Abstract
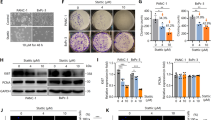
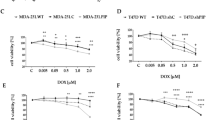
The p57Kip2 gene belongs to the Cip/Kip family of cyclin-dependent kinase (CDK) inhibitors and has been suggested to be a tumor suppressor gene, being inactivated in various types of human cancers. However, little is known concerning p57Kip2 possible interplay with the apoptotic cell death machinery and its possible implication for cancer. Here, we report that selective p57Kip2 expression sensitizes cancer cells to apoptotic agents such as cisplatin, etoposide and staurosporine (STS) via a mechanism, which does not require p57Kip2-mediated inhibition of CDK. Translocation of p57Kip2 to mitochondria occurs within 20 min after STS application. In fact, p57Kip2 primarily promotes the intrinsic apoptotic pathways, favoring Bax activation and loss of mitochondrial transmembrane potential, consequent release of cytochrome-c into cytosol, caspase-9 and caspase-3 activation. In accordance, Bcl2 overexpression or voltage-dependent anion channel (VDAC) inhibition is able to inhibit p57Kip2 cell death promoting effect. Thus, in addition to its established function in control of proliferation, these results reveal a mechanism whereby p57Kip2 influences the mitochondrial apoptotic cell death pathway in cancer cells.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Abbreviations
- αFas:
-
agonistic anti-Fas Abs
- AIF:
-
apoptosis inducing factor
- CDK:
-
cyclin-dependent kinase
- CKI:
-
CDK inhibitor
- CP:
-
cisplatin
- cyt-c:
-
cytochrome-c
- ΔΨm:
-
mitochondrial transmembrane potential
- DIDS:
-
4,4′-diisothiocyanatostilbene-2,2′-disulfonic acid
- Dox:
-
doxycycline
- FACS:
-
fluorescence-activated cell sorting
- G3PDH:
-
glyceraldehyde-3-phosphate dehydrogenase
- GFP:
-
green fluorescent protein
- MEF:
-
mouse embryo fibroblasts
- NLS:
-
nuclear localization signal
- PARP:
-
poly(ADP-ribose)polymerase
- PI:
-
propidium iodide
- STS:
-
staurosporine
- TMRE:
-
tetramethylrhodamine ethyl ester
- VP16:
-
etoposide
- VDAC:
-
voltage-dependent anion channel
References
Evan GI, Vousden KH . Proliferation, cell cycle and apoptosis in cancer. Nature 2001; 411: 342–348.
Lee MH, Reynisdottir I, Massague J . Cloning of p57KIP2, a cyclin-dependent kinase inhibitor with unique domain structure and tissue distribution. Genes Dev 1995; 9: 639–649.
Matsuoka S, Edwards MC, Bai C, Parker S, Zhang P, Baldini A et al. p57KIP2, a structurally distinct member of the p21CIP1 Cdk inhibitor family, is a candidate tumor suppressor gene. Genes Dev 1995; 9: 650–662.
Yan Y, Frisen J, Lee MH, Massague J, Barbacid M . Ablation of the CDK inhibitor p57Kip2 results in increased apoptosis and delayed differentiation during mouse development. Genes Dev 1997; 11: 973–983.
Zhang P, Liegeois NJ, Wong C, Finegold M, Hou H, Thompson JC et al. Altered cell differentiation and proliferation in mice lacking p57KIP2 indicates a role in Beckwith–Wiedemann syndrome. Nature 1997; 387: 151–158.
Dyer MA, Cepko CL . p57(Kip2) regulates progenitor cell proliferation and amacrine interneuron development in the mouse retina. Development 2000; 127: 3593–3605.
Joseph B, Wallen-Mackenzie A, Benoit G, Murata T, Okret S, Perlmann T . p57(Kip2) cooperates with Nurr1 in developing dopamine cells. Proc Natl Acad Sci USA 2003; 100: 15619–15624.
Li JQ, Wu F, Usuki H, Kubo A, Masaki T, Fujita J et al. Loss of p57KIP2 is associated with colorectal carcinogenesis. Int J Oncol 2003; 23: 1537–1543.
Nakai S, Masaki T, Shiratori Y, Ohgi T, Morishita A, Kurokohchi K et al. Expression of p57(KIP2) in hepatocellular carcinoma: relationship between tumor differentiation and patient survival. Int J Oncol 2002; 20: 769–775.
Sui L, Dong Y, Ohno M, Watanabe Y, Sugimoto K, Tokuda M . Expression of p57kip2 and its clinical relevance in epithelial ovarian tumors. Anticancer Res 2002; 22: 3191–3196.
Shin JY, Kim HS, Lee KS, Kim J, Park JB, Won MH et al. Mutation and expression of the p27KIP1 and p57KIP2 genes in human gastric cancer. Exp Mol Med 2000; 32: 79–83.
Kobatake T, Yano M, Toyooka S, Tsukuda K, Dote H, Kikuchi T et al. Aberrant methylation of p57KIP2 gene in lung and breast cancers and malignant mesotheliomas. Oncol Rep 2004; 12: 1087–1092.
Samuelsson MK, Pazirandeh A, Davani B, Okret S . p57Kip2, a glucocorticoid-induced inhibitor of cell cycle progression in HeLa cells. Mol Endocrinol 1999; 13: 1811–1822.
Watanabe H, Pan ZQ, Schreiber-Agus N, DePinho RA, Hurwitz J, Xiong Y . Suppression of cell transformation by the cyclin-dependent kinase inhibitor p57KIP2 requires binding to proliferating cell nuclear antigen. Proc Natl Acad Sci USA 1998; 95: 1392–1397.
Hengartner MO . The biochemistry of apoptosis. Nature 2000; 407: 770–776.
Desagher S, Martinou JC . Mitochondria as the central control point of apoptosis. Trends Cell Biol 2000; 10: 369–377.
Hsu YT, Youle RJ . Bax in murine thymus is a soluble monomeric protein that displays differential detergent-induced conformations. J Biol Chem 1998; 273: 10777–10783.
Martinou JC, Green DR . Breaking the mitochondrial barrier. Nat Rev Mol Cell Biol 2001; 2: 63–67.
Tsujimoto Y, Shimizu S . VDAC regulation by the Bcl-2 family of proteins. Cell Death Differ 2000; 7: 1174–1181.
Pateras IS, Apostolopoulou K, Koutsami M, Evangelou K, Tsantoulis P, Liloglou T et al. Downregulation of the KIP family members p27(KIP1) and p57(KIP2) by SKP2 and the role of methylation in p57(KIP2) inactivation in nonsmall cell lung cancer. Int J Cancer 2006; 119: 2546–2556.
Yue H, Na YL, Feng XL, Ma SR, Song FL, Yang B . Expression of p57kip2, Rb protein and PCNA and their relationships with clinicopathology in human pancreatic cancer. World J Gastroenterol 2003; 9: 377–380.
Ito Y, Yoshida H, Nakano K, Kobayashi K, Yokozawa T, Hirai K et al. Expression of p57/Kip2 protein in normal and neoplastic thyroid tissues. Int J Mol Med 2002; 9: 373–376.
Shen L, Toyota M, Kondo Y, Obata T, Daniel S, Pierce S et al. Aberrant DNA methylation of p57KIP2 identifies a cell-cycle regulatory pathway with prognostic impact in adult acute lymphocytic leukemia. Blood 2003; 101: 4131–4136.
Hatada I, Inazawa J, Abe T, Nakayama M, Kaneko Y, Jinno Y et al. Genomic imprinting of human p57KIP2 and its reduced expression in Wilms' tumors. Hum Mol Genet 1996; 5: 783–788.
Kikuchi T, Toyota M, Itoh F, Suzuki H, Obata T, Yamamoto H et al. Inactivation of p57KIP2 by regional promoter hypermethylation and histone deacetylation in human tumors. Oncogene 2002; 21: 2741–2749.
Chim CS, Wong AS, Kwong YL . Epigenetic inactivation of the CIP/KIP cell-cycle control pathway in acute leukemias. Am J Hematol 2005; 80: 282–287.
Yang H, Hoshino K, Sanchez-Gonzalez B, Kantarjian H, Garcia-Manero G . Antileukemia activity of the combination of 5-aza-2'-deoxycytidine with valproic acid. Leuk Res 2005; 29: 739–748.
Gonzalez S, Perez-Perez MM, Hernando E, Serrano M, Cordon-Cardo C . p73beta-Mediated apoptosis requires p57kip2 induction and IEX-1 inhibition. Cancer Res 2005; 65: 2186–2192.
Hsu S, Yu FS, Lewis J, Singh B, Borke J, Osaki T et al. Induction of p57 is required for cell survival when exposed to green tea polyphenols. Anticancer Res 2002; 22: 4115–4120.
Chang TS, Kim MJ, Ryoo K, Park J, Eom SJ, Shim J et al. p57KIP2 modulates stress-activated signaling by inhibiting c-Jun NH2-terminal kinase/stress-activated protein kinase. J Biol Chem 2003; 278: 48092–48098.
Kamura T, Hara T, Kotoshiba S, Yada M, Ishida N, Imaki H et al. Degradation of p57Kip2 mediated by SCFSkp2-dependent ubiquitylation. Proc Natl Acad Sci USA 2003; 100: 10231–10236 [E-pub 2003 Aug 18].
Denicourt C, Dowdy SF . Cip/Kip proteins: more than just CDKs inhibitors. Genes Dev 2004; 18: 851–855.
Matsumoto M, Furihata M, Ohtsuki Y, Sasaguri S, Ogoshi S . Immunohistochemical characterization of p57KIP2 expression in human esophageal squamous cell carcinoma. Anticancer Res 2000; 20: 1947–1952.
Coqueret O . New roles for p21 and p27 cell-cycle inhibitors: a function for each cell compartment? Trends Cell Biol 2003; 13: 65–70.
Yokoo T, Toyoshima H, Miura M, Wang Y, Iida KT, Suzuki H et al. p57Kip2 regulates actin dynamics by binding and translocating LIM-kinase 1 to the nucleus. J Biol Chem 2003; 278: 52919–52923.
Suzuki A, Tsutomi Y, Yamamoto N, Shibutani T, Akahane K . Mitochondrial regulation of cell death: mitochondria are essential for procaspase 3–p21 complex formation to resist Fas-mediated cell death. Mol Cell Biol 1999; 19: 3842–3847.
Morisaki T, Katano M . Mitochondria-targeting therapeutic strategies for overcoming chemoresistance and progression of cancer. Curr Med Chem 2003; 10: 2517–2521.
Joseph B, Marchetti P, Formstecher P, Kroemer G, Lewensohn R, Zhivotovsky B . Mitochondrial dysfunction is an essential step for killing of non-small cell lung carcinomas resistant to conventional treatment. Oncogene 2002; 21: 65–77.
Acknowledgements
We thank Drs JC Cross (Samuel Lunenfeld Research Institute), J Massagué (Sloan-Kettering Cancer Center), DJ McConkey (MD Anderson Cancer Center), S Okret (Karolinska Institute), CL Stewart (National Cancer Institute), Y Xiong (University of North Carolina), A Vartapetian (Moscow State University), G Kochs (University of Freiburg) and LG Larsson (Swedish University of agricultural sciences) for providing us with different DNA constructs, HeLa and MEF cell lines. We thank Drs B Zhivotovsky, S Orrenius, O Hermansson, M Malewicz and T Perlmann for critical reading of the manuscript. This work was supported by grants from the Swedish Research Council, the Swedish Cancer Society, the Åke Wiberg Foundation and the Swedish Medical Society.
Author information
Authors and Affiliations
Corresponding author
Additional information
Edited by K Vousden
Supplementary Information accompanies the paper on Cell Death and Differentiation website (http://www.nature.com/cdd)
Supplementary information
Rights and permissions
About this article
Cite this article
Vlachos, P., Nyman, U., Hajji, N. et al. The cell cycle inhibitor p57Kip2 promotes cell death via the mitochondrial apoptotic pathway. Cell Death Differ 14, 1497–1507 (2007). https://doi.org/10.1038/sj.cdd.4402158
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/sj.cdd.4402158
Keywords
This article is cited by
-
The bone marrow niche regulates redox and energy balance in MLL::AF9 leukemia stem cells
Leukemia (2022)
-
Combined inhibition of BET proteins and class I HDACs synergistically induces apoptosis in urothelial carcinoma cell lines
Clinical Epigenetics (2018)
-
Genome-wide identification of directed gene networks using large-scale population genomics data
Nature Communications (2018)
-
p57Kip2 is an unrecognized DNA damage response effector molecule that functions in tumor suppression and chemoresistance
Oncogene (2015)
-
Combined inhibition of BET family proteins and histone deacetylases as a potential epigenetics-based therapy for pancreatic ductal adenocarcinoma
Nature Medicine (2015)