P. & Miller, F. H. Predicting common solid renal tumors using machine learning models of classification of radiologist-assessed magnetic resonance characteristics. Abdom. Radiol. 45, 2797–2809 (2020).
Skounakis, E. et al. ATD: A multiplatform for semiautomatic 3-D detection of kidneys and their pathology in real time. IEEE Trans. Human-Machine Syst. 44 (1), 146–153 (2013).
Kahani, M. et al. A novel approach to classify urinary stones using dual-energy kidney, ureter and bladder (DEKUB) X-ray imaging. Appl. Radiat. Isot. 164, 109267 (2020).
Liu, Y. Y., Huang, Z. H. & Ko-Wei Huang Deep learning model for computer-aided diagnosis of urolithiasis detection from kidney–ureter–bladder images. Bioengineering 9 (12), 811 (2022).
Chittora, P. et al. Prediction of chronic kidney disease-a machine learning perspective. IEEE Access. 9, 17312–17334 (2021).
Yamamoto, T. & Nakazawa, A. General improvement method of specular component separation using high-emphasis filter and similarity function. ITE Trans. Media Technol. Appl. 7 (2), 92–102 (2019).
Saha, R. et al. Combining highlight removal and low-light image enhancement technique for HDR‐like image generation. IET Image Proc. 14 (9), 1851–1861 (2020).
Wang, T. H. et al. Pseudo-multiple-exposure-based tone fusion with local region adjustment. IEEE Transactions on Multimedia 17(4), 470–484 (2015).
Majid, M., Gulzar, Y., Ayoub, S., Khan, F., Reegu, F. A., Mir, M. S., … Soomro, A.B. (2023). Enhanced transfer learning strategies for effective kidney tumor classification with CT imaging. Int J Adv Comput Sci Appl, 14, 2023.
Liu, J., Yildirim, O., Akin, O. & Tian, Y. AI-driven robust kidney and renal mass segmentation and classification on 3D CT images. Bioengineering 10 (1), 116 (2023).
Chanchal, A. K., Lal, S., Kumar, R., Kwak, J. T. & Kini, J. A novel dataset and efficient deep learning framework for automated grading of renal cell carcinoma from kidney histopathology images. Sci. Rep. 13 (1), 1–16 (2023).
Pande, S. D. & Agarwal, R. Multi-class Kidney Abnormalities Detecting Novel System Through Computed Tomography (IEEE Access, 2024).
Ye, Y. et al. Deep learning-enabled classification of kidney allograft rejection on whole slide histopathologic images. Frontiers in Immunology, 15, 1438247 (2024).
Abubeker, K. M. & Baskar, S. B2-Net: an artificial intelligence powered machine learning framework for the classification of pneumonia in chest x-ray images. Mach. Learning: Sci. Technol. 4 (1), 015036 (2023).
Yang, Y., Wei, J., Yu, Z. & Zhang, R. A trustworthy neural architecture search framework for pneumonia image classification utilizing blockchain technology. J. Supercomput. 80, 1–34. https://doi.org/10.1007/s11227-023-05541-4 (2024).
Reznichenko, A., et al. Unbiased kidney-centric molecular categorization of chronic kidney disease as a step towards precision medicine. Kidney International, 105(6), 1263–1278 (2024).
Saifullah, S. et al. Detection of chest X-ray abnormalities using CNN based on hyperparameter optimization. Eng. Proc. 56 (1), 223 (2023).
Hindarto, D. Model accuracy analysis: comparing weed detection in soybean crops with EfficientNet-B0, B1, and B2. Jurnal JTIK (Jurnal Teknologi Informasi Dan. Komunikasi). 7 (4), 734–744 (2023).
Acknowledgements
The authors extend their appreciation to the Deanship of Scientific Research at Princess Nourah bint Abdulrahman University, through the Research Groups Program Grant number RGP-1444-0054.
Funding
This work was funded by the Deanship of Scientific Research at Princess Nourah bint Abdulrahman University, through the Research Groups Program Grant no. (RGP-1444-0054).
Author information
Authors and Affiliations
Contributions
All authors are equally contributed.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
All authors contributed and accepted to submit the current work.
Consent to participate
All authors contributed and accepted to submit the current work.
Consent to publish
All authors accepted to submit and publish the submitted work.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
El-Hag, N.A., El-Shafai, W., El-Hameed, H.A.A. et al. A two-stage deep learning framework for kidney disease detection using modified specular-free imaging and EfficientNetB2. Sci Rep (2026). https://doi.org/10.1038/s41598-025-04606-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-025-04606-z