Abstract
Aims To evaluate the efficacy and safety of latanoprost/timolol maleate fixed combination (LTFC) given once daily vsthe concomitant therapy of brimonidine twice daily and latanoprost once daily in primary open-angle glaucoma or ocular hypertensive subjects.
Methods A prospective, double-masked, active-controlled comparison in which qualified subjects had all glaucoma medicines discontinued for 1 month and then were randomized to either LTFC or brimonidine and latanoprost concomitant therapy for 6 weeks. They were then switched to the other treatment regimen. The intraocular pressure (IOP) was measured at 0800, 1200, and 1600 h at baseline and at the end of Periods 1 and Period 2.
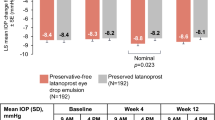
Results In 32 subjects, the diurnal curve of the untreated IOP of 26.0±3.4 decreased to 17.8±2.5 on LTFC and 17.2±2.8 mmHg on brimonidine and latanoprost (P=0.31). At 0800 and 1600 h, the IOPs were statistically similar between the groups (P>0.05). At 1200 h the latanoprost and brimonidine treatment IOP was statistically lower (16.2±3.2) than LTFC (18.0±2.8 mmHg). However, the reduced IOP from untreated baseline was not statistically different at each time point and for the diurnal curve for each therapy (P<0.05). Safety was similar between groups for both solicited and unsolicited side effects (P>0.05).
Conclusion This study suggests that LTFC and concomitant therapy of brimonidine and latanoprost provide statistically similar diurnal IOP reduction from an untreated baseline.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Stewart WC, Stewart JA, Day D, Sharpe ED . Efficacy and safety of latanoprost/timolol maleate fixed combination versus timolol maleate and brimonidine given twice daily. Acta Ophthalmol Scand 2003; 81: 242–246.
Book SA Essentials of Statistics. McGraw Hill Book Company: New York, 1978, pp 117–122, 205–215.
Mundorf TK, Cate EA, Sine CS, Otero DW, Stewart JA, Stewart WC . The safety and efficacy of switching timolol maleate 0.5% solution to timolol hemihydrate 0.5% solution given twice daily. J Ocular Pharm Therap 1998; 14: 129–135.
Duff GR . A double-masked crossover study comparing the effects of carteolol 1% and 2% on intraocular pressure. Acta Ophthalmol 1987; 65: 618–621.
Stewart WC, Sharpe ED, Harbin Jr TS, Pastor SA, Day DG, Holmes KT et al. Brimonidine 0.2% versus dorzolamide 2% each given three times daily to reduce the intraocular pressure. Am J Ophthalmol 2000; 129: 723–727.
Stewart WC, Day DG, Sharpe ED, Dubiner HB, Holmes KT, Stewart JA . Efficacy and safety of timolol solution once daily vs timolol gel added to latanoprost. Am J Ophthalmol 1999; 128: 692–696.
Siegel S Nonparametric Statistics for the Behavioral Science. Series in Psychology. McGraw Hill Book Company,. New York, 1956, pp 63–67.
Pfeiffer N, and the German Latanoprost Fixed Combination Study Group. A comparison of the fixed combination of latanoprost and timolol with IST individual components in patients with glaucoma or ocular hypertension. Invest Ophthalmol Vis Sci 2000; 41 (suppl): S754.
Higginbotham EJ, Feldman R, Stiles M, Dubiner H, Fixed Combination Investigative Group. Latanoprost and timolol combination therapy versus monotherapy. Arch Ophthalmol 2002; 120: 915–922.
Stewart WC, Day DG, Stewart JA, Schuhr J, Latham KE . The efficacy and safety of latanoprost 0.005% once daily versus brimonidine 0.2% twice daily in open-angle glaucoma or ocular hypertension. Am J Ophthalmol 2001; 131: 631–635.
Stewart WC, Holmes KT, Johnson MA . Washout periods for brimonidine 0.2% and latanoprost 0.005%. Am J Ophthalmol 2001; 131: 798–799.
Alm A, Stjernschantz J and the Scandinavian Latanoprost Study Group. Effects on intraocular pressure and side effects of 0.005% latanoprost applied once daily, evening or morning: a comparison with timolol. Ophthalmology 1995; 102: 1743–1752.
Camras CB and the United States Latanoprost Study Group. Comparison of latanoprost and timolol in patients with ocular hypertension and glaucoma: a six-month, masked, multicenter trial in the United States. Ophthalmology 1996; 103: 138–147.
Watson P, Stjernschantz J, and the Latanoprost Study Group. A six-month, randomized, double-masked study comparing latanoprost with timolol in open-angle glaucoma and ocular hypertension. Ophthalmology 1996; 103: 126–137.
Konstas AGP, Maltezos AC, Gandi S, Hudgins AC, Stewart WC . Comparison of the 24 hour intraocular pressure reduction with two dosing regimes of latanoprost and timolol in patients with open-angle glaucoma. Am J Ophthalmol 1999; 128: 15–20.
Konstas AGP, Nakos E, Tersis I, Lallos NA, Leech JN, Stewart WC . A comparison of once daily morning versus evening dosing of concomitant latanoprost/timolol. Am J Ophthalmol 2002; 133: 753–757.
Author information
Authors and Affiliations
Corresponding author
Additional information
This study was sponsored by Pfizer. The authors have no proprietary interest in any of the product or companies listed.
Rights and permissions
About this article
Cite this article
Stewart, W., Stewart, J., Day, D. et al. Efficacy and safety of the latanoprost/timolol maleate fixed combination vs concomitant brimonidine and latanoprost therapy. Eye 18, 990–995 (2004). https://doi.org/10.1038/sj.eye.6701375
Received:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/sj.eye.6701375
Keywords
This article is cited by
-
Repurposing Ophthalmologic Timolol for Dermatologic Use: Caveats and Historical Review of Adverse Events
American Journal of Clinical Dermatology (2021)
-
Efficacy and safety of fixed combinations of latanoprost/timolol and dorzolamide/timolol in open-angle glaucoma or ocular hypertension
Eye (2010)
-
Reporting of noninferiority and equivalence randomized trials for major prostaglandins: A systematic survey of the ophthalmology literature
Trials (2008)