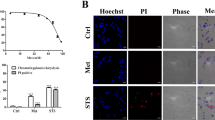
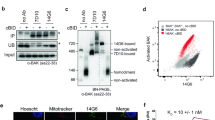
Abstract
The ubiquitous vacuolar H+-ATPase, a multisubunit proton pump, is essential for intraorganellar acidification. Disruption of its function leads to disturbances of organelle function and cell death. Here, we report that overexpression of the B2 subunit of the H+-ATPase inhibits apoptosis. This antiapoptotic effect is not mediated by an increase in H+-ATPase activity but through activation of the Ras-mitogen-activated protein kinase (MAPK)-signaling pathway that results in the serine phosphorylation of Bad at residues 112 and 155. Increased Bad phosphorylation reduces its translocation to mitochondria, limits the release of mitochondrial cytochrome c and apoptosis-inducing factor and increases the resistance of the B2 overexpressing cells to apoptosis. Screening experiments of kinase inhibitors, including inhibitors of cAMP-activated protein kinase, protein kinase C, protein kinase B, (MAPK/extracellular signal-regulated (ERK) kinase) MEK and Ste-MEK113, a cell permeable ERK activation inhibitor peptide, revealed that the B2 subunit of H+-ATPase acts upstream of MEK activation in the MEK/ERK pathway to ameliorate apoptosis.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Abbreviations
- MAPK:
-
mitogen-activated protein kinase
- ERK:
-
extracellular-signal-regulated kinase
- MEK:
-
MAPK/ERK kinase
- RSK:
-
ribosomal S6 kinase
- PKA:
-
cAMP-activated protein kinase
- PKC:
-
protein kinase C
- AKT:
-
protein kinase B
- AIF:
-
apoptosis-inducing factor
- GFP:
-
green fluorescent protein
References
Wagner CA, Finberg KE, Breton S, Marshansky V, Brown D and Geibel JP (2004) Renal vacuolar H+-ATPase. Physiol. Rev. 84: 1263–1314.
Hishita T, Tada-Oikawa S, Tohyama K, Miura Y, Nishihara T, Tohyama Y, Yoshida Y, Uchiyama T and Kawanishi S (2001) Caspase-3 activation by lysosomal enzymes in cytochrome c-independent apoptosis in myelodysplastic syndrome-derived cell line P39. Cancer Res. 61: 2878–2884.
Torigoe T, Izumi H, Ishiguchi H, Uramoto H, Murakami T, Ise T, Yoshida Y, Tanabe M, Nomoto M, Itoh H and Kohno K (2002) Enhanced expression of the human vacuolar H+-ATPase c subunit gene (ATP6L) in response to anticancer agents. J. Biol. Chem. 277: 36534–36543.
Aiko K, Tsujisawa T, Koseki T, Hashimoto S, Morimoto Y, Amagasa T and Nishihara T (2002) Involvement of cytochrome c and caspases in apoptotic cell death of human submandibular gland ductal cells induced by concanamycin A. Cell Signal 14: 717–722.
Zhan H, Yokoyama K, Otani H, Tanigaki K, Shirota N, Takano S and Ohkuma S (2003) Different roles of proteolipids and 70-kDa subunits of V-ATPase in growth and death of cultured human cells. Genes Cells 8: 501–513.
Ishisaki A, Hashimoto S, Amagasa T and Nishihara T (1999) Caspase-3 activation during the process of apoptosis induced by a vacuolar type H(+)-ATPase inhibitor. Biol. Cell 91: 507–513.
Niessen H, Meisenholder GW, Li HL, Gluck SL, Lee BS, Bowman B, Engler RL, Babior BM and Gottlieb RA (1997) Granulocyte colony-stimulating factor upregulates the vacuolar proton ATPase in human neutrophils. Blood 90: 4598–4601.
Lee BS, Krits I, Crane-Zelkovic MK and Gluck SL (1997) A novel transcription factor regulates expression of the vacuolar H+-ATPase B2 subunit through AP-2 sites during monocytic differentiation. J. Biol. Chem. 272: 174–181.
Anderson CL and Williams GT (2003) Apoptosis gene hunting using retroviral expression cloning: identification of vacuolar ATPase subunit E. Sci World J 3: 51–58.
Hengartner MO (2000) The biochemistry of apoptosis. Nature 407: 770–776.
van Loo G, Saelens X, van Gurp M, MacFarlane M, Martin SJ and Vandenabeele P (2002) The role of mitochondrial factors in apoptosis: a Russian roulette with more than one bullet. Cell Death Differ. 9: 1031–1042.
Susin SA, Lorenzo HK, Zamzami N, Marzo I, Snow BE, Brothers GM, Mangion J, Jacotot E, Costantini P, Loeffler M, Larochette N, Goodlett DR, Aebersold R, Siderovski DP, Penninger JM and Kroemer G (1999) Molecular characterization of mitochondrial apoptosis-inducing factor. Nature 397: 441–446.
Datta SR, Ranger AM, Lin MZ, Sturgill JF, Ma YC, Cowan CW, Dikkes P, Korsmeyer SJ and Greenberg ME (2002) Survival factor-mediated Bad phosphorylation raises the mitochondrial threshold for apoptosis. Dev. Cell 3: 631–643.
Zhou XM, Liu Y, Payne G, Lutz RJ and Chittenden T (2000) Growth factors inactivate the cell death promoter Bad by phosphorylation of its BH3 domain on Ser155. J. Biol. Chem. 275: 25046–25051.
Kolch W (2000) Meaningful relationships: the regulation of the Ras/Raf/MEK/ERK pathway by protein interactions. Biochem. J. 2: 289–305.
Datta SR, Dudek H, Tao X, Masters S, Fu H, Gotoh Y and Greenberg ME (1997) Akt phosphorylation of Bad couples survival signals to the cell-intrinsic death machinery. Cell 91: 231–241.
Blume-Jensen P, Janknecht R and Hunter T (1998) The kit receptor promotes cell survival via activation of PI3-kinase and subsequent Akt-mediated phosphorylation of Bad on Ser136. Curr. Biol. 8: 779–782.
Bertolotto C, Maulon L, Filippa N, Baier G and Auberger P (2000) Protein kinase C theta and epsilon promote T-cell survival by a rsk-dependent phosphorylation and inactivation of BAD. J. Biol. Chem. 275: 37246–37250.
Tan Y, Ruan H, Demeter MR and Comb MJ (1999) p90(RSK) blocks bad-mediated cell death via a protein kinase C-dependent pathway. J. Biol. Chem. 274: 34859–34867.
Harada H, Becknell B, Wilm M, Mann M, Huang LJ, Taylor SS, Scott JD and Korsmeyer SJ (1999) Phosphorylation and inactivation of Bad by mitochondria anchored protein kinase A. Mol. Cell 3: 413–422.
Kelemen BR, Hsiao K and Goueli SA (2002) Selective in vivo inhibition of mitogen-activated protein kinase activation using cell-permeable peptides. J. Biol. Chem. 277: 8741–8748.
Brognard J and Dennis PA (2002) Variable apoptotic response of NSCLC cells to inhibition of the MEK/ERK pathway by small molecules or dominant negative mutants. Cell Death Differ. 9: 893–904.
Bonni A, Brunet A, West AE, Datta SR, Takasu MA and Greenberg ME (1999) Cell survival promoted by the Ras-MAPK signaling pathway by transcription-dependent and independent mechanism. Science 286: 1358–1362.
Chiang CW, Kanies C, Kim KW, Fang WB, Parkhurst C, Xie M, Henry T and Yang E (2003) Protein phosphatase 2A dephosphorylation of phosphoserine 112 plays the gatekeeper role for Bad-mediated apoptosis. Mol. Cell Biol. 23: 6350–6362.
Schurmann A, Mooney AF, Sanders LC, Sells MA, Wang HG, Reed JC and Bokoch GM (2000) p21-activated kinase 1 phosphorylates the death agonist Bad and protects cells from apoptosis. Mol. Cell Biol. 20: 453–461.
Tan Y, Demeter MR, Ruan H and Comb MJ (2000) Bad Ser-155 phosphorylation regulates Bad/Bcl-XL interaction and cell survival. J. Biol. Chem. 275: 25865–25869.
Jouret F, Auzanneau C, Debaix H, Wada GH, Pretto C, Marbaix E, Karet FE, Courtoy PJ and Devuyst O (2005) Ubiquitous and kidney-specific subunits of vacuolar H+-ATPase are differentially expressed during nephrogenesis. J. Am. Soc. Nephrol. 16: 3235–3246.
Ruchalski K, Mao H, Singh SK, Wang Y, Mosser DD, Li F, Schwartz JH and Borkan SC (2003) HSP72 inhibits apoptosis-inducing factor release in ATP-depleted renal epithelial cells. Am. J. Physiol. 285: C1483–C1493.
Huang TG and Hackney DD (1994) Drosophilia kinesin minimal motor domain expressed in Escherichia coli. J. Biol. Chem. 269: 16493–16501.
Kiianitsa K, Solinger JA and Heyer WD (2003) NADH-coupled microplate photometric assay for kinetic studies of ATP-hydrolyzed enzymes with low and high specific activities. Anal. Biochem. 321: 266–271.
Acknowledgements
This research is supported by grants obtained from National Institutes of Health (National Institute of Diabetes and Digestive and Kidney Diseases): JH Schwartz (DK-52898) and (DK 59529) and SC Borkan (DK-53387). We also appreciate the gift of rabbit antibody to the 31 kD subunit H+-ATPase from Dr Dennis Brown.
Author information
Authors and Affiliations
Corresponding author
Additional information
Edited by JA Cidlowski
Supplementary Information accompanies the paper on Cell Death and Differentiation website (http://www.nature.com/cdd)
Rights and permissions
About this article
Cite this article
Li, G., Yang, Q., Krishnan, S. et al. A novel cellular survival factor – the B2 subunit of vacuolar H+-ATPase inhibits apoptosis. Cell Death Differ 13, 2109–2117 (2006). https://doi.org/10.1038/sj.cdd.4401970
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/sj.cdd.4401970
Keywords
This article is cited by
-
Interaction between V-ATPase B2 and (Pro) renin Receptors in Promoting the progression of Renal Tubulointerstitial Fibrosis
Scientific Reports (2016)
-
The medaka mutation tintachina sheds light on the evolution of V-ATPase B subunits in vertebrates
Scientific Reports (2013)
-
NMR solution structure of subunit E (fragment E1–69) of the Saccharomyces cerevisiae V1VO ATPase
Journal of Bioenergetics and Biomembranes (2011)
-
Cytosolic pH is a second messenger for glucose and regulates the PKA pathway through V-ATPase
The EMBO Journal (2010)