Abstract
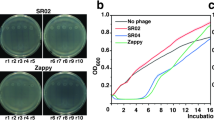
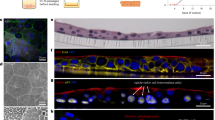
Urinary tract infections (UTIs) caused by uropathogenic Escherichia coli (UPEC) are one of the most common bacterial infections in humans. The rise of multidrug-resistant UPEC strains increases the urgent need for alternative treatment. Two diverse lytic bacteriophages (phages), SR02 and SR04, recently exhibited an in vitro anti-UPEC activity. In this study, we reported the interplay among UPEC, phages, and the microenvironment of mammalian urinary tract in UTI phage therapy using both in vitro (human bladder cell line) and in vivo (murine acute UTI) models. A gentamicin protection invasion assay was performed in UPEC-infected human bladder cells (UM-UC-3). Both monophages and the phage cocktail significantly reduced UPEC invasion into UM-UC-3 with a synergistic effect between SR02 and SR04. Female C57BL/6 mice were transurethrally infected with 107 colony-forming units of UPEC, and 2 h later, 108 plaque-forming units of monophages and cocktail were single transurethrally administered to the mouse bladder. At 24 h post-UPEC infection, the cocktail significantly reduced UPEC colonization in the mouse bladder and kidney, but not in the urine. The synergism between SR02 and SR04 was observed only in the mouse bladder. Both monophages and cocktail markedly reduced UPEC ascension into mouse kidneys without a synergism or robust tissue proinflammatory cytokine gene expression. However, increased polymorphonuclear cell infiltration was observed in the bladders of SR04-treated mice. In conclusion, we report the contribution of different host urinary tract microenvironments (urine, bladder, and kidney) in the outcomes of UTI phage therapy with two lytic phages and their combination.
Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article and the Supplementary Information files.
References
Yang, X. et al. Disease burden and long-term trends of urinary tract infections: A worldwide report. Front. Public Health 10, 888205 (2022).
Advani, S. D. et al. State-of-the-art review: Recurrent uncomplicated urinary tract infections in women. Clin. Infect. Dis. 80(3), e31–e42 (2025).
Deltourbe, L. et al. The impact of biological sex on diseases of the urinary tract. Mucosal Immunol. 15(5), 857–866 (2022).
Azimzadeh, P. N. et al. Mechanisms of uropathogenic E. coli mucosal association in the gastrointestinal tract. Sci. Adv. 11(5), eadp7066 (2025).
Barber, A. E. et al. Urinary tract infections: current and emerging management strategies. Clin. Infect. Dis. 57(5), 719–724 (2013).
Ozturk, R. & Murt, A. Epidemiology of urological infections: A global burden. World J. Urol. 38(11), 2669–2679 (2020).
Jo, J. K. et al. A comparative study of urinary tests and cultures for the effectiveness of fosfomycin in catheter-related urinary tract infections. J. Clin. Med. 11(23) (2022).
Farahat, E. M. et al. Characterization of multidrug-resistant Escherichia coli isolated from clinical (UPEC) and environmental samples. Mol. Biol. Rep. 52(1), 535 (2025).
Medugu, N. et al. Whole genome sequencing reveals virulence-mobile element linkages and phylogenetic diversity in multidrug-resistant Escherichia coli from Nigeria. Front. Microbiol. 16, 1579175 (2025).
Pootong, A. et al. Phylogenetic background, drug susceptibility and virulence factors of uropathogenic E. coli isolate in a tertiary university hospital in central Thailand. Trop. Biomed. 35(1), 195–204 (2018).
Ukoaka, B. M. et al. Updated WHO list of emerging pathogens for a potential future pandemic: Implications for public health and global preparedness. Infez. Med. 32(4), 463–477 (2024).
Gordillo Altamirano, F. L. & Barr, J. J. Phage therapy in the postantibiotic era. Clin. Microbiol. Rev. 32(2) (2019).
Delbruck, M. The growth of bacteriophage and lysis of the host. J. Gen. Physiol. 23(5), 643–660 (1940).
Lamy-Besnier, Q. et al. Prophylactic administration of a bacteriophage cocktail is safe and effective in reducing Salmonella enterica serovar Typhimurium burden in vivo. Microbiol. Spectr. 9(1), e0049721 (2021).
Zulk, J. J., Patras, K. A. & Maresso, A. W. The rise, fall, and resurgence of phage therapy for urinary tract infection. EcoSal Plus 12(1), eesp00292023 (2024).
Gonzalez-Villalobos, E. et al. |Isolation and characterization of novel bacteriophages as a potential therapeutic option for Escherichia coli urinary tract infections. Appl. Microbiol. Biotechnol. 105(13), 5617–5629 (2021).
Kongsomboonchoke, P. et al. Rapid formulation of a genetically diverse phage cocktail targeting uropathogenic Escherichia coli infections using the UTI89 model. Sci. Rep. 15(1), 12832 (2025).
Tamadonfar, K. O. et al. Reaching the end of the line: urinary tract infections. Microbiol. Spectr. 7(3) (2019).
Zhou, Y. et al. Urinary tract infections caused by uropathogenic Escherichia coli: Mechanisms of infection and treatment options. Int. J. Mol. Sci. 24(13), 10537 (2023).
Sati, H. et al. The WHO Bacterial Priority Pathogens List 2024: A prioritisation study to guide research, development, and public health strategies against antimicrobial resistance. Lancet Infect. Dis. (2025).
Giuliano, G. et al. Urinary tract infections caused by Gram-negative bacteria in elderly hospitalized patients: Epidemiology, clinical features and outcomes in the era of antimicrobial resistance. J. Glob. Antimicrob. Resist. 44, 116–126 (2025).
Morgan, C. J. et al. Phage therapy for urinary tract infections: progress and challenges ahead. Int. Urogynecol. J. (2025).
Larcher, R. et al. Phage therapy in patients with urinary tract infections: A systematic review. Expert Rev. Anti Infect. Ther. 1–12 (2025).
Chan, B. K., Abedon, S. T. & Loc-Carrillo, C. Phage cocktails and the future of phage therapy. Future Microbiol. 8(6), 769–783 (2013).
Niu, Y. D. et al. Efficacy of individual bacteriophages does not predict efficacy of bacteriophage cocktails for control of Escherichia coli O157. Front. Microbiol. 12, 616712 (2021).
Alteri, C. J. & Mobley, H. L. Metabolism and fitness of urinary tract pathogens. Microbiol. Spectr. 3(3) (2015).
Ragnarsdottir, B. et al. Genetics of innate immunity and UTI susceptibility. Nat. Rev. Urol. 8(8), 449–468 (2011).
Li, X. et al. Characterization of a novel phage vB_EcoP_P64441 and its potential role in controlling uropathogenic Escherichia coli (UPEC) and biofilms formation. Virology 609, 110570 (2025).
Hussein, A. H. et al. Characterization and therapeutic potential of phage vB_Eco_ZCEC08 against multidrug-resistant uropathogenic Escherichia coli. BMC Microbiol. 25(1), 221 (2025).
Xing, Y. et al. Phage-induced protection against lethal bacterial reinfection. Proc. Natl. Acad. Sci. USA 122(22), e2423286122 (2025).
Huh, H. et al. Bacteriophage interactions with mammalian tissue: Therapeutic applications. Adv. Drug Deliv. Rev. 145, 4–17 (2019).
Nishikawa, H. et al. T-even-related bacteriophages as candidates for treatment of Escherichia coli urinary tract infections. Arch. Virol. 153(3), 507–515 (2008).
Mijbel Ali, B. et al. A novel phage cocktail therapy of the urinary tract infection in a mouse model. Arch. Razi Inst. 76(5), 1229–1236 (2021).
Dufour, N. et al. Bacteriophage LM33_P1, a fast-acting weapon against the pandemic ST131-O25b:H4 Escherichia coli clonal complex. J. Antimicrob. Chemother. 71(11), 3072–3080 (2016).
Wu, R. et al. Bacteriophage P2–71: A promising therapeutic against multidrug-resistant Proteus mirabilis in urinary tract infections. Front. Vet. Sci. 11, 1445264 (2024).
Tothova, L. et al. Phage therapy of Cronobacter-induced urinary tract infection in mice. Med. Sci. Monit. 17(7), BR173–BR178 (2011).
Vera-Mansilla, J. et al. Isolation and characterization of novel lytic phages infecting multidrug-resistant Escherichia coli. Microbiol. Spectr. 10(1), e0167821 (2022).
Bhargava, K. et al. Bacteriophage therapy for Escherichia coli-induced urinary tract infection in rats. Future Microbiol. 18, 323–334 (2023).
Hannan, T. J. et al. Early severe inflammatory responses to uropathogenic E. coli predispose to chronic and recurrent urinary tract infection. PLoS Pathog. 6(8), e1001042 (2010).
Hopkins, W. J. et al. Time course and host responses to Escherichia coli urinary tract infection in genetically distinct mouse strains. Infect. Immun. 66(6), 2798–2802 (1998).
Wright, K. J., Seed, P. C. & Hultgren, S. J. Uropathogenic Escherichia coli flagella aid in efficient urinary tract colonization. Infect. Immun. 73(11), 7657–7668 (2005).
Hsieh, Y. J., Fu, C. L. & Hsieh, M. H. Helminth-induced interleukin-4 abrogates invariant natural killer T cell activation-associated clearance of bacterial infection. Infect. Immun. 82(5), 2087–2097 (2014).
Winter, S. E. et al. Contribution of flagellin pattern recognition to intestinal inflammation during Salmonella enterica serotype typhimurium infection. Infect. Immun. 77(5), 1904–1916 (2009).
Sarichai, P. et al. Pathogenicity of clinical Salmonella enterica serovar Typhimurium isolates from Thailand in a mouse colitis model. Microbiol. Immunol. 64(10), 679–693 (2020).
Tolstrup, L. K. et al. Cystitis: Significant associations between pathology, histology, and quantitative bacteriology in sows, a cross-sectional study. Porcine Health Manag. 9(1), 41 (2023).
Acknowledgements
We would like to thank the Research Administration Section, Faculty of Medicine, Chiang Mai University, and Support the Children Foundation for laboratory equipment support. We are also grateful to Pattida Kongsomboonchoke for her assistance with inter-laboratory transfers and for her helpful suggestions on phage preparation.
Funding
This research was funded by the Faculty of Medicine, Chiang Mai University (Grant. No. MIC-132/2567 (P.M. Mongkolkarvin and P.T. Thiennimitr). This research was also partially supported by the Center of Excellence in Microbial Diversity and Sustainable Utilization, Chiang Mai University. C.S. and P.M. were supported by scholarships from the Faculty of Medicine, Chiang Mai University. V.C. would like to acknowledge Thailand Science Research and Innovation Fund Chulalongkorn University (FF69).
Author information
Authors and Affiliations
Contributions
Conceptualization: PT, PM, VC Methodology: PT, PM, CS, PN Validation: PT, PM, VC Formal analysis: PT, PM, CS, SB Investigations: PT, PM, CS, WS, SB, IEI Resources: PT, VC, MHH Supervision: PT & VC Visualization: PT, PM, WS Data Curation: PT, PM, VS Writing – Original Draft Preparation: PT, PM, WS Writing – Review & Editing: PT, PM, VC, MHH Funding acquisition: PT & VC Project administration: PT.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Mongkolkarvin, P., Sukjoi, C., Suyapoh, W. et al. Cocktail of genetically diverse lytic phages reduces uropathogenic Escherichia coli colonization in mouse urinary tract. Sci Rep (2026). https://doi.org/10.1038/s41598-026-39877-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-39877-7