Abstract
Purpose
To compare the efficacy and safety of brimonidine 0.2% vs unoprostone 0.15%, both added to timolol maleate 0.5% each given twice daily.
Methods
In this prospective, multi-centred, double-masked, crossover comparison, patients were randomized to one treatment group for a 6-week treatment period, and then crossed over to the opposite treatment. Measurements were performed at 0800, 1000, 1600, 1800, and 2000 h at baseline and at the end of each treatment period.
Results
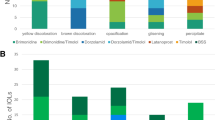
In all, 33 patients entered this trial and 29 completed. The baseline trough intraocular pressure (IOP) was 23.3±2.4 and the diurnal curve IOP was 22.0±1.3 mmHg. For the brimonidine and timolol maleate treatment group, the trough IOP was 21.6±3.3 and the diurnal curve IOP was 19.8±2.1 mmHg, while the timolol and unoprostone treatment showed a trough IOP of 20.9±3.8 and a diurnal curve IOP of 19.3±2.4 mmHg. There was no significant difference between treatment groups at any time point for the diurnal curve, or in the reduction from baseline (P>0.05). Both treatments failed to statistically reduce the IOP from baseline at 1800 h. There was no difference between treatment groups regarding ocular and systemic unsolicited adverse events, but patients admitted to more dryness (P=0.02) and burning upon instillation (P<0.0001) with unoprostone by survey.
Conclusion
Brimonidine 0.2% or unoprostone 0.15% added to timolol maleate 0.5% provide similar efficacy and safety throughout the daytime diurnal curve.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Stewart WC . Clinical Practice of Glaucoma. SLACK Inc.: Thorofare, 1990, pp 149–183.
Shields MB . Textbook of Glaucoma, 2nd ed. Williams & Wilkins: Baltimore, 1987, pp 449–455, 466–469.
Day DG, Schacknow PN, Wand M, Sharpe ED, Stewart JA, Leech J et al. Timolol 0.5%/dorzolamide 2% fixed combination versus timolol maleate 0.5% and unoprostone 0.15% given twice daily to patients with primary open-angle glaucoma or ocular hypertension. Am J Ophthalmol 2003; 135: 138–143.
Stewart WC, Stewart JA, Day DG, Leech J . The safety and efficacy of unoprostone 0.15% vs brimonidine 0.2%. Acta Opthalmol Scand 2004; in press.
Book SA . Essentials of Statistics. McGraw Hill Book Company: New York, 1978, pp 117–122, 205–215.
Mundorf TK, Cate EA, Sine CS, Otero DW, Stewart JA, Stewart WC . The safety and efficacy of switching timolol maleate 0.5% solution to timolol hemihydrate 0.5% solution given twice daily. J Ocul Pharmacol Ther 1998; 14: 129–135.
Duff GR . A double-masked crossover study comparing the effects of carteolol 1% and 2% on intraocular pressure. Acta Ophthalmol 1987; 65: 618–621.
Konstas AGP, Lake S, Maltezos AC, Holmes KT, Stewart WC . 24 hour intraocular pressure reduction with latanoprost compared to pilocarpine as third line therapy in exfoliation glaucoma. Eye 2001; 15: 59–62.
Stewart WC, Day DG, Stewart JA, Schuhr J, Latham KE . The efficacy and safety of latanoprost 0.005% once daily versus brimonidine 0.2% twice daily in open-angle glaucoma or ocular hypertension. Am J Ophthalmol 2001; 131: 631–635.
Siegel S . Nonparametric Statistics for the Behavioral Sciences. McGraw Hill Book Company: New York, 1956, pp 63–67.
Katz LJ . The Brimonidine Study Group. Brimonidine tartrate 0.2% twice daily vs timolol 0.5% twice daily: 1-year results in glaucoma patients. Am J Ophthalmol 1999; 127: 20–26.
Schuman JS . Clinical experience with brimonidine 0.2% and timolol 0.5% in glaucoma and ocular hypertension. Surv Ophthalmol 1996; 41: S27–S37.
Walters TR . Development and use of brimonidine in treating acute and chronic elevations of intraocular pressure: a review of safety, efficacy, dose response, and dosing studies. Surv Ophthalmol 1996; 41: S19–S26.
LeBlanc RP . Twelve-month results of an ongoing randomized trial comparing brimonidine tartrate 0.2% and timolol 0.5% given twice daily in patients with glaucoma or ocular hypertension. Brimonidine Study Group 2. Ophthalmology 1998; 105: 1960–1967.
Stewart WC . New perspectives in the medical treatment of glaucoma. Curr Opin 1999; 127: 142–147.
Hutzelmann J, Owens S, Shedden A, Adamsons I, Vargas E . Comparison of the safety and efficacy of the fixed combination of dorzolamide/timolol and the concomitant administration of dorzolamide and timolol: a clinical equivalence study. International Clinical Equivalence Study Group. Br J Ophthalmol 1998; 82: 1249–1253.
Sakurai M, Araie M, Oshika T, Mori M, Masuda K, Ueno R et al. Effects of topical application of UF-021, a novel prostaglandin derivative, on aqueous humor dynamics in normal human eyes. Jpn J Ophthalmol 1991; 35: 156–165.
Stewart WC, Mundorf T, Haque R, Brown A, Kapik B, Shams N . Comparison of the IOP-lowering efficacy and safety of the docosanoid unoprostone 0.15% versus timolol maleate 0.5% dosed twice daily for 6 months in patients with primary open-angle glaucoma or ocular hypertension. Invest Ophthalmol Vis Sci 2001; 42: S557.
Susanna Jr R, Giampani Jr J, Borges AS, Vessani RM, Jordao ML . A double-masked, randomized clinical trial comparing latanoprost with unoprostone in patients with open-angle glaucoma or ocular hypertension. Ophthalmology 2001; 108: 259–263.
Aung T, Chew PTK, Yip CC, Chan YH, See JLS, Khng CG et al. A randomized double-masked crossover study comparing latanoprost 0.005% with unoprostone 0.12% in patients with primary open-angle glaucoma and ocular hypertension. Am J Ophthalmol 2001; 131: 636–642.
Nordmann JP, Mertz B, Yannoulis NC, Schwenniger C, Kapik B, Shams N . A double-masked randomized comparison of the efficacy and safety of unoprostone with timolol and betaxolol in patients with primary open-angle glaucoma including pseudoexfoliation glaucoma or ocular hypertension. Six month data. Am J Ophthalmol 2002; 133: 1–10.
Stewart WC, Stewart JA, Day DG, Sharpe ED . Efficacy and safety of timolol maleat/ketanoprost fixed combination vs timolol maleate and brimonidine given twice daily. Acta Ophthalmol Scand 2003; 81: 242–246.
Stewart WC, Stewart JA, Kapik BM . The effect of unoprostone isopropylate 0.12% vs timolol maleate 0.5% on the diurnal IOP. J Glaucoma 1998; 6: 388–397.
Higginbotham EJ, Feldman R, Stiles M, Dubiner H . Fixed Combination Investigative Group. Latanoprost and timolol combination therapy vs monotherapy: one-year randomized trial. Arch Ophthalmol 2002; 120: 915–922.
Pfeiffer N, European Latanoprost Fixed Combination Study Group. A comparison of the fixed combination of latanoprost and timolol with its individual components. Graefes Arch Clin Exp Ophthalmol 2002; 240: 893–899.
Boyle JE, Ghosh K, Gieser DK, Adamsons IA . A randomized trial comparing the dorzolamide–timolol combination given twice daily to monotherapy with timolol and dorzolamide. Dorzolamide–Timolol Study Group. Ophthalmology 1998; 105: 1945–1951.
Clineschmidt CM, Williams RD, Snyder E, Adamsons IA . A randomized trial in patients inadequately controlled with timolol alone comparing the dorzolamide–timolol combination to monotherapy with timolol or dorzolamide. Dorzolamide–Timolol Combination Study Group. Ophthalmology 1998; 105: 1952–1959.
Hommer A, Kapik B, Shams N . The Unoprostone Adjunctive Therapy Study Group. Unoprostone as adjunctive therapy to timolol: a double masked randomised study versus brimonidine and dorzolamide. Br J Ophthalmol 2003; 87: 592–598.
Author information
Authors and Affiliations
Corresponding author
Additional information
This study was supported by Novartis Ophthalmics, Atlanta, GA, USA.
Rights and permissions
About this article
Cite this article
Sharpe, E., Henry, C., Mundorf, T. et al. Brimonidine 0.2% vs unoprostone 0.15% both added to timolol maleate 0.5% given twice daily to patients with primary open-angle glaucoma or ocular hypertension. Eye 19, 35–40 (2005). https://doi.org/10.1038/sj.eye.6701392
Received:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/sj.eye.6701392