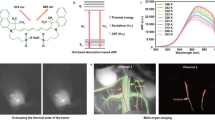
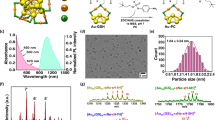
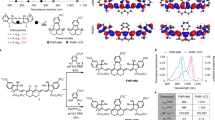
Abstract
The first near-infrared (NIR-I) spectral window (700–900 nm) enables deeper tissue penetration and reduced autofluorescence, improving sensitivity for in vivo imaging. However, the relatively low fluorescence intensity of many NIR fluorophores limits their effectiveness. Here, we report the development of gold nanodye (AuND) by conjugating gold nanospheres (AuNSs) with IRDye 800 NHS ester via polyethylene glycol (PEG) linkers and functionalizing them with epidermal growth factor receptor (EGFR) targeting antibodies (Anti-AuND). This design enhances fluorescence emission through plasmonic interactions while simultaneously exploiting the high X-ray attenuation of gold for computed tomography (CT) contrast. Using Anti-AuND, we established a multimodal imaging platform integrating CT, NIR fluorescence, and fluorescence lifetime (FLT) imaging in a preclinical mouse model of EGFR-overexpressing head and neck cancer. Anti-AuND enabled high-sensitivity, high-resolution tumor visualization in vivo. The combined imaging approach provides complementary anatomical and molecular information, highlighting the potential of Anti-AuND for early cancer detection and translational biomedical imaging applications.
Similar content being viewed by others
Data availability
Data is available under request.
References
Bai, J. W., Qiu, S. Q. & Zhang, G. J. Molecular and functional imaging in cancer-targeted therapy: current applications and future directions. Signal. Transduct. Target. Ther. 8, 89. https://doi.org/10.1038/s41392-023-01366-y (2023).
Garvey, C. J. Computed tomography in clinical practice. BMJ 324, 1077–1080. https://doi.org/10.1136/bmj.324.7345.1077 (2002).
Pansare, V. J., Hejazi, S., Faenza, W. J. & Prud’Homme, R. K. Review of long-wavelength optical and NIR imaging materials: contrast agents, fluorophores, and multifunctional nanocarriers. Chem. Mater. 24, 812–827. https://doi.org/10.1021/cm2028367 (2012).
Luo, S., Zhang, E., Su, Y., Cheng, T. & Shi, C. A review of NIR dyes in cancer targeting and imaging. Biomaterials 32, 7127–7138. https://doi.org/10.1016/j.biomaterials.2011.06.024 (2011).
Kang, H., Kang, M. W., Kashiwagi, S. & Choi, H. S. NIR fluorescence imaging and treatment for cancer immunotherapy. J. Immunother Cancer. 10, e004936. https://doi.org/10.1136/jitc-2022-004936 (2022).
Hong, G., Antaris, A. L. & Dai, H. Near-infrared fluorophores for biomedical imaging. Nat. Biomed. Eng. 1, 0010. https://doi.org/10.1038/s41551-016-0010 (2017).
Barber, W. C. et al. Combined fluorescence and X-ray tomography for quantitative in vivo detection of fluorophore. Technol. Cancer Res. Treat. 9, 45–51. https://doi.org/10.1177/153303461000900106 (2010).
Van Keulen, S. et al. The clinical application of fluorescence-guided surgery in head and neck cancer. J. Nucl. Med. 60, 758–763 (2019). https://pubmed.ncbi.nlm.nih.gov/30733319/
Argiris, A., Karamouzis, M. V., Raben, D. & Ferris, R. L. Head and neck cancer. Lancet 371, 1695–1709. https://doi.org/10.1016/S0140-6736(08)60728-X (2008).
Holzmann, K. L. K. et al. Improved ex vivo fluorescence imaging of human head and neck cancer using the peptide tracer TPP-IRDye800 targeting membrane-bound Hsp70. Br. J. Cancer. 131, 1814–1824. https://doi.org/10.1038/s41416-024-02872-8 (2024).
Voskuil, F. J. et al. Fluorescence-guided imaging for resection margin evaluation in head and neck cancer using cetuximab-800CW. Theranostics 10, 3994–4005. https://pubmed.ncbi.nlm.nih.gov/32226534/ (2020).
Chacko, N., Motiei, M., Dar, N. & Ankri, R. Cyanine-conjugated gold nanospheres for NIR fluorescence-based biomedical imaging. ACS Appl. Nano Mater. 8, 10104–10115. https://doi.org/10.1021/acsanm.5c01398 (2025). https://pubs.acs.org/doi/
Chacko, N. et al. Au nanodyes as enhanced contrast agents in wide-field NIR fluorescence lifetime imaging. Discover Nano. 19, 18. https://doi.org/10.1186/s11671-024-03958-1 (2024). https://link.springer.com/article/
Darvill, D., Centeno, A. & Xie, F. Plasmonic fluorescence enhancement by metal nanostructures. Phys. Chem. Chem. Phys. 15, 15709 (2013).
Knoblauch, R. & Geddes, C. D. Review of advances in metal-enhanced fluorescence. In Metal-Enhanced Fluorescence 253–283 https://doi.org/10.1007/978-3-030-18834-4_10 (Springer, 2019).
Theodorou, I. G. et al. Fluorescence enhancement from single gold nanostars. Nanoscale 10, 15854–15864 (2018). https://pubs.rsc.org/en/content/articlelanding/2018/nr/c8nr04567d
Strickler, S. J. & Berg, R. A. Relationship between absorption intensity and fluorescence lifetime. J. Chem. Phys. 37, 814–822. https://doi.org/10.1063/1.1733166 (1962).
Harel, M., Arbiv, U. & Ankri, R. Time-gated fluorescence lifetime imaging in the NIR regime. bioRxiv. https://doi.org/10.1101/2023.05.21.541614 (2023).
Michalet, X. AlliGator: Open-source fluorescence lifetime imaging analysis in G. SoftwareX 31, 102255. https://doi.org/10.1016/j.softx.2025.102255 (2025).
Anaki, A. et al. Impact of synthesis methods on antibody-conjugated gold nanoparticles. Nanoscale Adv. 6, 5420–5429. https://doi.org/10.1039/D4NA00134F (2024).
Patel, P. R. & De Jesus, O. CT Scan StatPearls. https://www.ncbi.nlm.nih.gov/books/NBK470414/ (2025).
Popovtzer, R. et al. Targeted gold nanoparticles enable molecular CT imaging. Nano Lett. 8, 4593–4596. https://doi.org/10.1021/nl8029114 (2008).
Escobedo, J. O., Rusin, O., Lim, S. & Strongin, R. M. NIR dyes for bioimaging. Curr. Opin. Chem. Biol. 14, 64–70. https://doi.org/10.1016/j.cbpa.2009.10.022 (2010).
Geddes, C. D. & Lakowicz, J. R. Metal-enhanced fluorescence. J. Fluoresc. 12, 121–129. https://doi.org/10.1023/A:1016875709579 (2002).
Dragan, A. I. et al. Distance dependence of metal-enhanced fluorescence. Plasmonics 7, 739–744. https://doi.org/10.1007/s11468-012-9366-0 (2012). https://link.springer.com/article/
Aslan, K. et al. Metal-enhanced fluorescence: an emerging tool. Curr. Opin. Biotechnol. 16, 55–62. https://doi.org/10.1016/j.copbio.2005.01.001 (2005).
Badshah, M. A. et al. Plasmonic nanostructures for MEF-based biosensing. Nanomaterials 10, 1749. https://doi.org/10.3390/nano10091749 (2020).
Murphy, C. J. et al. Gold nanoparticles in biology. Acc. Chem. Res. 41, 1721–1730. https://doi.org/10.1021/ar800035u (2008).
Sardar, R., Funston, A. M., Mulvaney, P. & Murray, R. W. Gold nanoparticles: past, present, and future. Langmuir 25, 13840–13851. https://doi.org/10.1021/la9019475 (2009).
Suk, J. S. et al. PEGylation for nanoparticle delivery. Adv. Drug Deliv Rev. 99, 28–51. https://doi.org/10.1016/j.addr.2015.09.012 (2016).
Soenen, S. J. et al. Cellular interactions of pegylated gold nanoparticles. Part. Part. Syst. Charact. 31, 794–800. https://doi.org/10.1002/ppsc.201300357 (2014).
Ashton, J. R. et al. EGFR-targeting antibodies for gold nanoparticle CT imaging. PLoS One. 13, e0206950. https://doi.org/10.1371/journal.pone.0206950 (2018).
Pal, R. et al. Fluorescence lifetime tumor contrast enhancement. Clin. Cancer Res. 25, 6653–6661 (2019). 10.1158/1078 – 0432.CCR-19-1686.
Mohanty, S. & Subuddhi, U. Fluorescence lifetime in biological systems. In Optical Spectroscopic and Microscopic Techniques. 77–111. https://doi.org/10.1007/978-981-16-4550-1_5 (2022).
Berezin, M. Y. & Achilefu, S. Fluorescence lifetime measurements and biological imaging. Chem. Rev. 110, 2641–2684. https://doi.org/10.1021/cr900343z (2010).
Leslie, T. K. et al. Sodium homeostasis in the tumour microenvironment. BBA Rev. Cancer. 1872, 188304 (2019). https://pubmed.ncbi.nlm.nih.gov/31348974/
Lin, J. et al. Calcium channels as therapeutic targets in HNSCC. Front. Oncol. 14, 1516357 (2024).
Rad, H. S. et al. Understanding the tumor microenvironment in HNSCC. Clin. Transl Immunol. 11, e1383 (2022). https://pubmed.ncbi.nlm.nih.gov/35686027/
Curry, J. M. et al. Tumor microenvironment in HNSCC. Semin Oncol. 41, 217–234. https://doi.org/10.1053/j.seminoncol.2014.03.003 (2014).
Shan, Y. et al. Tumor microenvironment for personalized immunotherapy in early-onset HNSCC. Front. Immunol. 15, (2025).
Ciepła, J. & Smolarczyk, R. Tumor hypoxia unveiled. Clin. Exp. Med. 24, 235 (2024). https://pubmed.ncbi.nlm.nih.gov/39361163/
Zhu, S. et al. NIR-II bioimaging via off-peak NIR-I emission. Theranostics 8, 4141–4151 (2018). https://www.thno.org/v08p4141.htm
Ye, J. et al. NIR-II-emitting organic fluorophores for bioimaging and phototherapy. Small. 21, (2025).
Acknowledgements
We gratefully acknowledge Prof. Rachela Popovtzer (Bar Ilan University) for generously providing access to her laboratory.
Funding
This work was supported by the Council for Higher Education, Israel (Grant No. RA2100000248).
Author information
Authors and Affiliations
Contributions
NC characterized the AuNDs, performed the FLI experiments, and drafted the initial version of the manuscript. MM synthesized the AuNSs and carried out the CT imaging. RR conducted the in vitro studies. RA supervised the research and revised the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chacko, N., Motiei, M., Rotbaum, R. et al. Antibody-functionalized gold nanospheres for multimodal imaging. Sci Rep (2026). https://doi.org/10.1038/s41598-026-35561-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-35561-y