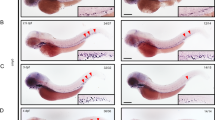
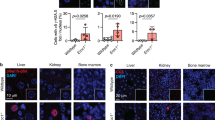
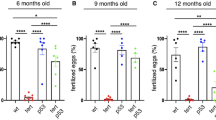
Abstract
Cell culture work has identified the tumor suppressor p53 as a component of the S-phase checkpoint control system, while in vivo studies of this role of p53 in whole-vertebrate systems were limited. Here, we describe zebrafish mutants in the DNA polymerase delta catalytic subunit 1, based on the positional cloning of the flathead (fla) gene. fla mutants display specific defects in late proliferative zones, such as eyes, brain and cartilaginous elements of the visceral head skeleton, where cells display compromised DNA replication, followed by apoptosis, and partial or complete loss of affected tissues. Antisense-mediated knockdown of p53 in fla mutants leads to a striking rescue of all phenotypic traits, including completion of replication, survival of cells, and normal differentiation and tissue formation. This indicates that under replication-compromised conditions, the p53 branch of the S-phase checkpoint is responsible for eliminating stalled cells that, given more time, would have otherwise finished their normal developmental program.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Accession codes
Abbreviations
- AP:
-
anterior–posterior
- ATR:
-
Ataxia telangiectasia mutated (ATM) and Rad3 related
- BrdU:
-
5′ Bromo-2′desoxyuridine
- Chk1:
-
checkpoint kinase 1
- DAPI:
-
4′,6-Diamidino-2-phenylindole
- dpf:
-
days postfertilization
- ef1α:
-
elongation factor 1 alpha
- FACS:
-
fluorescence-activated cell sorting
- fla:
-
flathead mutant
- hpf:
-
hours postfertilization
- MO:
-
morpholino oligonucleotide
- pcna:
-
proliferating-cell nuclear antigen
- polδ1:
-
DNA polymerase delta1
- RT-PCR:
-
polymerase chain reaction after reverse transcription of RNA
- SSLP:
-
simple sequence length polymorphism
- TGFβ:
-
transforming growth factor beta
- TUNEL:
-
terminal desoxynucleotidyl transferase-mediated biotinylated UTP nick end labeling
- 4mm:
-
four mismatch control
References
Hindges R and Hübscher U (1997) DNA polymerase delta, an essential enzyme for DNA transactions. Biol. Chem. 378: 345–362
Hübscher U, Maga G and Spadari S (2002) Eukaryotic DNA polymerases. Annu. Rev. Biochem. 71: 33–63
Waga S and Stillman B (1994) Anatomy of a DNA replication fork revealed by reconstitution of SV40 DNA replication in vitro. Nature 369: 207–212
Francesconi S, Park H and Wang TS (1993) Fission yeast with DNA polymerase delta temperature-sensitive alleles exhibits cell division cycle phenotype. Nucleic Acids Res. 21: 3821–3828
Fukui T, Yamauchi K, Muroya T, Akiyama M, Maki H, Sugino A and Waga S (2004) Distinct roles of DNA polymerases delta and epsilon at the replication fork in Xenopus egg extracts. Genes Cells 9: 179–191
Goldsby AE, Lawrence NA, Hays LE, Olmsted EA, Chen X, Singh M and Preston BD (2001) Defective DNA polymerase-δ proofreading causes cancer susceptibility in mice. Nat. Med. 7: 638–639
Goldsby AE, Hays LE, Chen X, Olmsted EA, Slayton WB, Spangrude GJ and Preston BD (2002) High incidence of epithelial cancers in mice deficient for DNA polymerase δ proofreading. Proc. Natl. Acad. Sci. USA 99: 15560–15565
Nyberg KA, Michelson RJ, Putnam CW and Weinert TA (2002) Toward maintaining the genome: DNA damage and replication checkpoints. Annu. Rev. Genet. 36: 617–656
Bartek J, Lukas C and Lukas J (2004) Checking on DNA damage in S phase. Nat. Rev. Mol. Cell Biol. 5: 792–804
Taylor WR, Agarwal ML, Agarwal A, Stacey DW and Stark GR (1999) p53 inhibits entry into mitosis when DNA synthesis is blocked. Oncogene 18: 283–295
Taylor WR and Stark GR (2001) Regulation of the G2/M transition by p53. Oncogene 20: 1803–1815
Haupt S, Berger M, Goldberg Z and Haupt Y (2003) Apoptosis – the p53 network. J. Cell Sci. 116: 4077–4085
Tibbets RS, Brumbaugh KM, Williams JM, Sarkaria JN, Cliby WA, Shieh S-Y, Taya Y, Prives C and Abraham RT (1999) A role of ATR in the DNA damage-induced phosphorylation of p53. Genes Dev. 13: 152–157
Lakin ND, Hann BC and Jackson SP (1999) The ataxia-telangiectasia related protein ATR mediates DNA-dependent phosphorylation of p53. Oncogene 18: 3989–3995
Shieh SY, Ahn J, Tamai K, Taya Y and Prives C (2000) The human homologs of checkpoint kinases Chk1 and Cds1 (Chk2) phosphorylate p53 at multiple DNA damage-inducible sites. Genes Dev. 14: 289–300
Sanchez Y, Wong C, Thoma RS, Richman R, Wu Z, Piwnica-Worms H and Elledge SJ (1997) Conservation of the Chk1 checkpoint pathway in mammals: linkage of DNA damage to Cdk regulation through Cdc25. Science 277: 1497–1501
Liu Q, Guntuku S, Cui XS, Matsuoka S, Cortez D, Tamai K, Luo G, Carattini-Rivera S, DeMayo F, Bradley A, Donehower LA and Elledge SJ (2000) Chk1 is an essential kinase that is regulated by Atr and required for the G(2)/M DNA damage checkpoint. Genes Dev. 14: 1448–1459
Takai H, Tominaga K, Motoyama N, Minamishima YA, Nagahama H, Tsukiyama T, Ikeda K, Nakayama K and Nakanishi M (2000) Aberrant cell cycle checkpoint function and early embryonic death in Chk1(−/−) mice. Genes Dev. 14: 1439–1447
Guo Z, Kumagai A, Wang SX and Dunphy WG (2000) Requirement for Atr in phosphorylation of Chk1 and cell cycle regulation in response to DNA replication blocks and UV-damaged DNA in Xenopus egg extracts. Genes Dev. 14: 2745–2756
Feijoo C, Hall-Jackson C, Wu R, Jenkins D, Leitch J, Gilbert DM and Smythe C (2001) Activation of mammalian Chk1 during DNA replication arrest: a role for Chk1 in the intra-S phase checkpoint monitoring replication origin firing. J. Cell Biol. 154: 913–923
Haffter P, Granato M, Brand M, Mullins MC, Hammerschmidt M, Kane DA, Odenthal J, van Eeden FJM, Jiang Y-J, Heisenberg C-P, Kelsh RN, Furutani-Seiki M, Warga RM, Vogelsang E, Beuchle D, Schach U, Fabian C and Nüsslein-Volhard C (1996) The identification of genes with unique and essential functions in the development of the zebrafish, Danio rerio. Development 123: 1–36
Driever W, Solnica-Krezel L, Schier AF, Neuhauss SCF, Malicki J, Stemple DL, Stanier DYR, Zwartkruis F, Abdeliah S, Rangini Z, Belak J and Boggs C (1996) A genetic screen for mutations affecting embryogenesis in zebrafish. Development 123: 37–46
Schilling TF, Piotrowski T, Grandel H, Brand M, Heisenberg CP, Jiang YJ, Beuchle D, Hammerschmidt M, Kane DA, Mullins MC, van Eeden FJ, Kelsh RN, Furutani-Seiki M, Granato M, Haffter P, Odenthal J, Warga RM, Trowe T and Nusslein-Volhard C (1996) Jaw and branchial arch mutants in zebrafish I: branchial arches. Development 123: 329–344
Westerfield M (1994) The Zebrafish Book: A Guide for the Laboratory use of Zebrafish (Eugene, Oregon: University of Oregon Press)
Schilling TF (1997) Genetic analysis of craniofacial development in the vertebrate embryo. BioEssays 19: 459–468
Rauch G-J, Granato M and Haffter P (1997) A polymorphic zebrafish line for genetic mapping using SSLPs on high-percentage agarose gels Technical Tips Online T01208
Geisler R (2002) In Zebrafish: A Practical Approach, Nüsslein-Volhard C, Dahm R, eds (Oxford: Oxford University Press) pp. 175–212
Hammerschmidt M, Pelegri F, Mullins MC, Kane DA, van Eeden FJM, Granato M, Brand M, Furutani-Seiki M, Haffter P, Heisenberg C-P, Jiang Y-J, Kelsh RN, Odenthal J, Warga RM and Nüsslein-Volhard C (1996) dino and mercedes, two genes regulating dorsal development in the zebrafish embryo. Development 123: 95–102
Akimenko M-A, Ekker M, Wegner J, Lin W and Westerfield M (1994) Combinatorial expression of three zebrafish genes related to distal-less: part of a homeobox gene code for the head. J. Neurosci. 16: 3475–3486
Chiang EF-L, Pai CI, Wyatt M, Yan Y-L, Postlethwait J and Chung B-C (2001) Two Sox9 genes on duplicated zebrafish chromosomes: expression of similar transcription activators in distinct sites. Dev. Biol. 231: 149–163
Lee J-S and Gye MC (1999) Zebrafish Danio rerio proliferating cell nuclear antigen (PCNA) cloning and characterization. Fish. Sci. 65: 955–958
Schulte-Merker S, Ho RK, Herrmann BG and Nüsslein-Volhard C (1992) The protein product of the zebrafish homologue of the mouse T gene is expressed in nuclei of the germ ring and the notochord of the early embryo. Development 116: 1021–1032
Cole LK and Ross LS (2001) Apoptosis in the developing zebrafish embryo. Dev. Biol. 240: 123–142
Herzog W, Sonntag C, von der Hardt S, Roehl HH, Varga ZM and Hammerschmidt M (2004) Fgf3 signaling from the ventral diencephalon is required for early specification and subsequent survival of the zebrafish adenohypophysis. Development 131: 3681–3692
Nasevicius A and Ekker SC (2000) Effective targeted gene ‘knockdown’ in zebrafish. Nat. Genet. 26: 216–220
Langheinrich U, Hennen E, Stott G and Vacun G (2002) Zebrafish as a model organism for the identification and characterization of drugs and genes affecting p53 signaling. Curr. Biol. 12: 2023–2028
Nordness S, Krauss S and Johansen T (1994) cDNA sequence of zebrafish (Brachydanio rerio) translation elongation factor-1 alpha: molecular phylogeny of eukaryotes based on elongation factor-1 alpha protein sequences. Biochim. Biophys. Acta 1219: 529–532
Cheng R, Ford BL, O’Neal PE, Mathews CZ, Bradford CS, Thongtan T, Barnes DW, Hendricks JD and Bailey GS (1997) Zebrafish (Danio rerio) p53 tumor suppressor gene: cDNA sequence and expression during embryogenesis. Mol. Mar. Biol. Biotechnol. 6: 88–97
Liu TX, Howlett NG, Deng M, Langenau DM, Hsu K, Rhodes J, Kanki JP, DAndrea AD and Look AT (2003) Knockdown of zebrafish Fancd2 causes developmental abnormalities via p53-dependent apoptosis. Dev. Cell 5: 903–914
Nowak M, Köster C and Hammerschmidt M (2005) Perp is required for tissue-specific cell survival during zebrafish development. Cell Death. Diff. 12: 52–64
Livak KJ and Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 25: 402–408
Schilling T, Walker C and Kimmel CK (1996) The chinless mutation and neural crest cell interactions in zebrafish jaw development. Development 122: 1417–1426
David NB, Saint-Etienne L, Tsang M, Schilling TF and Rosa FM (2002) Requirement for endoderm and FGF3 in ventral skeleton formation. Development 129: 4457–4468
Mizuno T, Yamagishi K, Miyazawa H and Hanaoka F (1999) Molecular architecture of the mouse DNA polymerase α-primase complex. Mol. Cell. Biol. 19: 7886–7896
Sanchez Garcia J, Cuifo LF, Kearsey SE and MacNeill SA (2004) The C-terminal zinc finger of the catalytic subunit of DNA polymerase δ is responsible for direct interaction with the B-subunit. Nucl. Acids Res. 32: 3005–3016
Dua R, Levy DL and Campbell JL (1999) Analysis of essential functions of the C-terminal protein/protein interaction domain of Saccharomyces cerevisiae pol e and its unexpected abilitiy to support growth in the absence of the DNA polymerase domain. J. Biol. Chem. 274: 22283–22288
Simon M, Giot L and Faye G (1993) A random mutagenesis procedure – application to the pol3 gene of Saccharomyces cerevisiae. Gene 127: 139–144
Kane DA and Kimmel CB (1993) The zebrafish midblastula transition. Development 119: 447–456
El-Deiry WS, Tokino T, Velculescu VE, Levy DB, Parsons R, Trent JM, Lin D, Mercer WE, Kinzler KW and Vogelstein B (1993) WAF1, a potential mediator of p53 tumor suppression. Cell 75: 817–825
Niculescu III AB, Chen X, Smeets M, Hengst L, Prives C and Reed SI (1998) Effects of p21 (Cip1/Waf1) at both the G1/S and the G2/M cell cycle transition: pRb is a critical determinant in blocking DNA replication and in preventing endoreduplication. Mol. Cell. Biol. 18: 629–643
Mayashita T and Reed JC (1995) Tumor suppressor p53 is a direct transcriptional activator of the human BAX gene. Cell 80: 293–299
Hengarter MO (2000) The biochemistry of apoptosis. Nature 407: 770–776
Donehower LA, Harvey M, Slagle BL, McArthur MJ, Montgomery Jr CA, Butel JS and Bradley A (1992) Mice deficient for p53 are developmentally normal but susceptible to spontaneous tumours. Nature 356: 215–221
Wallingford JB, Seufert DW, Virta VC and Vize PD (1997) p53 activity is essential for normal development in Xenopus. Curr. Biol. 7: 747–757
Takebayashi-Suzuki K, Funami J, Tokumori D, Saito A, Watabe T, Miyazono K, Kanda A and Suzuki A (2003) Interplay between the tumor suppressor p53 and TFGb signaling shapes embryonic body axes in Xenopus. Development 130: 3929–3939
Cordenonsi M, Dupont S, Maretto S, Insinga A, Imbriano C and Piccolo S (2003) Links between tumor suppressors: p53 is required for TGF-beta gene responses by cooperating with Smads. Cell 113: 301–314
Ikegami R, Rivera-Bennetts AK, Brooker DL and Yager TD (1997) Effects of inhibitors of DNA replication on early zebrafish embryos. Evidence for coordinate activation of multiple intrinstic checkpoints at midblastula transition. Zygote 5: 153–175
Postlethwait JH, Yan Y-L, Gates MA, Horne S, Amores A, Brownlie A, Donovan A, Egan ES, Force A, Gong Z, Goutel C, Fritz A, Kelsh R, Knapik E, Liao E, Paw B, Ransom D, Singer A, Thomson M, Abduljabbar TS, Yelik P, Beier D, Joly J-S, Larhammar D, Rosa F, Westerfield M, Zon LI, Johnson SL and Talbot WS (1998) Vertebrate genome evolution and the zebrafish gene map. Nat. Genet. 18: 345–349
Waga S, Masuda T, Takisawa H and Sugino A (2001) DNA polymerase e is required for coordinated and efficient chromosomal DNA replication in Xenopus egg extracts. Proc. Natl. Acad. Sci. USA 98: 4978–4983
Hammerschmidt M, Blader P and Strähle U (1999) In Methods in Cell Biology, Vol. 59, Detrich HW, Westerfield M, Zon LI (eds) (San Diego: Academic Press) pp. 87–115
Sugo N, Niimi N, Aratani Y, Takiguchi-Hayashi K and Koyama H (2004) p53 deficiency rescues neuronal apoptosis but not differentiation in DNA polymerase beta-deficient mice. Mol. Cell. Biol. 24: 9470–9477
Kim JM, Nakao K, Nakamura K, Saito I, Katsuki M, Arai K-i and Masai H (2002) Inactivation of Cdc kinase in mouse ES cells results in S-phase arrest and p53-dependent cell death. EMBO J. 21: 2168–2179
Rich T, Allen RL and Wyllie AH (2000) Defying death after DNA damage. Nature 407: 777–783
Schilling TF and Kimmel CB (1997) Musculoskeletal patterning in the pharyngeal segments of the zebrafish. Development 124: 2945–2960
Neuhauss SCF, Solnica-Krezel L, Schier AF, Zwartkruis F, Stemple DL, Malicki DL, Abdelilah S, Stainier DYR and Driever W (1996) Mutations affecting craniofacial development in zebrafish. Development 123: 357–367
Acknowledgements
We thank M Nowak and T Schilling for stimulating discussions, Soojin Ryu and Wolfgang Driever for communicating unpublished results, F van Eeden (pcna), Robert Kelsh (sox9) and Marc Ekker (dlx2) for reagents, and HG Fronhoefer from the Tuebingen stock center for fla mutant fish. NP is grateful to the Alexander von Humboldt Foundation and to EMBO for her long-term postdoctoral fellowships. Work in MH's laboratory was supported by the Max-Planck Society.
Author information
Authors and Affiliations
Corresponding author
Additional information
Edited by V De Laurenzi
Supplementary Information accompanies the paper on Cell Death and Differentiation website (http://www.nature.com/cdd)
Supplementary information
Rights and permissions
About this article
Cite this article
Plaster, N., Sonntag, C., Busse, C. et al. p53 deficiency rescues apoptosis and differentiation of multiple cell types in zebrafish flathead mutants deficient for zygotic DNA polymerase δ1. Cell Death Differ 13, 223–235 (2006). https://doi.org/10.1038/sj.cdd.4401747
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/sj.cdd.4401747
Keywords
This article is cited by
-
Antagonistic interactions safeguard mitotic propagation of genetic and epigenetic information in zebrafish
Communications Biology (2024)
-
Zebrafish Noxa promotes mitosis in early embryonic development and regulates apoptosis in subsequent embryogenesis
Cell Death & Differentiation (2014)
-
Plasticity of photoreceptor-generating retinal progenitors revealed by prolonged retinoic acid exposure
BMC Developmental Biology (2011)
-
Chromatin remodelling complex dosage modulates transcription factor function in heart development
Nature Communications (2011)
-
The zebrafish as a model organism for the study of apoptosis
Apoptosis (2010)