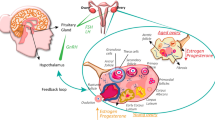
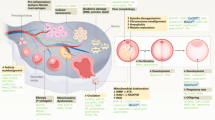
Abstract
The female reproductive axis is one of the first organ systems to age, which has consequences for fertility and overall health. Here, we provide a comprehensive overview of the biological process of female reproductive aging across reproductive organs, tissues and cells based on research with widely used physiologic aging mouse models, and describe the mechanisms that underpin these phenotypes. Overall, aging is associated with dysregulation of the hypothalamic–pituitary–ovarian axis, perturbations of the ovarian stroma, reduced egg quantity and quality, and altered uterine morphology and function that contributes to reduced capacity for fertilization and impaired embryo development. Ultimately, these age-related phenotypes contribute to altered pregnancy outcomes and adverse consequences in offspring. Conserved mechanisms of aging, as well as those unique to the reproductive system, underlie these phenotypes. The knowledge of such mechanisms will lead to development of therapeutics to extend female reproductive longevity and support endocrine function and overall health.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Broekmans, F. J., Soules, M. R. & Fauser, B. C. Ovarian aging: mechanisms and clinical consequences. Endocr. Rev. 30, 465–493 (2009).
Traub, M. L. & Santoro, N. Reproductive aging and its consequences for general health. Ann. N. Y Acad. Sci. 1204, 179–187 (2010).
Gameiro, C. M., Romao, F. & Castelo-Branco, C. Menopause and aging: changes in the immune system–a review. Maturitas 67, 316–320 (2010).
Adashi, E. Y. & Gutman, R. Delayed childbearing as a growing, previously unrecognized contributor to the national plural birth excess. Obstet. Gynecol. 132, 999–1006 (2018).
Martinez, G. M. & Daniels, K. Fertility of men and women aged 15–49 in the United States: National Survey of Family Growth, 2015–2019. Natl Health Stat. Report 1–22 (2023).
Gold, E. B. The timing of the age at which natural menopause occurs. Obstet. Gynecol. Clin. North Am. 38, 425–440 (2011).
Lu, H. et al. Current animal model systems for ovarian aging research. Aging Dis. 13, 1183–1195 (2022).
Magden, E. R., Mansfield, K. G., Simmons, J. H. & Abee, C. R. in Laboratory Animal Medicine (Third Edition) (eds. J. G. Fox et al.) 771–930 (Academic Press, 2015).
Hickman, D. L., Johnson, J., Vemulapalli, T. H., Crisler, J. R. & Shepherd, R. in Principles of Animal Research for Graduate and Undergraduate Students (eds. Suckow, M.A. & Stewart, K.L.), 117–175 (Academic Press, 2017).
Rydell-Törmänen, K. & Johnson, J. R. The applicability of mouse models to the study of human disease. Methods Mol. Biol. 1940, 3–22 (2019).
Danilovich, N. & Ram Sairam, M. Recent female mouse models displaying advanced reproductive aging. Exp. Gerontol. 41, 117–122 (2006).
Gilmer, G. et al. Female aging: when translational models don't translate. Nat. Aging 3, 1500–1508 (2023).
Kumar, T. R. Fshb knockout mouse model, two decades later and into the future. Endocrinology 159, 1941–1949 (2018).
Winkler, I. & Goncalves, A. Do mammals have menopause? Cell 186, 4729–4733 (2023).
Koebele, S. V. & Bimonte-Nelson, H. A. Modeling menopause: the utility of rodents in translational behavioral endocrinology research. Maturitas 87, 5–17 (2016).
Kawamura, T. et al. Identification of genes associated with endometrial cell ageing. Mol. Hum. Reprod. 27, gaaa078 (2021).
Parkening, T. A., Lau, I. F., Saksena, S. K. & Chang, M. C. Circulating plasma levels of pregnenolone, progesterone, estrogen, luteinizing hormone, and follicle stimulating hormone in young and aged C57BL/6 mice during various stages of pregnancy. J. Gerontol. 33, 191–196 (1978).
Parkening, T. A., Collins, T. J. & Smith, E. R. Plasma and pituitary concentrations of LH, FSH, and prolactin in aging C57BL/6 mice at various times of the estrous cycle. Neurobiol. Aging 3, 31–35 (1982).
Pal, D., Collins, T. J. & Parkening, T. A. Levels of alpha-inhibin in aging female mice. Biol. Reprod. 45, 869–875 (1991).
Bernstein, L. R. et al. Shortened estrous cycle length, increased FSH levels, FSH variance, oocyte spindle aberrations, and early declining fertility in aging senescence-accelerated mouse prone-8 (SAMP8) mice: concomitant characteristics of human midlife female reproductive aging. Endocrinology 155, 2287–2300 (2014).
Bergman, M. D., Karelus, K., Felicio, L. S. & Nelson, J. F. Differential effects of aging on estrogen receptor dynamics in hypothalamus, pituitary and uterus of the C57BL/6J mouse. J. Steroid Biochem. 33, 1027–1033 (1989).
Bergman, M. D., Karelus, K., Felicio, L. S. & Nelson, J. F. Age-related alterations in estrogen receptor dynamics are independent of cycling status in middle-aged C57BL/6J mice. J. Steroid Biochem. Mol. Biol. 38, 127–133 (1991).
Flurkey, K., Gee, D. M., Sinha, Y. N., Wisner, J. R. Jr. & Finch, C. E. Age effects on luteinizing hormone, progesterone and prolactin in proestrous and acyclic C57BL/6J mice. Biol. Reprod. 26, 835–846 (1982).
Gee, D. M., Flurkey, K. & Finch, C. E. Aging and the regulation of luteinizing hormone in C57BL/6J mice: impaired elevations after ovariectomy and spontaneous elevations at advanced ages. Biol. Reprod. 28, 598–607 (1983).
Finch, C. E., Felicio, L. S., Mobbs, C. V. & Nelson, J. F. Ovarian and steroidal influences on neuroendocrine aging processes in female rodents. Endocr. Rev. 5, 467–497 (1984).
Handelsman, D. J. et al. Ultrasensitive serum estradiol measurement by liquid chromatography-mass spectrometry in postmenopausal women and mice. J. Endocr. Soc. 4, bvaa086 (2020).
Albrecht, E. D., Koos, R. D. & Wehrenberg, W. B. Ovarian delta-5,3-beta-hydroxysteroid dehydrogenase and cholesterol in aged mouse during pregnancy. Biol. Reprod. 13, 158–162 (1975).
Albrecht, E. D., Koos, R. D. & Gottlieb, S. F. Pregnant mare serum and human chorionic gonadotropin stimulate ovarian delta5-3beta-hydroxysteroid dehydrogenase in aged mice. Fertil. Steril. 28, 762–765 (1977).
Wehrenberg, W. B., Gottlieb, S. F. & Albrecht, E. D. Ageing and ovarian delta 5-3 beta-hydrosteroid dehydrogenase in the pregnant mouse. J. Endocrinol. 70, 183–187 (1976).
Kevenaar, M. E. et al. Serum anti-Mullerian hormone levels reflect the size of the primordial follicle pool in mice. Endocrinology 147, 3228–3234 (2006).
Tappa, B., Amao, H., Ogasa, A. & Takahashi, K. W. Changes in the estrous cycle and number of ovulated and fertilized ova in aging female IVCS mice. Jikken Dobutsu 38, 115–119 (1989).
Boot, L. M., Muhlbock, O. & Thung, P. J. Senile changes in the oestrous cycle and in ovarian structure in some inbred strains of mice. Acta Endocrinol. 23, 8–32 (1956).
Nelson, J. F., Felicio, L. S., Osterburg, H. H. & Finch, C. E. Altered profiles of estradiol and progesterone associated with prolonged estrous cycles and persistent vaginal cornification in aging C57BL/6J mice. Biol. Reprod. 24, 784–794 (1981).
Nelson, J. F., Felicio, L. S., Randall, P. K., Sims, C. & Finch, C. E. A longitudinal study of estrous cyclicity in aging C57BL/6J mice: I. Cycle frequency, length and vaginal cytology. Biol. Reprod. 27, 327–339 (1982).
Felicio, L. S., Nelson, J. F. & Finch, C. E. Longitudinal studies of estrous cyclicity in aging C57BL/6J mice: II. Cessation of cyclicity and the duration of persistent vaginal cornification. Biol. Reprod. 31, 446–453 (1984).
Weixelbaumer, K. M. et al. Estrus cycle status defined by vaginal cytology does not correspond to fluctuations of circulating estrogens in female mice. Shock 41, 145–153 (2014).
Cora, M. C., Kooistra, L. & Travlos, G. Vaginal cytology of the laboratory rat and mouse: review and criteria for the staging of the estrous cycle using stained vaginal smears. Toxicol. Pathol. 43, 776–793 (2015).
Takasugi, N. Cytological basis for permanent vaginal changes in mice treated neonatally with steroid hormones. Int. Rev. Cytol. 44, 193–224 (1976).
Green, J. A. Some effects of advancing age on the histology and reactivity of the mouse ovary. Anat. Rec. 129, 333–347 (1957).
Jones, E. C. & Krohn, P. L. Relationships between age, numbers of oocytes and fertility in virgin and multiparous mice. J. Endocrinol. 21, 469–495 (1961).
Faddy, M. J., Jones, E. C. & Edwards, R. G. An analytical model for ovarian follicle dynamics. J. Exp. Zool. 197, 173–185 (1976).
Kalich-Philosoph, L. et al. Cyclophosphamide triggers follicle activation and "burnout"; AS101 prevents follicle loss and preserves fertility. Sci. Transl. Med. 5, 185ra162 (2013).
Goldman, K. N. et al. mTORC1/2 inhibition preserves ovarian function and fertility during genotoxic chemotherapy. Proc. Natl Acad. Sci. USA 114, 3186–3191 (2017).
Lawley, S. D. & Johnson, J. Why is there an "oversupply" of human ovarian follicles?. Biol. Reprod. 108, 814–821 (2023).
Dou, X. et al. Short-term rapamycin treatment increases ovarian lifespan in young and middle-aged female mice. Aging Cell 16, 825–836 (2017).
Selesniemi, K., Lee, H. J. & Tilly, J. L. Moderate caloric restriction initiated in rodents during adulthood sustains function of the female reproductive axis into advanced chronological age. Aging Cell 7, 622–629 (2008).
Solon-Biet, S. M. et al. Macronutrient balance, reproductive function, and lifespan in aging mice. Proc. Natl Acad. Sci. USA 112, 3481–3486 (2015).
Xiang, C. et al. Hippo signaling pathway reveals a spatio-temporal correlation with the size of primordial follicle pool in mice. Cell Physiol. Biochem. 35, 957–968 (2015).
Fu, X., Cheng, J., Hou, Y. & Zhu, S. The association between the oocyte pool and aneuploidy: a comparative study of the reproductive potential of young and aged mice. J. Assist. Reprod. Genet. 31, 323–331 (2014).
Duncan, F. E. et al. Age-associated dysregulation of protein metabolism in the mammalian oocyte. Aging Cell 16, 1381–1393 (2017).
Babayev, E. et al. Reproductive aging is associated with changes in oocyte mitochondrial dynamics, function, and mtDNA quantity. Maturitas 93, 121–130 (2016).
Babayev, E. et al. Cumulus expansion is impaired with advanced reproductive age due to loss of matrix integrity and reduced hyaluronan. Aging Cell 22, e14004 (2023).
Lim, J. & Luderer, U. Oxidative damage increases and antioxidant gene expression decreases with aging in the mouse ovary. Biol. Reprod. 84, 775–782 (2011).
Qian, Y. et al. Implication of differential peroxiredoxin 4 expression with age in ovaries of mouse and human for ovarian aging. Curr. Mol. Med. 16, 243–251 (2016).
Lin, N., Lin, J., Plosch, T., Sun, P. & Zhou, X. An oxidative stress-related gene signature in granulosa cells is associated with ovarian aging. Oxid. Med. Cell Longev. 2022, 1070968 (2022).
Alcazar-Fabra, M., Navas, P. & Brea-Calvo, G. Coenzyme Q biosynthesis and its role in the respiratory chain structure. Biochim. Biophys. Acta 1857, 1073–1078 (2016).
Ben-Meir, A. et al. Coenzyme Q10 restores oocyte mitochondrial function and fertility during reproductive aging. Aging Cell 14, 887–895 (2015).
Liu, C. et al. Granulosa cell mevalonate pathway abnormalities contribute to oocyte meiotic defects and aneuploidy. Nat. Aging 3, 670–687 (2023).
Amjad, S. et al. Role of NAD+ in regulating cellular and metabolic signaling pathways. Mol. Metab. 49, 101195 (2021).
Mara, J. N. et al. Ovulation and ovarian wound healing are impaired with advanced reproductive age. Aging 12, 9686–9713 (2020).
Umehara, T. et al. Female reproductive life span is extended by targeted removal of fibrotic collagen from the mouse ovary. Sci. Adv. 8, eabn4564 (2022).
Suebthawinkul, C., Babayev, E., Lee, H. C. & Duncan, F. E. Morphokinetic parameters of mouse oocyte meiotic maturation and cumulus expansion are not affected by reproductive age or ploidy status. J. Assist. Reprod. Gen. 40, 1197–1213 (2023).
Bajwa, P. et al. Age related increase in mTOR activity contributes to the pathological changes in ovarian surface epithelium. Oncotarget 7, 19214–19227 (2016).
Tan, O. L. & Fleming, J. S. Proliferating cell nuclear antigen immunoreactivity in the ovarian surface epithelium of mice of varying ages and total lifetime ovulation number following ovulation. Biol. Reprod. 71, 1501–1507 (2004).
Clow, O. L., Hurst, P. R. & Fleming, J. S. Changes in the mouse ovarian surface epithelium with age and ovulation number. Mol. Cell Endocrinol. 191, 105–111 (2002).
Fleming, J. S., McQuillan, H. J., Millier, M. J., Beaugie, C. R. & Livingstone, V. E-cadherin expression and bromodeoxyuridine incorporation during development of ovarian inclusion cysts in age-matched breeder and incessantly ovulated CD-1 mice. Reprod. Biol. Endocrinol. 5, 14 (2007).
Eppig, J. J. & O'Brien, M. In vitro maturation and fertilization of oocytes isolated from aged mice: a strategy to rescue valuable genetic resources. J. Assist. Reprod. Genet. 12, 269–273 (1995).
Lee, S. H. et al. Ovarian cysts in MRL/MpJ mice are derived from the extraovarian rete: a developmental study. J. Anat. 219, 743–755 (2011).
Long, G. G. Apparent mesonephric duct (rete anlage) origin for cysts and proliferative epithelial lesions in the mouse ovary. Toxicol. Pathol. 30, 592–598 (2002).
Kon, Y., Konno, A., Hashimoto, Y. & Endoh, D. Ovarian cysts in MRL/MpJ mice originate from rete ovarii. Anat. Histol. Embryol. 36, 172–178 (2007).
Tan, O. L., Hurst, P. R. & Fleming, J. S. Location of inclusion cysts in mouse ovaries in relation to age, pregnancy, and total ovulation number: implications for ovarian cancer. J. Pathol. 205, 483–490 (2005).
Kinnear, H. M. et al. The ovarian stroma as a new frontier. Reproduction 160, R25–R39 (2020).
Briley, S. M. et al. Reproductive age-associated fibrosis in the stroma of the mammalian ovary. Reproduction 152, 245–260 (2016).
Landry, D. A. et al. Metformin prevents age-associated ovarian fibrosis by modulating the immune landscape in female mice. Sci. Adv. 8, eabq1475 (2022).
Amargant, F. et al. Ovarian stiffness increases with age in the mammalian ovary and depends on collagen and hyaluronan matrices. Aging Cell 19, e13259 (2020).
Amargant, F., Magalhaes, C., Pritchard, M. T. & Duncan, F. E. Systemic low-dose anti-fibrotic treatment attenuates ovarian aging in the mouse. Geroscience https://doi.org/10.1007/s11357-024-01322-w (2024).
Dipali, S. S. et al. Proteomic quantification of native and ECM-enriched mouse ovaries reveals an age-dependent fibro-inflammatory signature. Aging 15, 10821–10855 (2023).
Lliberos, C. et al. Evaluation of inflammation and follicle depletion during ovarian ageing in mice. Sci. Rep. 11, 278 (2021).
Rowley, J. E. et al. Low molecular weight hyaluronan induces an inflammatory response in ovarian stromal cells and impairs gamete development in vitro. Int J. Mol. Sci. 21, 1036 (2020).
Ben Yaakov, T., Wasserman, T., Aknin, E. & Savir, Y. Single-cell analysis of the aged ovarian immune system reveals a shift towards adaptive immunity and attenuated cell function. Elife 12, e74915 (2023).
Asano, Y. Age-related accumulation of non-heme ferric and ferrous iron in mouse ovarian stroma visualized by sensitive non-heme iron histochemistry. J. Histochem. Cytochem. 60, 229–242 (2012).
Galaris, D., Barbouti, A. & Pantopoulos, K. Iron homeostasis and oxidative stress: an intimate relationship. Biochim. Biophys. Acta Mol. Cell Res. 1866, 118535 (2019).
Covarrubias, A. J., Perrone, R., Grozio, A. & Verdin, E. NAD+ metabolism and its roles in cellular processes during ageing. Nat. Rev. Mol. Cell Biol. 22, 119–141 (2021).
Yang, Q. et al. Deletion of enzymes for de novo NAD+ biosynthesis accelerated ovarian aging. Aging Cell 22, e13904 (2023).
Yang, Q. et al. Increasing ovarian NAD+ levels improve mitochondrial functions and reverse ovarian aging. Free Radic. Biol. Med. 156, 1–10 (2020).
Bertoldo, M. J. et al. NAD+ repletion rescues female fertility during reproductive aging. Cell Rep. 30, 1670–1681 (2020).
Perrone, R. et al. CD38 regulates ovarian function and fecundity via NAD+ metabolism. iScience 26, 107949 (2023).
Yang, Q. et al. NADase CD38 is a key determinant of ovarian aging. Nat. Aging 4, 110–128 (2024).
Maruyama, N. et al. Accumulation of senescent cells in the stroma of aged mouse ovary. J. Reprod. Dev. 69, 328–336 (2023).
Jia, L. et al. Analyzing the cellular and molecular atlas of ovarian mesenchymal cells provides a strategy against female reproductive aging. Sci. China Life Sci. 66, 2818–2836 (2023).
Deng, J. et al. miR-143-3p promotes ovarian granulosa cell senescence and inhibits estradiol synthesis by targeting UBE2E3 and LHCGR. Int. J. Mol. Sci. 24, 12560 (2023).
Henderson, S. A. & Edwards, R. G. Chiasma frequency and maternal age in mammals. Nature 218, 22–28 (1968).
Luthardt, F. W., Palmer, C. G. & Yu, P. L. Chiasma and univalent frequencies in aging female mice. Cytogenet. Cell Genet. 12, 68–79 (1973).
Speed, R. M. The effects of ageing on the meiotic chromosomes of male and female mice. Chromosoma 64, 241–254 (1977).
Brunet, S. & Maro, B. Germinal vesicle position and meiotic maturation in mouse oocyte. Reproduction 133, 1069–1072 (2007).
Manosalva, I. & Gonzalez, A. Aging changes the chromatin configuration and histone methylation of mouse oocytes at germinal vesicle stage. Theriogenology 74, 1539–1547 (2010).
Cui, L. B. et al. The Kunming mouse: as a model for age-related decline in female fertility in human. Zygote 21, 367–376 (2013).
Lister, L. M. et al. Age-related meiotic segregation errors in mammalian oocytes are preceded by depletion of cohesin and Sgo2. Curr. Biol. 20, 1511–1521 (2010).
Martin, R. H., Dill, F. J. & Miller, J. R. Nondisjunction in aging female mice. Cytogenet. Cell. Genet. 17, 150–160 (1976).
Duncan, F. E., Chiang, T., Schultz, R. M. & Lampson, M. A. Evidence that a defective spindle assembly checkpoint is not the primary cause of maternal age-associated aneuploidy in mouse eggs. Biol. Reprod. 81, 768–776 (2009).
Eichenlaub-Ritter, U. & Boll, I. Age-related non-disjunction, spindle formation and progression through maturation of mammalian oocytes. Prog. Clin. Biol. Res. 318, 259–269 (1989).
Koncicka, M. et al. Increased expression of maturation promoting factor components speeds up meiosis in oocytes from aged females. Int. J. Mol. Sci. 19, 2841 (2018).
Cheng, J. M. et al. Elevated intracellular pH appears in aged oocytes and causes oocyte aneuploidy associated with the loss of cohesion in mice. Cell Cycle 15, 2454–2463 (2016).
Sakakibara, Y. et al. Bivalent separation into univalents precedes age-related meiosis I errors in oocytes. Nat. Commun. 6, 7550 (2015).
Shomper, M., Lappa, C. & FitzHarris, G. Kinetochore microtubule establishment is defective in oocytes from aged mice. Cell Cycle 13, 1171–1179 (2014).
Yun, Y. et al. Reduced ability to recover from spindle disruption and loss of kinetochore spindle assembly checkpoint proteins in oocytes from aged mice. Cell Cycle 13, 1938–1947 (2014).
Mihajlovic, A. I., Haverfield, J. & FitzHarris, G. Distinct classes of lagging chromosome underpin age-related oocyte aneuploidy in mouse. Dev. Cell 56, 2273–2283 (2021).
Nakagawa, S. & FitzHarris, G. Intrinsically defective microtubule dynamics contribute to age-related chromosome segregation errors in mouse oocyte meiosis-I. Curr. Biol. 27, 1040–1047 (2017).
Yin, L., Mihajlovic, A. I., Yang, G. S. & FitzHarris, G. Kinetochore deterioration concommitant with centromere weakening during aging in mouse oocyte meiosis-I. FASEB J. 37, e22922 (2023).
Pan, H., Ma, P., Zhu, W. & Schultz, R. M. Age-associated increase in aneuploidy and changes in gene expression in mouse eggs. Dev. Biol. 316, 397–407 (2008).
Qiu, D. et al. Sirt2-BubR1 acetylation pathway mediates the effects of advanced maternal age on oocyte quality. Aging Cell 17, e12698 (2018).
Zhang, L. et al. Sirt2 functions in spindle organization and chromosome alignment in mouse oocyte meiosis. FASEB J. 28, 1435–1445 (2014).
Tian, N. et al. Three-dimensional quantitative analysis of chromosomes in the oocytes of aging mice during meiosis I in vitro. Theriogenology 79, 249–256 (2013).
Hornick, J. E. et al. Age-associated alterations in the micromechanical properties of chromosomes in the mammalian egg. J. Assist. Reprod. Genet. 32, 765–769 (2015).
Tachibana-Konwalski, K. et al. Rec8-containing cohesin maintains bivalents without turnover during the growing phase of mouse oocytes. Genes Dev. 24, 2505–2516 (2010).
Cheng, J. M. et al. Merotelic kinetochore attachment in oocyte meiosis II causes sister chromatids segregation errors in aged mice. Cell Cycle 16, 1404–1413 (2017).
Chiang, T., Duncan, F. E., Schindler, K., Schultz, R. M. & Lampson, M. A. Evidence that weakened centromere cohesion is a leading cause of age-related aneuploidy in oocytes. Curr. Biol. 20, 1522–1528 (2010).
Yun, Y., Lane, S. I. & Jones, K. T. Premature dyad separation in meiosis II is the major segregation error with maternal age in mouse oocytes. Development 141, 199–208 (2014).
Nabti, I., Grimes, R., Sarna, H., Marangos, P. & Carroll, J. Maternal age-dependent APC/C-mediated decrease in securin causes premature sister chromatid separation in meiosis II. Nat. Commun. 8, 15346 (2017).
Zielinska, A. P. et al. Meiotic kinetochores fragment into multiple lobes upon cohesin loss in aging eggs. Curr. Biol. 29, 3749–3765 (2019).
Kouznetsova, A., Liu, J. G., Valentiniene, S., Brismar, H. & Höög, C. Age-dependent aneuploidy in mammalian oocytes instigated at the second meiotic division. Aging Cell 21, e13649 (2022).
Mihajlovic, A. I., Byers, C., Reinholdt, L. & FitzHarris, G. Spindle assembly checkpoint insensitivity allows meiosis-II despite chromosomal defects in aged eggs. EMBO Rep. 24, e57227 (2023).
Chiang, T., Schultz, R. M. & Lampson, M. A. Age-dependent susceptibility of chromosome cohesion to premature separase activation in mouse oocytes. Biol. Reprod. 85, 1279–1283 (2011).
Sun, Q. et al. Shugoshin regulates cohesin, kinetochore-microtubule attachments, and chromosomal instability. Cytogenet. Genome Res. 162, 283–296 (2022).
Liu, Y. et al. Low estrogen level in aged mice leads to abnormal oogenesis affecting the quality of surrounded nucleolus-type immature oocytes. Reprod. Fert. Dev. 34, 991–1001 (2022).
Perez, G. I. & Tilly, J. L. Cumulus cells are required for the increased apoptotic potential in oocytes of aged mice. Hum. Reprod. 12, 2781–2783 (1997).
Fujino, Y. et al. DNA fragmentation of oocytes in aged mice. Hum. Reprod. 11, 1480–1483 (1996).
Titus, S. et al. Impairment of BRCA1-related DNA double-strand break repair leads to ovarian aging in mice and humans. Sci. Transl. Med. 5, 172ra121 (2013).
Horta, F., Ravichandran, A., Catt, S., Vollenhoven, B. & Temple-Smith, P. Ageing and ovarian stimulation modulate the relative levels of transcript abundance of oocyte DNA repair genes during the germinal vesicle-metaphase II transition in mice. J. Assist. Reprod. Genet. 38, 55–69 (2021).
Zhang, M., Lu, Y., Chen, Y., Zhang, Y. & Xiong, B. Insufficiency of melatonin in follicular fluid is a reversible cause for advanced maternal age-related aneuploidy in oocytes. Redox Biol. 28, 101327 (2020).
Marangos, P. et al. DNA damage-induced metaphase I arrest is mediated by the spindle assembly checkpoint and maternal age. Nat. Commun. 6, 8706 (2015).
Guli, C. L. & Smyth, D. R. Lack of effect of maternal age on UV-induced DNA repair in mouse oocytes. Mutat. Res. 210, 323–328 (1989).
Wasserzug-Pash, P. et al. Loss of heterochromatin and retrotransposon silencing as determinants in oocyte aging. Aging Cell 21, e13568 (2022).
Yamada-Fukunaga, T. et al. Age-associated telomere shortening in mouse oocytes. Reprod. Biol. Endocrinol. 11, 108 (2013).
Ge, J. et al. SIRT6 participates in the quality control of aged oocytes via modulating telomere function. Aging 11, 1965–1976 (2019).
Ramos-Ibeas, P. et al. Longitudinal analysis of somatic and germ-cell telomere dynamics in outbred mice. Mol. Reprod. Dev. 86, 1033–1043 (2019).
Hamatani, T. et al. Age-associated alteration of gene expression patterns in mouse oocytes. Hum. Mol. Genet. 13, 2263–2278 (2004).
Yue, M. X. et al. Abnormal DNA methylation in oocytes could be associated with a decrease in reproductive potential in old mice. J. Assist. Reprod. Genet. 29, 643–650 (2012).
Shao, G. B. et al. Aging alters histone H3 lysine 4 methylation in mouse germinal vesicle stage oocytes. Reprod. Fertil. Dev. 27, 419–426 (2015).
Zhang, L., Lu, D. Y., Ma, W. Y. & Li, Y. Age-related changes in the localization of DNA methyltransferases during meiotic maturation in mouse oocytes. Fertil. Steril. 95, 1531–1534 (2011).
Castillo-Fernandez, J. et al. Increased transcriptome variation and localised DNA methylation changes in oocytes from aged mice revealed by parallel single-cell analysis. Aging Cell 19, e13278 (2020).
Paczkowski, M., Schoolcraft, W. B. & Krisher, R. L. Dysregulation of methylation and expression of imprinted genes in oocytes and reproductive tissues in mice of advanced maternal age. J. Assist. Reprod. Genet. 32, 713–723 (2015).
Wu, Y. W. et al. Dynamic mRNA degradome analyses indicate a role of histone H3K4 trimethylation in association with meiosis-coupled mRNA decay in oocyte aging. Nat. Commun. 13, 3191 (2022).
He, Y. F. et al. Melatonin ameliorates histone modification disorders in mammalian aged oocytes by neutralizing the alkylation of HDAC1. Free Radic. Biol. Med. 208, 361–370 (2023).
Potabattula, R. et al. Ribosomal DNA methylation in human and mouse oocytes increases with age. Aging 14, 1214–1232 (2022).
Akiyama, T., Nagata, M. & Aoki, F. Inadequate histone deacetylation during oocyte meiosis causes aneuploidy and embryo death in mice. Proc. Natl Acad. Sci. USA 103, 7339–7344 (2006).
Manosalva, I. & Gonzalez, A. Aging alters histone H4 acetylation and CDC2A in mouse germinal vesicle stage oocytes. Biol. Reprod. 81, 1164–1171 (2009).
Suo, L. et al. Changes in acetylation on lysine 12 of histone H4 (acH4K12) of murine oocytes during maternal aging may affect fertilization and subsequent embryo development. Fertil. Steril. 93, 945–951 (2010).
Huang, J. A. et al. Multi-omics analysis reveals translational landscapes and regulations in mouse and human oocyte aging. Adv. Sci. 10, e2301538 (2023).
Mihalas, B. P. et al. Oxidative damage in naturally aged mouse oocytes is exacerbated by dysregulation of proteasomal activity. J. Biol. Chem. 293, 18944–18964 (2018).
Harasimov, K. et al. The maintenance of oocytes in the mammalian ovary involves extreme protein longevity. Nat. Cell Biol. 26, 1124–1138 (2024).
Bomba-Warczak, E. K. et al. Exceptional longevity of mammalian ovarian and oocyte macromolecules throughout the reproductive lifespan. eLife 13, RP93172 (2024).
Peters, A. E. et al. The impact of aging on macroautophagy in the pre-ovulatory mouse oocyte. Front. Cell Dev. Biol. 9, 691826 (2021).
Mishina, T. et al. Single-oocyte transcriptome analysis reveals aging-associated effects influenced by life stage and calorie restriction. Aging Cell 20, e13428 (2021).
Del Llano, E. et al. Age-related differences in the translational landscape of mammalian oocytes. Aging Cell 19, e13231 (2020).
Kim, A. M., Vogt, S., O'Halloran, T. V. & Woodruff, T. K. Zinc availability regulates exit from meiosis in maturing mammalian oocytes. Nat. Chem. Biol. 6, 674–681 (2010).
Tian, X. & Diaz, F. J. Zinc depletion causes multiple defects in ovarian function during the periovulatory period in mice. Endocrinology 153, 873–886 (2012).
Tian, X. & Diaz, F. J. Acute dietary zinc deficiency before conception compromises oocyte epigenetic programming and disrupts embryonic development. Dev. Biol. 376, 51–61 (2013).
Tian, X., Anthony, K., Neuberger, T. & Diaz, F. J. Preconception zinc deficiency disrupts postimplantation fetal and placental development in mice. Biol. Reprod. 90, 83 (2014).
Lisle, R. S., Anthony, K., Randall, M. A. & Diaz, F. J. Oocyte-cumulus cell interactions regulate free intracellular zinc in mouse oocytes. Reproduction 145, 381–390 (2013).
Kujjo, L. L. et al. Ceramide and its transport protein (CERT) contribute to deterioration of mitochondrial structure and function in aging oocytes. Mech. Ageing Dev. 134, 43–52 (2013).
Kushnir, V. A. et al. Reproductive aging is associated with decreased mitochondrial abundance and altered structure in murine oocytes. J. Assist. Reprod. Genet. 29, 637–642 (2012).
Kim, M. J. et al. Association between functional activity of mitochondria and actin cytoskeleton instability in oocytes from advanced age mice. Reprod. Sci. 27, 1037–1046 (2020).
Al-Zubaidi, U. et al. Mitochondria-targeted therapeutics, MitoQ and BGP-15, reverse aging-associated meiotic spindle defects in mouse and human oocytes. Hum. Reprod. 36, 771–784 (2021).
Czajkowska, K. & Ajduk, A. Mitochondrial activity and redox status in oocytes from old mice: the interplay between maternal and postovulatory aging. Theriogenology 204, 18–30 (2023).
Simsek-Duran, F. et al. Age-associated metabolic and morphologic changes in mitochondria of individual mouse and hamster oocytes. PLoS ONE 8, e64955 (2013).
Khan, S. A., Reed, L., Schoolcraft, W. B., Yuan, Y. & Krisher, R. L. Control of mitochondrial integrity influences oocyte quality during reproductive aging. Mol. Hum. Reprod. 29, gaad028b (2023).
Tatone, C. et al. Evidence that carbonyl stress by methylglyoxal exposure induces DNA damage and spindle aberrations, affects mitochondrial integrity in mammalian oocytes and contributes to oocyte ageing. Hum. Reprod. 26, 1843–1859 (2011).
Ma, H. et al. Germline and somatic mtDNA mutations in mouse aging. PLoS ONE 13, e0201304 (2018).
Pasquariello, R. et al. Alterations in oocyte mitochondrial number and function are related to spindle defects and occur with maternal aging in mice and humans. Biol. Reprod. 100, 971–981 (2019).
Hartley, F. et al. No evidence for age-related differences in mitochondrial RNA quality in the female germline. Reprod. Fertil. 3, 198–206 (2022).
Mok, H. J. et al. Age-associated lipidome changes in metaphase II mouse oocytes. PLoS ONE 11, e0148577 (2016).
Bogliolo, L. et al. Raman spectroscopy-based approach to detect aging-related oxidative damage in the mouse oocyte. J. Assist. Reprod. Genet. 30, 877–882 (2013).
Mihalas, B. P., De Iuliis, G. N., Redgrove, K. A., McLaughlin, E. A. & Nixon, B. The lipid peroxidation product 4-hydroxynonenal contributes to oxidative stress-mediated deterioration of the ageing oocyte. Sci. Rep. 7, 6247 (2017).
Goud, P. T. et al. Chronological age enhances aging phenomena and protein nitration in oocyte. Front. Endocrinol. 14, 1251102 (2023).
Park, M. J. et al. Prediction of ovarian aging using ovarian expression of BMP15, GDF9, and C-KIT. Exp. Biol. Med. 245, 711–719 (2020).
Tappa, B., Amao, H., Saito, T. R., Sugiyama, M. & Takahashi, K. W. In vitro development of two-cell embryos obtained from female mice of different ages. Jikken Dobutsu 42, 623–626 (1993).
Tarin, J. J., Gomez-Piquer, V., Pertusa, J. F., Hermenegildo, C. & Cano, A. Association of female aging with decreased parthenogenetic activation, raised MPF, and MAPKs activities and reduced levels of glutathione S-transferases activity and thiols in mouse oocytes. Mol. Reprod. Dev. 69, 402–410 (2004).
Crocker, K., Calder, M. D., Edwards, N. A., Betts, D. H. & Watson, A. J. CD-1 mouse fertility rapidly declines and is accompanied with early pregnancy loss under conventional housing conditions. Theriogenology 108, 245–254 (2018).
Jurisicova, A., Rogers, I., Fasciani, A., Casper, R. F. & Varmuza, S. Effect of maternal age and conditions of fertilization on programmed cell death during murine preimplantation embryo development. Mol. Hum. Reprod. 4, 139–145 (1998).
Tsunoda, Y. & Chang, M. C. The suppressive effect of sera from old female mice on in vitro fertilization and blastocyst development. Biol. Reprod. 20, 355–361 (1979).
Saunders, C. M. et al. PLC zeta: a sperm-specific trigger of Ca2+ oscillations in eggs and embryo. Development 129, 3533–3544 (2002).
Nozawa, K., Satouh, Y., Fujimoto, T., Oji, A. & Ikawa, M. Sperm-borne phospholipase C zeta-1 ensures monospermic fertilization in mice. Sci. Rep. 8, 1315 (2018).
McDonough, C. E., Bernhardt, M. L. & Williams, C. J. Mouse strain-dependent egg factors regulate calcium signals at fertilization. Mol. Reprod. Dev. 87, 284–292 (2020).
Czajkowska, K., Walewska, A., Ishikawa, T., Szczepanska, K. & Ajduk, A. Age-related alterations in fertilization-induced Ca2+ oscillations depend on the genetic background of mouse oocytes. Biol. Reprod. 103, 986–999 (2020).
Haverfield, J. et al. Ca2+ dynamics in oocytes from naturally-aged mice. Sci. Rep. 6, 19357 (2016).
Huang, J. et al. Generation of pluripotent stem cells from eggs of aging mice. Aging Cell 9, 113–125 (2010).
Carbone, M. C. & Tatone, C. Alterations in the protein kinase C signaling activated by a parthenogenetic agent in oocytes from reproductively old mice. Mol. Reprod. Dev. 76, 122–131 (2009).
Verlhac, M. H., Kubiak, J. Z., Clarke, H. J. & Maro, B. Microtubule and chromatin behavior follow MAP kinase activity but not MPF activity during meiosis in mouse oocytes. Development 120, 1017–1025 (1994).
Yan, J. et al. Cryo-survival, fertilization and early embryonic development of vitrified oocytes derived from mice of different reproductive age. J. Assist. Reprod. Genet. 27, 605–611 (2010).
Horta, F., Catt, S., Ramachandran, P., Vollenhoven, B. & Temple-Smith, P. Female ageing affects the DNA repair capacity of oocytes in IVF using a controlled model of sperm DNA damage in mice. Hum. Reprod. 35, 529–544 (2020).
Santalo, J. et al. The genetic risks of in vitro fertilization techniques: the use of an animal model. J. Assist. Reprod. Genet. 9, 462–474 (1992).
Jiao, Z. X., Xu, M. & Woodruff, T. K. Age-associated alteration of oocyte-specific gene expression in polar bodies: potential markers of oocyte competence. Fertil. Steril. 98, 480–486 (2012).
Kawagoe, Y. et al. CXCL5-CXCR2 signaling is a senescence-associated secretory phenotype in preimplantation embryos. Aging Cell 19, e13240 (2020).
May-Panloup, P. et al. Maternal ageing impairs mitochondrial DNA kinetics during early embryogenesis in mice. Hum. Reprod. 34, 1313–1324 (2019).
Tao, X. et al. Mitochondrial DNA content is associated with ploidy status, maternal age, and oocyte maturation methods in mouse blastocysts. J. Assist. Reprod. Genet. 34, 1587–1594 (2017).
Gu, Y. M., Xu, J. J., Sun, F. & Cheng, J. M. Elevated intracellular pH of zygotes during mouse aging causes mitochondrial dysfunction associated with poor embryo development. Mol. Cell. Endocrinol. 574, 111991 (2023).
Almohammed, Z. N. H. et al. The effect of melatonin on mitochondrial function and autophagy in in vitro matured oocytes of aged mice. Cell J. 22, 9–16 (2020).
Iljas, J. D., Wei, Z. & Homer, H. A. Sirt1 sustains female fertility by slowing age-related decline in oocyte quality required for post-fertilization embryo development. Aging Cell 19, e13204 (2020).
Tarin, J. J. et al. Long-term effects of delayed motherhood in mice on postnatal development and behavioural traits of offspring. Hum. Reprod. 18, 1580–1587 (2003).
Lopes, F. L. et al. Reproductive and epigenetic outcomes associated with aging mouse oocytes. Hum. Mol. Genet. 18, 2032–2044 (2009).
Holinka, C. F. & Finch, C. E. Efficiency of mating in C57BL/6J female mice as a function of age and previous parity. Exp. Gerontol. 16, 393–398 (1981).
Wirth-Dzieciolowska, E. & Czuminska, K. Longevity and aging of mice from lines divergently selected for body weight for over 90 generations. Biogerontology 1, 171–178 (2000).
Patel, R. et al. Effect of reproductive ageing on pregnant mouse uterus and cervix. J. Physiol. 595, 2065–2084 (2017).
Holinka, C. F., Tseng, Y. C. & Finch, C. E. Prolonged gestation, elevated preparturitional plasma progesterone and reproductive aging in C57BL/6J mice. Biol. Reprod. 19, 807–816 (1978).
Gosden, R. G. & Fowler, R. E. Corpus luteum function in ageing inbred mice. Experientia 35, 128–130 (1979).
Fabricant, J. D., Dunn, G. & Schneider, E. L. Maternal age related pre- and post-implantation fetal mortality: a strain survey. Mech. Ageing Dev. 8, 227–231 (1978).
Harman, S. M. & Talbert, G. B. The effect of maternal age on ovulation, corpora lutea of pregnancy, and implantation failure in mice. J. Reprod. Fertil. 23, 33–39 (1970).
Khalyfa, A. et al. Transcription profiling of estrogen target genes in young and old mouse uterus. Exp. Gerontol. 38, 1087–1099 (2003).
Jiang, Y. et al. Decreased expression of elastin and lysyl oxidase family genes in urogenital tissues of aging mice. J. Obstet. Gynaecol. Res. 40, 1998–2004 (2014).
Finch, C. E. & Holinka, C. F. Aging and uterine growth during implantation in C57BL/6J mice. Exp. Gerontol. 17, 235–241 (1982).
van der Heijden, O. W. et al. Aging blunts remodeling of the uterine artery during murine pregnancy. J. Soc. Gynecol. Investig. 11, 304–310 (2004).
Miller, M. M., Teng, C. J., Mitmaker, B. & Wang, E. Characterization of the tissue regression process in the uterus of older mice as apoptotic by the presence of Tp30, an isoform of terminin. Eur. J. Histochem. 39, 91–100 (1995).
Barnett, S. A., Munro, K. M. H. & Stoddart, R. C. Growth and pathology of aged house mice. Exp. Gerontol. 9, 275–279 (1974).
Malinin, G. I. & Malinin, I. M. Age-related spontaneous uterine lesions in mice. J. Gerontol. 27, 193–196 (1972).
Silva, E. et al. Antioxidant supplementation modulates age-related placental bed morphology and reproductive outcome in mice. Biol. Reprod. 93, 56 (2015).
Shimizu, K. & Yamada, J. Relationship of decrease in fecundity with advancing age to structural changes in mouse endometrium. J. Anat. 196, 111–114 (2000).
Belisle, S., Beaudry, C. & Lehoux, J. G. Endocrine aging in CBA mice: characterization of uterine cytosolic and nuclear sex steroid receptors. Exp. Gerontol. 17, 417–423 (1982).
Belisle, S., Bellabarba, D., Lehoux, J. G., Robel, P. & Baulieu, E. E. Effect of aging on the dissociation kinetics and estradiol receptor nuclear interactions in mouse uteri: correlation with biological effects. Endocrinology 118, 750–758 (1986).
Belisle, S., Bellabarba, D. & Lehoux, J. G. Estradiol receptor-nuclear interactions in aging mouse uteri: the role of DNA and nuclear matrix. J. Steroid Biochem. 32, 41–43 (1989).
Combatsiaris, E. P. & Nelson, J. F. Dissociation kinetics of the uterine estrogen receptor-estradiol complex are unaltered in aging C57BL/6 mice. Mech. Ageing Dev. 65, 137–147 (1992).
Holinka, C. F., Tseng, Y. C. & Finch, C. E. Reproductive aging in C57BL/6J mice: plasma progesterone, viable embryos and resorption frequency throughout pregnancy. Biol. Reprod. 20, 1201–1211 (1979).
Woods, L. et al. Decidualisation and placentation defects are a major cause of age-related reproductive decline. Nat. Commun. 8, 352 (2017).
Talbert, G. B. Effect of maternal age on postimplantation reproductive failure in mice. J. Reprod. Fertil. 24, 449–452 (1971).
Wang, H. & Dey, S. K. Roadmap to embryo implantation: clues from mouse models. Nat. Rev. Genet. 7, 185–199 (2006).
Kokorudz, C., Radford, B. N., Dean, W. & Hemberger, M. Advanced maternal age differentially affects embryonic tissues with the most severe impact on the developing brain. Cells 12, 76 (2022).
Woods, L. et al. Epigenetic changes occur at decidualisation genes as a function of reproductive ageing in mice. Development 147, dev185629 (2020).
Barry, C. M. et al. Morphological and neurochemical differences in peptidergic nerve fibers of the mouse vagina. J. Comp. Neurol. 525, 2394–2410 (2017).
Kokorudz, C., Radford, B. N., Dean, W. & Hemberger, M. Advanced maternal age differentially affects embryonic tissues with the most severe impact on the developing brain. Cells 12, 76 (2023).
Tarin, J. J. et al. Delayed motherhood decreases life expectancy of mouse offspring. Biol. Reprod. 72, 1336–1343 (2005).
Morello, G. M. et al. High laboratory mouse pre-weaning mortality associated with litter overlap, advanced dam age, small and large litters. PLoS ONE 15, e0236290 (2020).
Weber, E. M., Hultgren, J., Algers, B. & Olsson, I. A. Do laboratory mouse females that lose their litters behave differently around parturition? PLoS ONE 11, e0161238 (2016).
Brajon, S. et al. All the pups we cannot see: cannibalism masks perinatal death in laboratory mouse breeding but infanticide is rare. Animals 11, 2327 (2021).
López-Otín, C., Blasco, M. A., Partridge, L., Serrano, M. & Kroemer, G. Hallmarks of aging: an expanding universe. Cell 186, 243–278 (2023).
Saraf, V. S. et al. Vaginal microbiome: normalcy vs dysbiosis. Arch. Microbiol 203, 3793–3802 (2021).
Bull, J. R. et al. Real-world menstrual cycle characteristics of more than 600,000 menstrual cycles. NPJ Digit. Med. 2, 83 (2019).
Byers, S. L., Wiles, M. V., Dunn, S. L. & Taft, R. A. Mouse estrous cycle identification tool and images. PLoS ONE 7, e35538 (2012).
Maybin, J. A. & Critchley, H. O. Menstrual physiology: implications for endometrial pathology and beyond. Hum. Reprod. Update 21, 748–761 (2015).
Catalini, L. & Fedder, J. Characteristics of the endometrium in menstruating species: lessons learned from the animal kingdom. Biol. Reprod. 102, 1160–1169 (2020).
Wesselink, A. K. et al. Age and fecundability in a North American preconception cohort study. Am. J. Obstet. Gynecol. 217, 667.e1–667.e8 (2017).
Lyndaker, C. & Hulton, L. The influence of age on symptoms of perimenopause. J. Obstet. Gynecol. Neonatal Nurs. 33, 340–347 (2004).
Santoro, N. Perimenopause: from research to practice. J. Women’s Health 25, 332–339 (2016).
Cohen, A. A. Female post-reproductive lifespan: a general mammalian trait. Biol. Rev. Camb. Philos. Soc. 79, 733–750 (2004).
Brust, V., Schindler, P. M. & Lewejohann, L. Lifetime development of behavioural phenotype in the house mouse (Mus musculus). Front Zool. 12, S17 (2015).
Kochanek, K. D., Murphy, S. L., Xu, J. Q. & Arias, E. Mortality in the United States, 2022. NCHS Data Brief no. 492 https://doi.org/10.15620/cdc:13585 (2024).
Yuan, R. et al. Aging in inbred strains of mice: study design and interim report on median lifespans and circulating IGF1 levels. Aging Cell 8, 277–287 (2009).
Garcia, D. N. et al. Effect of caloric restriction and rapamycin on ovarian aging in mice. Geroscience 41, 395–408 (2019).
Liu, W. J. et al. Calorie restriction inhibits ovarian follicle development and follicle loss through activating SIRT1 signaling in mice. Eur. J. Med. Res. 20, 22 (2015).
Selesniemi, K., Lee, H. J., Muhlhauser, A. & Tilly, J. L. Prevention of maternal aging-associated oocyte aneuploidy and meiotic spindle defects in mice by dietary and genetic strategies. Proc. Natl Acad. Sci. USA 108, 12319–12324 (2011).
Kushnir, V. A., Seifer, D. B., Barad, D. H., Sen, A. & Gleicher, N. Potential therapeutic applications of human anti-Mullerian hormone (AMH) analogues in reproductive medicine. J. Assist. Reprod. Genet. 34, 1105–1113 (2017).
Yan, F. et al. The role of oxidative stress in ovarian aging: a review. J. Ovarian Res 15, 100 (2022).
Secomandi, L., Borghesan, M., Velarde, M. & Demaria, M. The role of cellular senescence in female reproductive aging and the potential for senotherapeutic interventions. Hum. Reprod. Update 28, 172–189 (2022).
Tokano, Y. et al. [A case of the hepatoma associated with hypergastrinemia]. Nihon Naika Gakkai Zasshi 78, 1490–1491 (1989).
Okamoto, N., Sato, Y., Kawagoe, Y., Shimizu, T. & Kawamura, K. Short-term resveratrol treatment restored the quality of oocytes in aging mice. Aging 14, 5628–5640 (2022).
McCloskey, C. W. et al. Metformin abrogates age-associated ovarian fibrosis. Clin. Cancer Res. 26, 632–642 (2020).
Kang, M. H. et al. Mitigating age-related ovarian dysfunction with the anti-inflammatory agent MIT-001. Int. J. Mol. Sci. 24, 15158 (2023).
Punzon-Jimenez, P. et al. Effect of aging on the human myometrium at single-cell resolution. Nat. Commun. 15, 945 (2024).
Ouni, E. et al. Proteome-wide and matrisome-specific atlas of the human ovary computes fertility biomarker candidates and open the way for precision oncofertility. Matrix Biol. 109, 91–120 (2022).
Ouni, E. et al. A blueprint of the topology and mechanics of the human ovary for next-generation bioengineering and diagnosis. Nat. Commun. 12, 5603 (2021).
Woessner, J. F. Jr. Age-related changes of the human uterus and its connective tissue framework. J. Gerontol. 18, 220–226 (1963).
van den Berg, I. M. et al. Defective deacetylation of histone 4 K12 in human oocytes is associated with advanced maternal age and chromosome misalignment. Hum. Reprod. 26, 1181–1190 (2011).
Uysal, F., Kosebent, E. G., Toru, H. S. & Ozturk, S. Decreased expression of TERT and telomeric proteins as human ovaries age may cause telomere shortening. J. Assist. Reprod. Genet. 38, 429–441 (2021).
Winkler, I. et al. The cycling and aging mouse female reproductive tract at single-cell resolution. Cell 187, 981–998 (2024).
Rodriguez-Garcia, M., Barr, F. D., Crist, S. G., Fahey, J. V. & Wira, C. R. Phenotype and susceptibility to HIV infection of CD4+ Th17 cells in the human female reproductive tract. Mucosal Immunol. 7, 1375–1385 (2014).
Yeaman, G. R. et al. Unique CD8+ T cell-rich lymphoid aggregates in human uterine endometrium. J. Leukoc. Biol. 61, 427–435 (1997).
Machlin, J. H. et al. Fibroinflammatory signatures increase with age in the human ovary and follicular fluid. Int. J. Mol. Sci. 22, 4902 (2021).
Baker, T. G. A quantitative and cytological study of germ cells in human ovaries. Proc. R. Soc. Lond. B Biol. Sci. 158, 417–433 (1963).
Kai, Y. et al. Transcriptomic signatures in trophectoderm and inner cell mass of human blastocysts classified according to developmental potential, maternal age and morphology. PLoS ONE 17, e0278663 (2022).
Ntostis, P. et al. The effects of aging on molecular modulators of human embryo implantation. iScience 24, 102751 (2021).
Strieby, A., McCallie, B. R., Parks, J. C., Schoolcraft, W. B. & Katz-Jaffe, M. G. Blastocysts derived from women of advanced maternal age have compromised gene expression profiles that may impact their implantation potential but do not influence the endometrial transcriptome. Fertil. Steril. https://doi.org/10.1016/j.fertnstert.2013.07.1641 (2013).
Menon, R. et al. Histological evidence of oxidative stress and premature senescence in preterm premature rupture of the human fetal membranes recapitulated in vitro. Am. J. Pathol. 184, 1740–1751 (2014).
Wu, M. et al. Spatiotemporal transcriptomic changes of human ovarian aging and the regulatory role of FOXP1. Nat. Aging 4, 527–545 (2024).
He, H. et al. Selective autophagic degradation of ACLY (ATP citrate lyase) maintains citrate homeostasis and promotes oocyte maturation. Autophagy 19, 163–179 (2023).
Vilser, C. et al. The variable expression of lectin-like oxidized low-density lipoprotein receptor (LOX-1) and signs of autophagy and apoptosis in freshly harvested human granulosa cells depend on gonadotropin dose, age, and body weight. Fertil. Steril. 93, 2706–2715 (2010).
Zhou, S., Zhao, L., Yi, T., Wei, Y. & Zhao, X. Author correction: menopause-induced uterine epithelium atrophy results from arachidonic acid/prostaglandin E2 axis inhibition-mediated autophagic cell death. Sci. Rep. 10, 6915 (2020).
Fujii, S. & Oguchi, T. Age- and endometrial microbiota-related delay in development of endometrial receptivity. Reprod. Med. Biol. 22, e12523 (2023).
So, K. A. et al. Menopausal status induces vaginal dysbiosis in women with human papillomavirus infection. Sci. Rep. 14, 7092 (2024).
Yoshikata, R. et al. Age-related changes, influencing factors, and crosstalk between vaginal and gut microbiota: a cross-sectional comparative study of pre- and postmenopausal women. J. Womens Health 31, 1763–1772 (2022).
Tatone, C. et al. Age-dependent changes in the expression of superoxide dismutases and catalase are associated with ultrastructural modifications in human granulosa cells. Mol. Hum. Reprod. 12, 655–660 (2006).
Burger, H. G. et al. Serum inhibins A and B fall differentially as FSH rises in perimenopausal women. Clin. Endocrinol. 48, 809–813 (1998).
Reame, N. E. et al. Age effects of follicle-stimulating hormone and pulsatile luteinizing hormone secretion across the menstrual cycle of premenopausal women. J. Clin. Endocrinol. Metab. 81, 1512–1518 (1996).
Overlie, I. et al. Inhibin A and B as markers of menopause: a five-year prospective longitudinal study of hormonal changes during the menopausal transition. Acta Obstet. Gynecol. Scand. 84, 281–285 (2005).
Lie Fong, S. et al. Serum anti-mullerian hormone levels in healthy females: a nomogram ranging from infancy to adulthood. J. Clin. Endocrinol. Metab. 97, 4650–4655 (2012).
Miro, F. et al. Sequential classification of endocrine stages during reproductive aging in women: the FREEDOM study. Menopause 12, 281–290 (2005).
MacNaughton, J., Banah, M., McCloud, P., Hee, J. & Burger, H. Age related changes in follicle stimulating hormone, luteinizing hormone, oestradiol and immunoreactive inhibin in women of reproductive age. Clin. Endocrinol. 36, 339–345 (1992).
Fogle, R. H., Li, A. & Paulson, R. J. Modulation of HOXA10 and other markers of endometrial receptivity by age and human chorionic gonadotropin in an endometrial explant model. Fertil. Steril. 93, 1255–1259 (2010).
Santoro, N. et al. Impaired folliculogenesis and ovulation in older reproductive aged women. J. Clin. Endocrinol. Metab. 88, 5502–5509 (2003).
Mihalas, B. P. et al. Age-dependent loss of cohesion protection in human oocytes. Curr. Biol. 34, 117–131 (2024).
Ito, M. et al. Age-associated changes in the subcellular localization of phosphorylated p38 MAPK in human granulosa cells. Mol. Hum. Reprod. 16, 928–937 (2010).
Robinson, W. P. et al. Maternal meiosis I non-disjunction of chromosome 15: dependence of the maternal age effect on level of recombination. Hum. Mol. Genet. 7, 1011–1019 (1998).
Zielinska, A. P., Holubcova, Z., Blayney, M., Elder, K. & Schuh, M. Sister kinetochore splitting and precocious disintegration of bivalents could explain the maternal age effect. Elife 4, e11389 (2015).
Duncan, F. E. et al. Chromosome cohesion decreases in human eggs with advanced maternal age. Aging Cell 11, 1121–1124 (2012).
Battaglia, D. E., Goodwin, P., Klein, N. A. & Soules, M. R. Influence of maternal age on meiotic spindle assembly in oocytes from naturally cycling women. Hum. Reprod. 11, 2217–2222 (1996).
Volarcik, K. et al. The meiotic competence of in-vitro matured human oocytes is influenced by donor age: evidence that folliculogenesis is compromised in the reproductively aged ovary. Hum. Reprod. 13, 154–160 (1998).
Ezoe, K. et al. Maternal age affects pronuclear and chromatin dynamics, morula compaction and cell polarity, and blastulation of human embryos. Hum. Reprod. 38, 387–399 (2023).
Smits, M. A. J. et al. Human ovarian aging is characterized by oxidative damage and mitochondrial dysfunction. Hum. Reprod. 38, 2208–2220 (2023).
Yang, L. et al. Mitochondrial DNA mutation exacerbates female reproductive aging via impairment of the NADH/NAD+ redox. Aging Cell 19, e13206 (2020).
Acknowledgements
We thank M. Beestrum at Northwestern University for assistance with the literature search and scoping review preparation. We acknowledge S. Tobin for critical input. This work was supported in part by National Institutes of Health grants (R01HD105752 to F.E.D., R01HD103384, AG029531 and AG056046 to T.R.K.), The Makowski Family Endowment (to T.R.K.), The Thomas J. Watkins Memorial Endowment (to F.E.D.) and the Global Consortium on Reproductive Longevity and Equity (T.R.K. and F.E.D.).
Author information
Authors and Affiliations
Contributions
J.L.B., S.S.D. and K.V. reviewed the literature, wrote the manuscript and prepared the figures. T.R.K. contributed to the conception of the review and revising and approving the manuscript for final submission. F.E.D. was responsible for conceiving, drafting and revising the manuscript and handling correspondence.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Aging thanks Qingling Yang, Lindsay Wu, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Text, Tables 1–6, References and Figs. 1–4.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Balough, J.L., Dipali, S.S., Velez, K. et al. Hallmarks of female reproductive aging in physiologic aging mice. Nat Aging 4, 1711–1730 (2024). https://doi.org/10.1038/s43587-024-00769-y
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s43587-024-00769-y
This article is cited by
-
Omega-3 dietary supplementation combined with exercise to keep telomere integrity in the liver of aged obese female mice
Journal of Physiology and Biochemistry (2026)
-
Novel multi-omic biomarkers to combat oocyte and ovarian aging
GeroScience (2026)
-
Research on the mechanism of human umbilical cord mesenchymal stem cells and their extracellular vesicles in the treatment of common reproductive diseases
Stem Cell Research & Therapy (2025)
-
Ovarian vascular aging: a hidden driver of mid-age female fertility decline
npj Aging (2025)
-
The Nexus of Iron, Senescence, and Fibrosis in Endometriosis: A Narrative Review
Reproductive Sciences (2025)