Abstract
Aim
To identify the relative incidence and profile of adverse drug reaction (ADR) to various topical ocular hypotensives in a hospital setting.
Methods
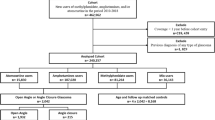
All the patients presenting in outpatients clinic and accident and emergency with an ADR to topical hypotensive agent from August 2000 to January 2001 were included in the study. Details regarding the type date of commencing the treatment, the date of developing ADR, time to resolution of the ADR were noted.
Results
Over the period of 6 months, 66 patients presented with 73 ADRs. Brimonidine was the most frequent offending agent. In total, 23 (34.8%) presented with ADR after being commenced on treatment for more than 12 months. In all, 16 (24%) patients had IOP>21 on presentation, eight (12%) patients underwent filtration surgery following the development of ADR.
Conclusion
Adverse drug reaction to ocular hypotensive agents is not uncommon and can have a major impact on glaucoma management. Delayed presentation and association with raised intraocular pressure presentation emphasise the need for effective patient education to encourage prompt reporting of ADR.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Bateman DN, Clark R, Azuara-Blanco A, Bain M, Forrest J . The effects of new topical treatments on management of glaucoma in Scotland: an examination of ophthalmological health care. Br J Ophthalmol 2002; 86: 551–554.
Bateman DN, Clark R, Azuara-Blanco, Bain M, Forrest J . The impact of new drugs on management of glaucoma in Scotland: observational study. BMJ 2001; 323: 1401–1402.
Whittaker KW, Gillow JT, Cunliffe IA . Is the role of trabeculectomy in glaucoma management changing? Eye 2001; 15: 449–452.
Schuman JS, Horwitz B, Choplin NT, David R, Albracht D, Chen K et al. A 1-year study of brimonidine twice daily in glaucoma and ocular hypertension. Arch Ophthalmol 1997; 115: 847–852.
Jay Katz L, The Brimonidine Study Group. Brimonidine tartrate 0.2% twice daily vs timolol 0.5% twice daily: 1-year results in glaucoma patients. Am J Ophthalmol 1999; 127: 20–26.
Blondeau P, Rousseau JA . Allergic reactions to brimonidine in patients treated for glaucoma. Can J Ophthalmol 2002; 37: 21–26.
LeBlanc RP, The Brimonidine Study Group 2. Twelve-month results of an ongoing randomised trial comparing brimonidine tartarate 0.2% and timolol 0.5% given twice daily in patients with glaucoma and ocular hypertension. Opthalmology 1998; 105: 1960–1967.
Watts P, Hawksworth N . Delayed hypersensitivity to brimonidine tartrate 0.2% associated with higher intraocular pressure. Eye 2002; 16: 132–135.
Garcia F, Blanco J, Juste S, Garces MM, Alonso L, Marcos ML et al. Contact dermatitis due to levobunolol in eyedrops. Contact Dermatitis 1997; 36: 230.
Nino M, Suppa F, Ayala F, Balato N . Allergic contact dermatitis due to the beta-lockers befunolol in eyedrops, with cross-sensitivity to carteolol. Contact Dermatitis 2001; 44: 369.
Delaney YM, Salmon JF, Mossa F, Gee B, Beehne K, Powell S . Periorbital dermatitis as a side effect of topical dorzolamide. Br J Ophthalmol 2002; 86: 378–380.
Koch P . Allergic contact dermatitis due to timolol and levobunolol in eye drops, with no cross sensitivity to other ophthalmic beta-blockers. Contact Dermatitis 1995; 33: 140.
Corazza M, Virgili A, Mantovani L, Taddei Masieri L . Allergic contact dermatitis from cross-reacting beta-blocking agents. Contact Dermatitis 1993; 28: 188–189.
Pisella PJ, Pouliquen P, Baudouin C . Prevalence of ocular symptoms and signs with preserved and preservative free glaucoma medication. Br J Ophthalmol 2002; 86: 418–423.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bhatt, R., Whittaker, K., Appaswamy, S. et al. Prospective survey of adverse reactions to topical antiglaucoma medications in a hospital population. Eye 19, 392–395 (2005). https://doi.org/10.1038/sj.eye.6701515
Received:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/sj.eye.6701515