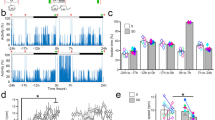
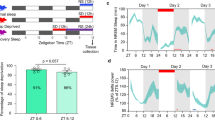
Abstract
Growing evidence shows that sleep can be enhanced in a non-invasive, drug-free manner through sensory stimulation. While modalities such as auditory and vestibular stimulation effectively increase sleep, the cognitive and cellular consequences of such enhancement remain unclear. Here, we investigated the effects of vestibular stimulation via rocking on sleep architecture, motor learning, cortical gene expression, and synaptic organization in the motor cortex. Eleven consecutive days of rocking enhanced sleep in mice, increasing both sleep duration and consolidation. These improvements were accompanied by greater motor learning performance, and the degree of learning enhancement positively correlated with total sleep amount. At the molecular level, improved learning was associated with transcriptional changes in genes involved in glutamatergic signalling and synaptic plasticity, alongside an increased density of excitatory synapses in the motor cortex. Together, these findings demonstrate that sleep enhancement via rocking facilitates learning by promoting neuroplastic mechanisms in the motor cortex.
Similar content being viewed by others
Data availability
Source data are included in the article and/or Supplementary Data file. Gene expression data are available at the NCBI GEO database (GSE 316798). All other raw data are available from the corresponding author upon reasonable request.
Code availability
All 3D printing files for the complex wheel and custom algorithms for data collection and extraction are available at https://github.com/RickAvv/ActiWheel. Codes for EEG and motion analysis are available at https://github.com/BSRLab.
References
Tononi, G. & Cirelli, C. Sleep and the price of plasticity: from synaptic and cellular homeostasis to memory consolidation and integration. Neuron 81, 12–34 (2014).
Walsh, J. K. Enhancement of slow wave sleep: implications for insomnia. J. Clin. Sleep. Med. 5, S27–S32 (2009).
Grimaldi, D., Papalambros, N. A., Zee, P. C. & Malkani, R. G. Neurostimulation techniques to enhance sleep and improve cognition in aging. Neurobiol. Dis. 141, 104865 (2020).
Malkani, R. G. & Zee, P. C. Brain stimulation for improving sleep and memory. Sleep. Med. Clin. 15, 101–115 (2020).
Marshall, L., Helgadóttir, H., Mölle, M. & Born, J. Boosting slow oscillations during sleep potentiates memory. Nature 444, 610–613 (2006).
Massimini, M. et al. Triggering sleep slow waves by transcranial magnetic stimulation. Proc. Natl. Acad. Sci. USA 104, 8496–8501 (2007).
Esfahani, M. J. et al. Closed-loop auditory stimulation of sleep slow oscillations: basic principles and best practices. Neurosci. Biobehav Rev. 153, 105379 (2023).
Tononi, G., Riedner, B. A., Hulse, B. K., Ferrarelli, F. & Sarasso, S. Enhancing sleep slow waves with natural stimuli. Medicamundi 54, 73–78 (2010).
Ngo, H.-V. V., Martinetz, T., Born, J. & Mölle, M. Auditory closed-loop stimulation of the sleep slow oscillation enhances memory. Neuron 78, 545–553 (2013).
Diep, C. et al. Acoustic enhancement of slow wave sleep on consecutive nights improves alertness and attention in chronically short sleepers. Sleep. Med. 81, 69–79 (2021).
Bellesi, M., Riedner, B. A., Garcia-Molina, G. N., Cirelli, C. & Tononi, G. Enhancement of sleep slow waves: underlying mechanisms and practical consequences. Front. Syst. Neurosci. 8, 208 (2014).
Carro-Domínguez, M. et al. Pupil size reveals arousal level fluctuations in human sleep. Nat. Commun. 16, 2070 (2025).
Kompotis, K. et al. Rocking promotes sleep in mice through rhythmic stimulation of the vestibular system. Curr. Biol. 29, 392–401.e4 (2019).
Perrault, A. A. et al. Whole-night continuous rocking entrains spontaneous neural oscillations with benefits for sleep and memory. Curr. Biol. 29, 402–411.e3 (2019).
Zhang, L. et al. Rocking during sleep reduces motor deficits and beta-amyloid levels in an Alzheimer’s mouse model. iScience 28, 112036 (2025).
Wunderlin, M. et al. Modulating overnight memory consolidation by acoustic stimulation during slow-wave sleep: a systematic review and meta-analysis. Sleep 44, zsaa296 (2021).
Choi, J., Won, K. & Jun, S. C. Acoustic stimulation following sleep spindle activity may enhance procedural memory consolidation during a nap. IEEE Access 7, 56297–56307 (2019).
Weigenand, A., Mölle, M., Werner, F., Martinetz, T. & Marshall, L. Timing matters: open-loop stimulation does not improve overnight consolidation of word pairs in humans. Eur. J. Neurosci. 44, 2357–2368 (2016).
Inami, S. & Koh, K. Sleep induced by mechanosensory stimulation provides cognitive and health benefits in Drosophila. Sleep 47, zsae226 (2024).
Maret, S., Faraguna, U., Nelson, A. B., Cirelli, C. & Tononi, G. Sleep and waking modulate spine turnover in the adolescent mouse cortex. Nat. Neurosci. 14, 1418–1420 (2011).
Bellesi, M. et al. Effects of sleep and wake on oligodendrocytes and their precursors. J. Neurosci. 33, 14288–14300 (2013).
Szklarczyk, D. et al. The STRING database in 2023: protein-protein association networks and functional enrichment analyses for any sequenced genome of interest. Nucleic Acids Res. 51, D638–D646 (2023).
Spano, G. M. et al. Sleep deprivation by exposure to novel objects increases synapse density and axon-spine interface in the hippocampal CA1 region of adolescent mice. J. Neurosci. 39, 6613–6625 (2019).
Nagai, H. et al. Sleep consolidates motor learning of complex movement sequences in mice. Sleep 40, zsw059 (2017).
Fischer, S., Hallschmid, M., Elsner, A. L. & Born, J. Sleep forms memory for finger skills. Proc. Natl. Acad. Sci. USA 99, 11987–11991 (2002).
Walker, M. P., Brakefield, T., Morgan, A., Hobson, J. A. & Stickgold, R. Practice with sleep makes perfect: sleep-dependent motor skill learning. Neuron 35, 205–211 (2002).
Korman, M., Raz, N., Flash, T. & Karni, A. Multiple shifts in the representation of a motor sequence during the acquisition of skilled performance. Proc. Natl. Acad. Sci. USA 100, 12492–12497 (2003).
Korman, M. et al. Daytime sleep condenses the time course of motor memory consolidation. Nat. Neurosci. 10, 1206–1213 (2007).
Moreira, C. G. et al. Closed-loop auditory stimulation method to modulate sleep slow waves and motor learning performance in rats. eLife 10, e68043 (2021).
Nicholls, R. E. et al. mGluR2 acts through inhibitory Galpha subunits to regulate transmission and long-term plasticity at hippocampal mossy fiber-CA3 synapses. Proc. Natl. Acad. Sci. USA 103, 6380–6385 (2006).
Ma, J. et al. GRM2 regulates functional integration of adult-born DGCs by paradoxically modulating MEK/ERK1/2 pathway. J. Neurosci. 43, 2822–2836 (2023).
Ahnaou, A., Ver Donck, L. & Drinkenburg, W. H. I. M. Blockade of the metabotropic glutamate (mGluR2) modulates arousal through vigilance states transitions: evidence from sleep-wake EEG in rodents. Behav. Brain Res. 270, 56–67 (2014).
Pritchett, D. et al. Deletion of metabotropic glutamate receptors 2 and 3 (mGlu2 & mGlu3) in mice disrupts sleep and wheel-running activity, and increases the sensitivity of the circadian system to light. PLoS ONE 10, e0125523 (2015).
Bjorness, T. E. et al. An essential role for MEF2C in the cortical response to loss of sleep in mice. Elife 9, e58331 (2020).
Cosgrove, D. et al. Genes influenced by MEF2C contribute to neurodevelopmental disease via gene expression changes that affect multiple types of cortical excitatory neurons. Hum. Mol. Genet. 30, 961–970 (2021).
Adolfsen, B. & Littleton, J. T. Genetic and molecular analysis of the synaptotagmin family. Cell Mol. Life Sci. 58, 393–402 (2001).
Kuhn, M. et al. Sleep recalibrates homeostatic and associative synaptic plasticity in the human cortex. Nat. Commun. 7, 12455 (2016).
Holst, S. C. et al. Functional polymorphisms in dopaminergic genes modulate neurobehavioral and neurophysiological consequences of sleep deprivation. Sci. Rep. 7, 45982 (2017).
Wu, M. et al. Dopamine pathways mediating affective state transitions after sleep loss. Neuron 112, 141–154.e8 (2024).
Wang, G., Grone, B., Colas, D., Appelbaum, L. & Mourrain, P. Synaptic plasticity in sleep: learning, homeostasis and disease. Trends Neurosci. 34, 452–463 (2011).
Huang, S. & Sigrist, S. J. Presynaptic and postsynaptic long-term plasticity in sleep homeostasis. Curr. Opin. Neurobiol. 69, 1–10 (2021).
Alonso-Nanclares, L. et al. Perisomatic glutamatergic axon terminals: a novel feature of cortical synaptology revealed by vesicular glutamate transporter 1 immunostaining. Neuroscience 123, 547–556 (2004).
Melone, M., Burette, A. & Weinberg, R. J. Light microscopic identification and immunocytochemical characterization of glutamatergic synapses in brain sections. J. Comp. Neurol. 492, 495–509 (2005).
Mao, H., Hamodeh, S. & Sultan, F. Quantitative comparison of vesicular glutamate transporters in rat deep cerebellar nuclei. Neuroscience 376, 152–161 (2018).
Mokin, M. & Keifer, J. Quantitative analysis of immunofluorescent punctate staining of synaptically localized proteins using confocal microscopy and stereology. J. Neurosci. Methods 157, 218–224 (2006).
Danielson, E. & Lee, S. H. SynPAnal: software for rapid quantification of the density and intensity of protein puncta from fluorescence microscopy images of neurons. PLoS ONE 9, e115298 (2014).
Carvalhais, L. G., Martinho, V. C., Ferreiro, E. & Pinheiro, P. S. Unraveling the nanoscopic organization and function of central mammalian presynapses with super-resolution microscopy. Front. Neurosci. 14, 578409 (2020).
Tononi, G. & Cirelli, C. Sleep function and synaptic homeostasis. Sleep. Med. Rev. 10, 49–62 (2006).
de Vivo, L. et al. Ultrastructural evidence for synaptic scaling across the wake/sleep cycle. Science 355, 507–510 (2017).
Cirelli, C. & Tononi, G. The why and how of sleep-dependent synaptic down-selection. Semin. Cell Dev. Biol. 125, 91–100 (2022).
Ramanathan, D. S., Gulati, T. & Ganguly, K. Sleep-dependent reactivation of ensembles in motor cortex promotes skill consolidation. PLoS Biol. 13, e1002263 (2015).
Kam, K., Pettibone, W. D., Shim, K., Chen, R. K. & Varga, A. W. Dynamics of sleep spindles and coupling to slow oscillations following motor learning in adult mice. Neurobiol. Learn Mem. 166, 107100 (2019).
Eckert, M. J., McNaughton, B. L. & Tatsuno, M. Neural ensemble reactivation in rapid eye movement and slow-wave sleep coordinate with muscle activity to promote rapid motor skill learning. Philos. Trans. R. Soc. Lond. B Biol. Sci. 375, 20190655 (2020).
Adler, A. et al. Sleep promotes the formation of dendritic filopodia and spines near learning-inactive existing spines. Proc. Natl. Acad. Sci. USA 118, e2114856118 (2021).
Miyamoto, D., Marshall, W., Tononi, G. & Cirelli, C. Net decrease in spine-surface GluA1-containing AMPA receptors after post-learning sleep in the adult mouse cortex. Nat. Commun. 12, 2881 (2021).
Squarcio, F. et al. Ultrastructural effects of learning and post-learning sleep on the dorsal striatum. Sleep 48, zsaf179 (2025).
Sulaman, B. A. et al. VTA dopaminergic neuronal activity during NREM sleep is modulated by learning and facilitates motor memory consolidation. Sci. Adv. 11, eadw7427 (2025).
Cusinato, R. et al. Workflow for the unsupervised clustering of sleep stages identifies light and deep sleep in electrophysiological recordings in mice. J. Neurosci. Methods 408, 110155 (2024).
Acknowledgements
This work was supported by (Wellcome Trust 215267/Z/19/Z to MB, 217546/Z/19/Z to LdV) and the Armenise-Harvard Foundation (CDA for LdV).
Author information
Authors and Affiliations
Contributions
Conceptualization: M.B., L.d.V. Investigation: R.S., L.S., S.G., R.A., E.B., L.d.V., and M.B. Wheel design and testing: R.A. Writing original draft: M.B., L.d.V. Writing, review and editing: All authors. Project supervision and funding: M.B., L.d.V.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Biology thanks the anonymous reviewers for their contribution to the peer review of this work. Primary handling editor: Benjamin Bessieres. [A peer review file is available.]
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Simayi, R., Santoni, L., Galizia, S. et al. Rocking-induced sleep enhancement promotes motor learning through transcriptional and synaptic remodelling. Commun Biol (2026). https://doi.org/10.1038/s42003-026-09666-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42003-026-09666-z