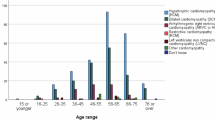
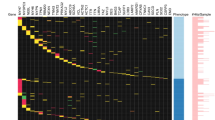
Abstract
The inherited cardiomyopathies exhibit a broad spectrum of disease, with some patients remaining asymptomatic throughout life, while, for others, the first symptom of disease is sudden cardiac death at a young age. The risk of malignant ventricular arrhythmia in these conditions has traditionally been linked to the degree of structural myocardial abnormalities and functional impairment. However, recent advances in genetic testing and knowledge of the genetic basis of the diseases have led to the identification of concealed cardiomyopathy, in which sudden cardiac arrest or sudden cardiac death occurs in the absence of observable clinical features of cardiomyopathy, with a diagnosis being made only after the identification of a causative genetic variant. Increased awareness of concealed cardiomyopathy, a better understanding of mechanisms of arrhythmia and identification of risk modulators will be vital to improve care for families with concealed cardiomyopathy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Ommen, S. R. & Semsarian, C. Hypertrophic cardiomyopathy: a practical approach to guideline directed management. Lancet 398, 2102–2108 (2021).
Arbelo, E. et al. 2023 ESC Guidelines for the management of cardiomyopathies. Eur. Heart J. 44, 3503–3626 (2023).
Basso, C. et al. Guidelines for autopsy investigation of sudden cardiac death: 2017 update from the Association for European Cardiovascular Pathology. Virchows Arch. 471, 691–705 (2017).
Ingles, J. et al. Evaluating the clinical validity of hypertrophic cardiomyopathy genes. Circ. Genom. Precis. Med. 12, e002460 (2019).
Alfares, A. A. et al. Results of clinical genetic testing of 2,912 probands with hypertrophic cardiomyopathy: expanded panels offer limited additional sensitivity. Genet. Med. 17, 880–888 (2015).
Tadros, R. et al. Shared genetic pathways contribute to risk of hypertrophic and dilated cardiomyopathies with opposite directions of effect. Nat. Genet. 53, 128–134 (2021).
Neubauer, S. et al. Distinct subgroups in hypertrophic cardiomyopathy in the NHLBI HCM Registry. J. Am. Coll. Cardiol. 74, 2333–2345 (2019).
Canepa, M. et al. Temporal trend of age at diagnosis in hypertrophic cardiomyopathy: an analysis of the international Sarcomeric Human Cardiomyopathy Registry. Circ. Heart Fail. 13, e007230 (2020).
Pinto, Y. M. et al. Proposal for a revised definition of dilated cardiomyopathy, hypokinetic non-dilated cardiomyopathy, and its implications for clinical practice: a position statement of the ESC Working Group on Myocardial and Pericardial Diseases. Eur. Heart J. 37, 1850–1858 (2016).
Jordan, E. et al. Evidence-based assessment of genes in dilated cardiomyopathy. Circulation 144, 7–19 (2021).
Corrado, D. & Basso, C. Arrhythmogenic left ventricular cardiomyopathy. Heart 108, 733–743 (2022).
Marcus, F. I. et al. Diagnosis of arrhythmogenic right ventricular cardiomyopathy/dysplasia: proposed modification of the Task Force Criteria. Eur. Heart J. 31, 806–814 (2010).
Corrado, D. et al. Diagnosis of arrhythmogenic cardiomyopathy: the Padua criteria. Int. J. Cardiol. 319, 106–114 (2020).
Sheppard, M. N. et al. Genetically determined cardiomyopathies at autopsy: the pivotal role of the pathologist in establishing the diagnosis and guiding family screening. Virchows Arch. 482, 653–669 (2023).
James, C. A. et al. International evidence based reappraisal of genes associated with arrhythmogenic right ventricular cardiomyopathy using the Clinical Genome Resource framework. Circ. Genom. Precis. Med. 14, e003273 (2021).
Zeppenfeld, K. et al. 2022 ESC Guidelines for the management of patients with ventricular arrhythmias and the prevention of sudden cardiac death. Eur. Heart J. 43, 3997–4126 (2022).
Ortiz-Genga, M. F. et al. Truncating FLNC mutations are associated with high-risk dilated and arrhythmogenic cardiomyopathies. J. Am. Coll. Cardiol. 68, 2440–2451 (2016).
Gigli, M. et al. Genetic risk of arrhythmic phenotypes in patients with dilated cardiomyopathy. J. Am. Coll. Cardiol. 74, 1480–1490 (2019).
van den Hoogenhof, M. M. G. et al. RBM20 mutations induce an arrhythmogenic dilated cardiomyopathy related to disturbed calcium handling. Circulation 138, 1330–1342 (2018).
Wilde, A. A. M. et al. European Heart Rhythm Association (EHRA)/Heart Rhythm Society (HRS)/Asia Pacific Heart Rhythm Society (APHRS)/Latin American Heart Rhythm Society (LAHRS) Expert Consensus Statement on the state of genetic testing for cardiac diseases. Europace 24, 1307–1367 (2022). A comprehensive international expert statement outlining the role of genetic testing in all cardiomyopathies and in the setting of SCA.
Ommen, S. R. et al. 2020 AHA/ACC Guideline for the Diagnosis and Treatment of Patients With Hypertrophic Cardiomyopathy: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation 142, e558–e631 (2020).
Kamp, N. J. et al. Risk stratification using late gadolinium enhancement on cardiac magnetic resonance imaging in patients with hypertrophic cardiomyopathy: a systematic review and meta-analysis. Prog. Cardiovasc. Dis. 66, 10–16 (2021).
Thiene, G. et al. Arrhythmogenic right ventricular cardiomyopathy a still underrecognized clinic entity. Trends Cardiovasc. Med. 7, 84–90 (1997).
Visser, M. et al. Long-term outcome of patients initially diagnosed with idiopathic ventricular fibrillation: a descriptive study. Circ. Arrhythm. Electrophysiol. 9, e004258 (2016).
Ingles, J. et al. Concealed arrhythmogenic right ventricular cardiomyopathy in sudden unexplained cardiac death events. Circ. Genom. Precis. Med. 11, e002355 (2018). One of the earliest descriptions of CCMs caused by plakophilin 2 leading to SCA and SCD.
Zipes, D. P. & Wellens, H. J. Sudden cardiac death. Circulation 98, 2334–2351 (1998).
Bagnall, R. D. et al. A prospective study of sudden cardiac death among children and young adults. N. Engl. J. Med. 374, 2441–2452 (2016).
Krahn, A. D. et al. Systematic assessment of patients with unexplained cardiac arrest: Cardiac Arrest Survivors With Preserved Ejection Fraction Registry (CASPER). Circulation 120, 278–285 (2009).
Waldmann, V. et al. Characteristics and clinical assessment of unexplained sudden cardiac arrest in the real-world setting: focus on idiopathic ventricular fibrillation. Eur. Heart J. 39, 1981–1987 (2018).
Alqarawi, W. et al. Defining idiopathic ventricular fibrillation: a systematic review of diagnostic testing yield in apparently unexplained cardiac arrest. Heart Rhythm 18, 1178–1185 (2021).
Conte, G., Giudicessi, J. R. & Ackerman, M. J. Idiopathic ventricular fibrillation: the ongoing quest for diagnostic refinement. Europace 23, 4–10 (2021).
Behr, E. R. et al. Investigation on Sudden Unexpected Death in the Young (SUDY) in Europe: results of the European Heart Rhythm Association survey. Europace 24, 331–339 (2022).
de Noronha, S. V. et al. The importance of specialist cardiac histopathological examination in the investigation of young sudden cardiac deaths. Europace 16, 899–907 (2014).
Papadakis, M. et al. Sudden cardiac death with autopsy findings of uncertain significance: potential for erroneous interpretation. Circ. Arrhythm. Electrophysiol. 6, 588–596 (2013).
Geisterfer-Lowrance, A. A. et al. A molecular basis for familial hypertrophic cardiomyopathy: a β cardiac myosin heavy chain gene missense mutation. Cell 62, 999–1006 (1990).
Curran, M. E. et al. A molecular basis for cardiac arrhythmia: HERG mutations cause long QT syndrome. Cell 80, 795–803 (1995).
Wang, Q. et al. SCN5A mutations associated with an inherited cardiac arrhythmia, long QT syndrome. Cell 80, 805–811 (1995).
Musunuru, K. et al. Genetic testing for inherited cardiovascular diseases: a scientific statement from the American Heart Association. Circ. Genom. Precis. Med. 13, e000067 (2020).
Ackerman, M. J., Tester, D. J., C-bJ, Porter & Edwards, W. D. Molecular diagnosis of the inherited long-QT syndrome in a woman who died after near-drowning. N. Engl. J. Med. 341, 1121–1125 (1999).
Di Paolo, M., Luchini, D., Bloise, R. & Priori, S. G. Postmortem molecular analysis in victims of sudden unexplained death. Am. J. Forensic Med. Pathol. 25, 182–184 (2004).
Chugh, S. S. et al. Postmortem molecular screening in unexplained sudden death. J. Am. Coll. Cardiol. 43, 1625–1629 (2004).
Tester, D. J., Spoon, D. B., Valdivia, H. H., Makielski, J. C. & Ackerman, M. J. Targeted mutational analysis of the RyR2-encoded cardiac ryanodine receptor in sudden unexplained death: a molecular autopsy of 49 medical examiner/coroner’s cases. Mayo Clin. Proc. 79, 1380–1384 (2004).
Ackerman, M. J., Tester, D. J. & Driscoll, D. J. Molecular autopsy of sudden unexplained death in the young. Am. J. Forensic Med. Pathol. 22, 105–111 (2001).
Skinner, J. R. et al. Prospective, population-based long QT molecular autopsy study of postmortem negative sudden death in 1 to 40 year olds. Heart Rhythm 8, 412–419 (2011).
Tester, D. J., Medeiros-Domingo, A., Will, M. L., Haglund, C. M. & Ackerman, M. J. Cardiac channel molecular autopsy: insights from 173 consecutive cases of autopsy-negative sudden unexplained death referred for postmortem genetic testing. Mayo Clin. Proc. 87, 524–539 (2012).
Ackerman, M. J. et al. HRS/EHRA Expert Consensus Statement on the State of Genetic Testing for the Channelopathies and Cardiomyopathies: this document was developed as a partnership between the Heart Rhythm Society (HRS) and the European Heart Rhythm Association (EHRA). EP Europace 13, 1077–1109 (2011).
Campuzano, O. et al. Post-mortem genetic analysis in juvenile cases of sudden cardiac death. Forensic Sci. Int. 245, 30–37 (2014).
Bagnall, R. D., Das, K. J., Duflou, J. & Semsarian, C. Exome analysis-based molecular autopsy in cases of sudden unexplained death in the young. Heart Rhythm 11, 655–662 (2014).
Grondin, S. et al. Importance of genetic testing in unexplained cardiac arrest. Eur. Heart J. 43, 3071–3081 (2022).
Tester, D. J. et al. Plakophilin-2 truncation variants in patients clinically diagnosed with catecholaminergic polymorphic ventricular tachycardia and decedents with exercise-associated autopsy negative sudden unexplained death in the young. JACC Clin. Electrophysiol. 5, 120–127 (2019).
Isbister, J. C. et al. ‘Concealed cardiomyopathy’ as a cause of previously unexplained sudden cardiac arrest. Int. J. Cardiol. 324, 96–101 (2021).
Song, J. S. et al. Identification of pathogenic variants in genes related to channelopathy and cardiomyopathy in Korean sudden cardiac arrest survivors. J. Hum. Genet. 62, 615–620 (2017).
Lahrouchi, N. et al. Utility of post-mortem genetic testing in cases of sudden arrhythmic death syndrome. J. Am. Coll. Cardiol. 69, 2134–2145 (2017).
Webster, G. et al. Genomic autopsy of sudden deaths in young individuals. JAMA Cardiol. 6, 1247–1256 (2021).
Guo, L. et al. Genetic variants associated with unexplained sudden cardiac death in adult white and African American individuals. JAMA Cardiol. 6, 1013–1022 (2021).
Neves, R. et al. Exome sequencing highlights a potential role for concealed cardiomyopathies in youthful sudden cardiac death. Circ. Genom. Precis. Med. 15, e003497 (2022). This study highlights the importance of including cardiomyopathy genes when assessing unexplained SCD in the young.
Isbister, J. C. et al. Concealed cardiomyopathy in autopsy-inconclusive cases of sudden cardiac death and implications for families. J. Am. Coll. Cardiol. 80, 2057–2068 (2022). An autopsy-based study showing both the key genetic causes of CCMs and the importance of screening family members to prevent further arrhythmic events.
Rodrigues, P. et al. Diagnosis and prognosis in sudden cardiac arrest survivors without coronary artery disease: utility of a clinical approach using cardiac magnetic resonance imaging. Circ. Cardiovasc. Imaging 10, e006709 (2017).
Groeneveld, S. A. et al. Echocardiographic deformation imaging unmasks global and regional mechanical dysfunction in patients with idiopathic ventricular fibrillation: a multicenter case–control study. Heart Rhythm 18, 1666–1672 (2021).
Haissaguerre, M. et al. Localized structural alterations underlying a subset of unexplained sudden cardiac death. Circ. Arrhythm. Electrophysiol. 11, e006120 (2018).
Akhtar, M. M. et al. Clinical phenotypes and prognosis of dilated cardiomyopathy caused by truncating variants in the TTN gene. Circ. Heart Fail. 13, e006832 (2020).
Lakdawala, N. K. et al. Associations between female sex, sarcomere variants, and clinical outcomes in hypertrophic cardiomyopathy. Circ. Genom. Precis. Med. 14, e003062 (2021).
Augusto, J. B. et al. Dilated cardiomyopathy and arrhythmogenic left ventricular cardiomyopathy: a comprehensive genotype-imaging phenotype study. Eur. Heart J. Cardiovasc. Imaging 21, 326–336 (2020).
van der Voorn, S. M. et al. Arrhythmogenic cardiomyopathy: pathogenesis, pro-arrhythmic remodelling, and novel approaches for risk stratification and therapy. Cardiovasc. Res. 116, 1571–1584 (2020).
Lota, A. S. et al. Genetic architecture of acute myocarditis and the overlap with inherited cardiomyopathy. Circulation 146, 1123–1134 (2022).
Kim, J. C. et al. Disruption of Ca2+i homeostasis and connexin 43 hemichannel function in the right ventricle precedes overt arrhythmogenic cardiomyopathy in plakophilin-2-deficient mice. Circulation 140, 1015–1030 (2019).
Jiang, X. et al. Uncovering inherited cardiomyopathy with human induced pluripotent stem cells. Front. Cell Dev. Biol. 9, 672039 (2021).
Jung, P. et al. Increased cytosolic calcium buffering contributes to a cellular arrhythmogenic substrate in iPSC-cardiomyocytes from patients with dilated cardiomyopathy. Basic Res. Cardiol. 117, 5 (2022).
Semsarian, C. et al. Precision medicine in cardiovascular disease: genetics and impact on phenotypes: JACC Focus Seminar 1/5. J. Am. Coll. Cardiol. 77, 2517–2530 (2021).
Richards, S. et al. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet. Med. 17, 405–424 (2015).
Davies, B. et al. Variant reinterpretation in survivors of cardiac arrest with preserved ejection fraction (the Cardiac Arrest Survivors With Preserved Ejection Fraction Registry) by clinicians and clinical commercial laboratories. Circ. Genom. Precis. Med. 14, e003235 (2021).
Behr, E. R. Explaining the unexplained: applying genetic testing after cardiac arrest and sudden death. Eur. Heart J. 43, 3082–3084 (2022).
Nafissi, D. A. et al. Prevalence and phenotypic burden of monogenic arrhythmias using integration of electronic health records with genetics. Circ. Genom. Precis. Med. 15, e003675 (2022).
Abdulrahim, J. W. et al. Identification of undetected monogenic cardiovascular disorders. J. Am. Coll. Cardiol. 76, 797–808 (2020).
Shah, R. A. et al. Frequency, penetrance, and variable expressivity of dilated cardiomyopathy-associated putative pathogenic gene variants in UK Biobank participants. Circulation 146, 110–124 (2022). Up to 20% of adults with putative pathogenic variants in DCM genes exhibit early DCM features, most commonly manifesting with arrhythmias in the absence of substantial ventricular dilation or dysfunction.
Carruth, E. D. et al. Loss-of-function FLNC variants are associated with arrhythmogenic cardiomyopathy phenotypes when identified through exome sequencing of a general clinical population. Circ. Genom. Precis. Med. 15, e003645 (2022).
Mellor, G. et al. Genetic testing in the evaluation of unexplained cardiac arrest: from the CASPER (Cardiac Arrest Survivors With Preserved Ejection Fraction Registry). Circ. Cardiovasc. Genet. 10, 278–285 (2017).
Lehman, S. J., Crocini, C. & Leinwand, L. A. Targeting the sarcomere in inherited cardiomyopathies. Nat. Rev. Cardiol. 19, 353–363 (2022).
Acknowledgements
C.S. is the recipient of a National Health and Medical Research Council Investigator grant (2016822) and a New South Wales Health Cardiovascular Disease Clinician Scientist grant. R.T. holds the Canada Research Chair in Translational Cardiovascular Genetics.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review information
Nature Cardiovascular Research thanks the anonymous reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Isbister, J.C., Tadros, R., Raju, H. et al. Concealed cardiomyopathy as an emerging cause of sudden cardiac arrest and sudden cardiac death. Nat Cardiovasc Res 3, 1274–1283 (2024). https://doi.org/10.1038/s44161-024-00558-1
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s44161-024-00558-1
This article is cited by
-
Brugada Syndrome: an exemplar for the genomic basis of sudden death
European Journal of Human Genetics (2025)