Abstract
Mammalian Sterile20-like kinase 1 (Mst1) is a member of the yeast Ste20-related kinase family known to be involved in the process of apoptosis initiated by a variety of stimuli. The aim of this study was to determine the prognostic significance of Mst1 expression in colorectal cancer. A series of 1197 mismatch-repair-proficient colorectal cancers retrieved from a tissue microarray were randomized into two study groups. On the first group (n=599) receiver operating characteristic curves were used to determine the most clinically useful cutoffs to describe Mst1 expression with respect to T stage, N stage, tumor grade, vascular invasion and overall survival. The association of Mst1 expression and each outcome was immunohistochemically evaluated on the second study group (n=598) as well as on a third study group comprising 141 mismatch-repair-deficient colorectal cancers. A univariate analysis in the second study group showed that loss of cytoplasmic Mst1 was associated with higher T stage (P=0.001), higher N stage (P=0.029), vascular invasion (P=0.017) and overall survival (P=0.014). Nuclear Mst1 expression was not significantly associated with N stage, T stage or vascular invasion but was associated with tumor grade. In mismatch-repair-deficient colorectal cancers, loss of cytoplasmic Mst1 was associated with higher N stage (P=0.019) and shortened survival (P=0.0001). In a multivariate analysis, loss of cytoplasmic Mst1 was an independent adverse prognostic factor in this group of patients. Methylation analysis on 32 cases showed that the loss of cytoplasmic Mst1 expression is not likely due to promoter methylation. In summary, loss of cytoplasmic Mst1 expression is a marker of tumor progression in mismatch-repair-proficient as well as mismatch-repair-deficient colorectal cancers. These results are suggestive of a tumor suppressor role for Mst1 in human colorectal cancer.
Similar content being viewed by others
Main
Mst1 (mammalian Sterile-like 1), a serine/threonine kinase, belongs to the Sterile 20-like superfamily that was originally identified as a stress-activated protein participating in a wide range of apoptotic responses.1, 2 Mst1 contains an N-terminal catalytic domain and an autoinhibitory segment followed by a dimerization domain and a nuclear localization motif at the non catalytic, C-terminal part.3 Mst1 also contains a caspase-3-recognition motif, between the catalytic and regulatory domains, which mediates the cleavage of the autoinhibitory domain and yields a highly active catalytic fragment during apoptosis (Figure 1).4, 5, 6 Despite extensive studies, the mechanisms and targets through which Mst1 regulates apoptosis are not well characterized. The pro-apoptotic function of Mst1—at least in part—is attributed to caspase-3 mediated cleavage of Mst1, although subsequent studies demonstrated that this is not essential for signaling and morphological features of apoptosis.7 In fact, Mst1 has also shown to be activated by phosphorylation of Thr183 and Thr187 in its activation loop which is promoted by dimerization. Mst1 phosphorylation results in caspase activation and apoptosis7, 8 indicating that Mst1 might function both upstream and downstream of caspases. Recent findings that most Ste20 group kinases activate mitogen-activated protein kinase (MAPK) and more specifically stressed-induced p38 MAPK and JNK (c-Jun N-terminal kinase) support this model (Figure 1).1, 8, 9, 10
Cellular localization of Mst1 is also regulated by caspase-mediated cleavage as well as threonine phosphorylation.7, 8, 11 Under resting conditions, Mst1 is localized predominantly in the cytoplasm but cleavage of Mst1 by caspases or phosphorylation in its activation loop lead to Mst1 translocation to the nucleus.7, 8, 11 Nuclear retention of Mst1 results in chromatin condensation and DNA fragmentation indicating that nuclear entry of Mst1 may be important in its pro-apoptotic function.11, 12 Extensive studies to identify potential Mst1 substrates in the nucleus have led to identification of Histone H2B as a physiologic substrate for the catalytic domain of Mst1, the phosphorylation of which leads to chromatin condensation and apoptosis.13
Based on its proposed function in apoptosis, Mst1 might play a tumor suppressor role in human cancers as has been suggested for its Drosophila homologs.14, 15, 16, 17 Nevertheless, there is no report on the biological significance of Mst1 expression in primary human cancer cells. Herein, we report the prognostic significance of cytoplasmic and nuclear expression of Mst1 in a large group of patients with colorectal cancer.
Materials and methods
Tissue Microarray Construction
A tissue microarray comprising of 1420 unselected, non-consecutive colorectal cancers was constructed.18 Formalin-fixed, paraffin-embedded tissue blocks of colorectal cancer resections were retrieved from the archives of the Institute of Pathology, University Hospital of Basel, Switzerland, the Institute of Clinical Pathology, Basel, Switzerland and the Institute of Pathology, Stadtspital Triemli, Zürich, Switzerland. One tissue cylinder with a diameter of 0.6 mm was punched from morphologically representative tissue areas of each ‘donor’ tissue block and brought into one recipient paraffin block (3 × 2.5 cm) using a homemade semiautomated tissue arrayer.
Clinico-Pathological Parameters
The clinico-pathologic data for 1420 patients included T stage (T1, T2, T3 and T4), N stage (N0, N1 and N2), tumor grade (G1, G2 and G3) (overall grading based on the most poorly differentiated area but excluding tumor budding at the tumor margin), vascular invasion (intramural or extramural) and survival. The distribution of these features has been described previously.19
Immunohistochemistry of Tissue Microarray
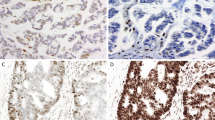
Sections (4 μm) of tissue microarray blocks were transferred to an adhesive-coated slide system (Instrumedics Inc., Hackensack, NJ, USA) to facilitate the transfer of tissue sections to slides and to minimize tissue loss. Standard indirect immunoperoxidase procedures were used for IHC (Dako Envision+). 1420 colorectal cancers and 57 normal colonic mucosa samples were immunostained for N-terminal region of Mst1 (dilution 1:200; Cell Signaling, Danvers, USA), MLH1 (clone MLH-1; dilution 1:100; BD Biosciences Pharmingen, San Jose, CA, USA), MSH2 (clone MSH-2; dilution 1:200; BD Biosciences Pharmingen, San Jose, CA, USA), MSH6 (clone 44; dilution 1:400; BD Biosciences Pharmingen, San Jose, CA, USA). After dewaxing and rehydration in dH2O, sections for immunostaining were subjected to heat antigen retrieval in a microwave oven (1200 W, 15 min) in 1 mM EDTA buffer pH 8.0 for Mst1, MLH1, MSH2 and in 0.01 mol/l citrate buffer pH 6.0 for MSH6. Endogenous peroxidase activity was blocked using 0.5% H2O2. After transfer to a humidified chamber, the sections were incubated with 10% normal goat serum (Dako Cytomation) for 20 min and incubated with primary antibody at 4°C overnight for hMLH1, hMSH2 and hMSH6 and Mst1. Subsequently, the sections were incubated with a peroxidase-labeled secondary antibody (K4005, EnVision+ System-HRP (AEC); DakoCytomation) for 30 min at room temperature. For visualization of the antigen, the sections were immersed in 3-amino-9-ethylcarbazole+substrate-chromogen (K4005, EnVision+ System-HRP (AEC); DakoCytomation) for 30 min, and counterstained lightly with Gill's haematoxylin (Figure 2).
Different colorectal cancer punches showing varying positivity of Mst1 staining (a) (× 2). Predominantly cytoplasmic Mst1 expression in two moderately differentiated MMR-proficient colorectal cancer (arrow: negative stroma with few scattered Mst1 positive inflammatory cells) (b, c) (× 40). Nuclear Mst1 expression in a moderately differentiated MMR-proficient colon cancer (d) (× 40).
Evaluation of Immunohistochemistry
Cytoplasmic and nuclear Mst1 immunoreactivity were separately evaluated semi-quantitatively using %—positivity defined as the proportion of Mst1 positive tumor cells over the total number of tumor cells ranging from 0 to 100%. Scores were based on 5% intervals (0, 5, 10%, etc). The 1420 colorectal cancers were scored by one experienced surgical pathologist (AL) blinded to the clinico-pathological features. MLH1, MSH2 and MSH6 were scored as negative (0% staining) or positive (>0% staining).
Mismatch-Repair Status
The 1420 colorectal cancers were stratified according to DNA mismatch-repair (MMR) status and consisted of 1197 MMR-proficient tumors expressing MLH1, MSH2 and MSH6, 141 MLH1-negative tumors, and 82 presumed HNPCC cases demonstrating loss of MSH2 and/or MSH6 at any age (N=73), or loss of MLH1 at <55 years (N=9).20 Only the MMR-proficient tumors were randomized into two matched groups in view of the large number of available cases (N=1197, 84.4%).
Randomization of MMR-Proficient Colorectal Cancers
The 1197 MMR-proficient colorectal cancers were randomly assigned into two subgroups, study group 1 (N=599) and study group 2 (N=598) (Table 1). Study group 1 served to determine the most relevant cutoffs of cytoplasmic and nuclear Mst1 positivity and the clinico-pathologic features. The associations of Mst1 expression at the proposed cutoffs with T stage, N stage, tumor grade, vascular invasion and survival were investigated on study group 2 as well as in the third group containing 141 cases with MLH1-negative tumors.
DNA Extraction and Bisulphite Modification
Samples were micro-dissected from paraffin-embedded tissue using two 8-micron-thick sections. Cell lysis and DNA extraction were performed using a QIAamp DNA mini kit (QIAGEN, Mississauga, ON, Canada) according to manufacturer's protocol. Extracted genomic DNA was diluted in 40 μl of distilled water and denatured by adding 6 μl of 2 N NaOH and incubation at 75°C for 20 min. 500 μl of freshly prepared 4.8 M sodium bisulphite and 28 μl of 10 mM hydroquinone were added to the denatured genomic DNA and the reaction was carried out overnight in dark at 55°C. DNA then was purified using a Wizard DNA clean-up (Promega, Madison, WI, USA) and then ethanol-precipitated after 5 min of alkali treatment with 8.8 μl of 2 N NaOH at room temperature.
Methylation of Mst1
Methylation of Mst1 promoter was examined by methylation-specific PCR (MSP) using AmpliTaq Gold kit (Roche, Branchburg, NJ, USA) as described previously.21 The primers for amplification of unmethylated sequence were 5′-TTTTGGTAATGTAGGAAGATAGTGT-3′ and 3′-AAACCACAACCCTAATCACATA-5′ and primer sequences for methylated reaction were 5′-GTTTCGGTAACGTAGGAAGATAGC-3′ and 3′-AACCACGACCCTAATCACGTA-5′. The condition for amplification was 10 min at 95°C followed by 39 cycles of denaturing at 95°C for 30 s, annealing at 56°C for 30 s and 30 s of extension at 72°C. PCR products were subjected to electrophoresis on 8% acrylamide gels and visualized by SYBR gold nucleic acid gel stain (Molecular Probes, Eugene, USA). CpGenome Universal Methylated DNA (Chemicon, Temecula, CA, USA) was used as a positive control for methylation.
Statistical Analysis
The selection of clinically important cutoff scores for Mst1 expression was based on receiver operating characteristic (ROC) curve analysis.22 At each percentage score, the sensitivity and specificity for the outcome under study was plotted, thus generating a ROC curve. The score having the shortest distance to the point with both maximum sensitivity and specificity, that is, the point (0.0, 1.0) on the curve, and thereby minimizing the trade-off between the two, was selected as the cutoff score. Categorizing tumors as positive or negative around this score leads to the greatest number of tumors correctly classified as having or not having the clinical outcome. The reliability of the selected cutoff scores was determined by 1000-bootstrapped replications to re-sample the data. With bootstrapping, 1000 re-samples of equal size were created. ROC curve analysis was performed for each sub-group and a cutoff score was obtained. The most frequently obtained score over the 1000 re-samples was selected as the cutoff score for Mst1 positivity. In order to use ROC curve analysis, the clinico-pathological features were dichotomized: T stage (early (T1+T2) or late (T3+T4)), N stage (N0 (no lymph node involvement) or >N0 (any lymph node involvement)), tumor grade (low (G1+G2) or high (G3)), vascular invasion (absent or present), and survival (death due to colorectal cancer or censored (lost to follow-up, alive or death from other causes)).
The association of cytoplasmic and nuclear Mst1 (retention/loss) and T stage, N stage, tumor grade and vascular invasion were evaluated at their respective cutoffs using the χ2 test. Univariate survival analysis was carried out using the Kaplan–Meier method, and by Cox proportional hazards regression. Multivariate survival analysis was performed by including T stage, N stage, tumor grade, vascular invasion and Mst1 expression. All analyses were carried out using SAS, version 9 (The SAS Institute, Cary, NC, USA).
Results
Normal Colonic Mucosa
In normal colonic mucosa Mst1 immunoreactivity regardless of the extent occurred in 73.7% of cases.
Cytoplasmic Mst1 in MMR-Proficient Colorectal Cancer
(Table 2) The optimum cutoff for loss of cytoplasmic Mst1 was <80% for all clinico-pathological parameters. In a χ2 test, loss of cytoplasmic Mst1 was observed more frequently in tumor stages T3 and T4, while retention of cytoplasmic Mst1 was associated with early (T1 and T2) tumor stages (P=0.001) (Table 2). Additionally there was an association between loss of cytoplasmic Mst1 and presence of lymph node metastasis (P=0.029), vascular invasion (P=0.017) and worse survival (P=0.014) (Mst1 retention: 81.0 months; Mst1 loss: 52.0 months) (Figure 3).
Nuclear Mst1 in MMR-Proficient Colorectal Cancer
(Table 3) The optimum cutoffs for nuclear Mst1 as determined on subgroup 1 differed for the various endpoints: 50% for T stage, 40% for N stage and vascular invasion, 70% for tumor grade and 30% for overall survival. In a univariate analysis nuclear Mst1 expression was not significantly associated with N stage, T stage or vascular invasion.
Cytoplasmic Mst1 in MMR-Deficient Colorectal Cancer
(Table 4) The optimum cutoff for cytoplasmic Mst1 was also found to be 80% for all clinico-pathological parameters. In a univariate analysis, loss of cytoplasmic Mst1 was associated with the presence of lymph node metastasis (P=0.019) and worse survival (P=0.0001) (Mst1 retention: 74.1 months; Mst1 loss: 31.7 months) (Figure 4).
Multivariate Analysis of Survival
In a multivariate analysis, which included T stage, N stage, tumor grade, vascular invasion and age, loss of cytoplasmic Mst1 was not an independent prognostic factor in MMR-proficient colorectal cancers (P=0.679). However, multivariate analysis using the same criteria demonstrated that loss of cytoplasmic Mst1 was an independent adverse prognostic factor in MMR-deficient colorectal cancers (P=0.03).
Methylation Analysis of Mst1 Promoter
In order to investigate whether loss of Mst1 expression is a result of promoter methylation, we carried out MSP studies on 16 cases with near complete or complete loss of Mst1 expression in MLH1 proficient group and 16 cases in MLH1 negative group. The results showed no methylation in the selected promoter region of Mst1 in both groups (data not shown).
Discussion
In this study, we analyzed the prognostic value of Mst1 expression in 1197 MMR-proficient as well as 141 MMR-deficient colorectal cancers using tissue microarray technology and immunohistochemistry. The tissue microarray technique is an accepted tool of investigation, especially when a huge number of samples are analyzed.18, 23, 24, 25, 26, 27, 28 The randomization of the study population into two subgroups allowed us to determine the best cutoffs for cytoplasmic and nuclear Mst1 expression on the first subgroup (N=599) based on the ROC curve analysis and to apply the determined cutoffs on the second subgroup (N=598) as well as on the third group (N=141). This approach has the important advantage of avoiding a scoring system that includes a subjectively selected cutoff or a complex composite scoring system. Indeed, the different cutoffs for nuclear Mst1 dependent on the clinico-pathological parameter analyzed clearly underlines the importance of ROC curve analysis in the immunohistochemical analysis of potential tumor markers.
In normal colon mucosa Mst1 was found predominantly in the cytoplasm but there was nuclear staining to a lesser extent. This is consistent with the finding that endogenous Mst1 accumulates in the nucleus upon treatment with leptomycin B—a specific inhibitor of nuclear export—suggesting that Mst1 shuttles continuously between cytoplasm and nucleus.7, 29
Univariate analysis in the MMR-proficient group showed that loss of cytoplasmic Mst1 was associated with higher T and N stages, vascular invasion and worse survival. These results are consistent with Mst1 function in induction of apoptosis and tumor suppression. Patients with MMR-proficient colorectal cancers are more likely to have APC, KRAS, and P53 mutations and are microsatellite stable (MSS).30 There has been increasing evidence that Ras and other oncogenes paradoxically induce both pro- and anti-apoptotic signaling.31, 32 NORE1 and RASSF1 are two members of the RASSF family known to have putative RAS-binding domains and mediate RAS-induced apoptotic signals.33, 34, 35 Further studies demonstrated that these two proteins are binding partners of Mst1 and their complex formation with Mst1 is critical to the mechanism of RAS-induced apoptosis (Figure 1).36, 37 Loss of Mst1 expression will change the balance of RAS-induced contradictory signals in favor of anti-apoptotic ones and provide a further explanation for the association of clinico-pathological outcomes and Mst1 expression in MMR proficient colorectal cancers.
Based on the proposed mechanism of Mst1 in induction of apoptosis (see introductory text) we expected to observe an association between the loss of Mst1 nuclear expression with worse clinico-pathological outcomes. However, in our analysis, Mst1 nuclear expression did not show a significant association with tumor stage, vascular invasion or survival. As nuclear localization of Mst1 is promoted by caspase-mediated cleavage and threonine phosphorylation, it is conceivable that these two modifications interfere with antigen/antibody complexing in immunohistochemistry.
In the MMR-deficient group, we found that 80% was also the best cutoff for different clinico-pathological variables. Univariate analysis in this group showed that the loss of cytoplasmic Mst1 is associated with lymph node metastasis and worse survival. These results firstly confirm the role of Mst1 as a tumor suppressor gene and secondly indicate that the 80% cutoff for cytoplasmic expression of Mst1 is a valid and useful criterion in prediction of clinico-pathologic features of a tumor. Loss of MLH1 expression is highly deleterious and results in apoptosis due to the rapid accumulation of DNA mismatch errors. Thus, loss of MLH1 expression is only tolerated when the mechanisms leading to apoptosis are extensively disrupted. In fact, inhibition of apoptosis has been regarded as a central pathogenic mechanism in development of serrated polyps which are the likely precursors of sporadic MSI-H colorectal cancers.38, 39 This might explain why in the multivariate analysis, loss of cytoplasmic Mst1 showed a significant effect on survival only in the MLH1-negative group.
CpG island methylation is an epigenetic mechanism commonly involved in silencing of gene transcription leading to development of cancer.40 Methylation in the promoter region of Mst1 has been reported in normal colon mucosa in patients with hyperplastic polyposis.21 Our methylation analysis showed that methylation of gene promoter is not a likely mechanism for explaining Mst1 loss of expression in MMR proficient and MMR deficient colorectal cancers. Hence, we suggest the presence of other mechanisms, (eg mutation, loss of heterozygosity) may be responsible for downregulation of Mst1 expression in colorectal cancers.
In summary, the results of this study indicate that loss of cytoplasmic Mst1 is associated with higher T and/or N stage, higher tumor grade and poor prognosis in colorectal cancer patients, suggesting a tumor suppressor role for Mst1 in human colorectal cancer.
References
Creasy CL, Chernoff J . Cloning and characterization of a member of the MST subfamily of Ste20-like kinases. Gene 1995;167:303–306.
Taylor LK, Wang HC, Erikson RL . Newly identified stress-responsive protein kinases, Krs-1 and Krs-2. Proc Natl Acad Sci USA 1996;93:10099–10104.
Creasy CL, Ambrose DM, Chernoff J . The Ste20-like protein kinase, Mst1, dimerizes and contains an inhibitory domain. J Biol Chem 1996;271:21049–21053.
Reszka AA, Halasy-Nagy JM, Masarachia PJ, et al. Bisphosphonates act directly on the osteoclast to induce caspase cleavage of mst1 kinase during apoptosis. A link between inhibition of the mevalonate pathway and regulation of an apoptosis-promoting kinase. J Biol Chem 1999;274:34967–34973.
Lee KK, Murakawa M, Nishida E, et al. Proteolytic activation of MST/Krs, STE20-related protein kinase, by caspase during apoptosis. Oncogene 1998;16:3029–3037.
Kakeya H, Onose R, Osada H . Caspase-mediated activation of a 36-kDa myelin basic protein kinase during anticancer drug-induced apoptosis. Cancer Res 1998;58:4888–4894.
Lee KK, Yonehara S . Phosphorylation and dimerization regulate nucleocytoplasmic shuttling of mammalian STE20-like kinase (MST). J Biol Chem 2002;277:12351–12358.
Glantschnig H, Rodan GA, Reszka AA . Mapping of MST1 kinase sites of phosphorylation. Activation and autophosphorylation. J Biol Chem 2002;277:42987–42996.
Graves JD, Gotoh Y, Draves KE, et al. Caspase-mediated activation and induction of apoptosis by the mammalian Ste20-like kinase Mst1. EMBO J 1998;17:2224–2234.
Ura S, Masuyama N, Graves JD, et al. MST1-JNK promotes apoptosis via caspase-dependent and independent pathways. Genes Cells 2001;6:519–530.
Lee KK, Ohyama T, Yajima N, et al. MST, a physiological caspase substrate, highly sensitizes apoptosis both upstream and downstream of caspase activation. J Biol Chem 2001;276:19276–19285.
Ura S, Masuyama N, Graves JD, et al. Caspase cleavage of MST1 promotes nuclear translocation and chromatin condensation. Proc Natl Acad Sci USA 2001;98:10148–10153.
Cheung WL, Ajiro K, Samejima K, et al. Apoptotic phosphorylation of histone H2B is mediated by mammalian sterile twenty kinase. Cell 2003;113:507–517.
Wu S, Huang J, Dong J, et al. hippo encodes a Ste-20 family protein kinase that restricts cell proliferation and promotes apoptosis in conjunction with salvador and warts. Cell 2003;114:445–456.
Jia J, Zhang W, Wang B, et al. The Drosophila Ste20 family kinase dMST functions as a tumor suppressor by restricting cell proliferation and promoting apoptosis. Genes Dev 2003;17:2514–2519.
Harvey KF, Pfleger CM, Hariharan IK . The Drosophila Mst ortholog, hippo, restricts growth and cell proliferation and promotes apoptosis. Cell 2003;114:457–467.
Pantalacci S, Tapon N, Leopold P . The Salvador partner Hippo promotes apoptosis and cell-cycle exit in Drosophila. Nat Cell Biol 2003;5:921–927.
Sauter G, Simon R, Hillan K . Tissue microarrays in drug discovery. Nat Rev Drug Discov 2003;2:962–972.
Lugli A, Zlobec I, Gunthert U, et al. Overexpression of the receptor for hyaluronic acid mediated motility is an independent adverse prognostic factor in colorectal cancer. Mod Pathol 2006;19:1302–1309.
Hampel H, Frankel WL, Martin E, et al. Screening for the Lynch syndrome (hereditary nonpolyposis colorectal cancer). N Engl J Med 2005;352:1851–1860.
Minoo P, Baker K, Goswami R, et al. Extensive DNA methylation in normal colorectal mucosa in hyperplastic polyposis. Gut 2006;55:1467–1474.
Hanley JA . Receiver operating characteristic (ROC) methodology: the state of the art. Crit Rev Diagn Imaging 1989;29:307–335.
Moch H, Schraml P, Bubendorf L, et al. High-throughput tissue microarray analysis to evaluate genes uncovered by cDNA microarray screening in renal cell carcinoma. Am J Pathol 1999;154:981–986.
Nocito A, Bubendorf L, Tinner EM, et al. Microarrays of bladder cancer tissue are highly representative of proliferation index and histological grade. J Pathol 2001;194:349–357.
Simon R, Nocito A, Hubscher T, et al. Patterns of her-2/neu amplification and overexpression in primary and metastatic breast cancer. J Natl Cancer Inst 2001;93:1141–1146.
Torhorst J, Bucher C, Kononen J, et al. Tissue microarrays for rapid linking of molecular changes to clinical endpoints. Am J Pathol 2001;159:2249–2256.
Goethals L, Perneel C, Debucquoy A, et al. A new approach to the validation of tissue microarrays. J Pathol 2006;208:607–614.
Barlund M, Forozan F, Kononen J, et al. Detecting activation of ribosomal protein S6 kinase by complementary DNA and tissue microarray analysis. J Natl Cancer Inst 2000;92:1252–1259.
Lin Y, Khokhlatchev A, Figeys D, et al. Death-associated protein 4 binds MST1 and augments MST1-induced apoptosis. J Biol Chem 2002;277:47991–48001.
Vogelstein B, Fearon ER, Hamilton SR, et al. Genetic alterations during colorectal-tumor development. N Engl J Med 1988;319:525–532.
Cox AD, Der CJ . The dark side of Ras: regulation of apoptosis. Oncogene 2003;22:8999–9006.
Feig LA, Buchsbaum RJ . Cell signaling: life or death decisions of ras proteins. Curr Biol 2002;12:R259–R261.
Vavvas D, Li X, Avruch J, et al. Identification of Nore1 as a potential Ras effector. J Biol Chem 1998;273:5439–5442.
Dammann R, Li C, Yoon JH, et al. Epigenetic inactivation of a RAS association domain family protein from the lung tumour suppressor locus 3p21.3. Nat Genet 2000;25:315–319.
Lerman MI, Minna JD . The 630-kb lung cancer homozygous deletion region on human chromosome 3p21.3: identification and evaluation of the resident candidate tumor suppressor genes. The International Lung Cancer Chromosome 3p21.3 Tumor Suppressor Gene Consortium. Cancer Res 2000;60:6116–6133.
Khokhlatchev A, Rabizadeh S, Xavier R, et al. Identification of a novel Ras-regulated proapoptotic pathway. Curr Biol 2002;12:253–265.
Praskova M, Khoklatchev A, Ortiz-Vega S, et al. Regulation of the MST1 kinase by autophosphorylation, by the growth inhibitory proteins, RASSF1 and NORE1, and by Ras. Biochem J 2004;381:453–462.
Tateyama H, Li W, Takahashi E, et al. Apoptosis index and apoptosis-related antigen expression in serrated adenoma of the colorectum: the saw-toothed structure may be related to inhibition of apoptosis. Am J Surg Pathol 2002;26:249–256.
Jass JR, Whitehall VL, Young J, et al. Emerging concepts in colorectal neoplasia. Gastroenterology 2002;123:862–876.
Herman JG, Baylin SB . Gene silencing in cancer in association with promoter hypermethylation. N Engl J Med 2003;349:2042–2054.
Acknowledgements
This study was supported by a grant from the Swiss National Foundation (Grant no. PBBSB-110417) and the Novartis Foundation, formerly Ciba-Geigy-Jubilee-Foundation. We thank Privatdozent Dr Hanspeter Spichtin, Institute of Clinical Pathology Basel, Switzerland and Professor Dr Robert Maurer, Institute of Pathology, Stadtspital Triemli, Zuerich, Switzerland for providing the cases.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Minoo, P., Zlobec, I., Baker, K. et al. Prognostic significance of mammalian sterile20-like kinase 1 in colorectal cancer. Mod Pathol 20, 331–338 (2007). https://doi.org/10.1038/modpathol.3800740
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/modpathol.3800740
Keywords
This article is cited by
-
RASSF1A–Hippo pathway link in patients with urothelial carcinoma of bladder: plausible therapeutic target
Molecular and Cellular Biochemistry (2020)
-
Identification of microRNA signature in the progression of gestational trophoblastic disease
Cell Death & Disease (2018)
-
Deacetylation of tumor-suppressor MST1 in Hippo pathway induces its degradation through HBXIP-elevated HDAC6 in promotion of breast cancer growth
Oncogene (2016)
-
Molecular chaperone Hsp27 regulates the Hippo tumor suppressor pathway in cancer
Scientific Reports (2016)
-
MicroRNA-18a is elevated in prostate cancer and promotes tumorigenesis through suppressing STK4 in vitro and in vivo
Oncogenesis (2014)