Abstract
Purpose
To study the incidence, clinical findings, and tumour characteristics of posterior uveal melanoma in Western Norway, and to report the results of a consistent treatment strategy (I-125 brachytherapy or primary enucleation) over a 13-year period.
Methods
The clinical records of all patients with posterior uveal melanoma referred between January 1993 and December 2005 were reviewed. Clinical data, radiation parameters, visual outcome, and mortality were analysed in a dedicated database.
Results
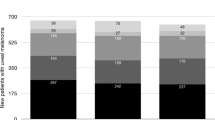
The study included 111 consecutive patients. The annual age-adjusted incidence (per million population) of posterior uveal melanoma was 8.5 for women and 8.9 for men. Fifty-six patients underwent I-125 brachytherapy, 52 were enucleated, and three received no treatment. The median follow-up time was 36 months (mean, 52 months; range, 2 months to 13 years). In the brachytherapy group, two eyes were enucleated owing to tumour recurrence and two because of neovascular glaucoma. A visual acuity of 0.1 or better, present in 87% of the patients before brachytherapy, was retained in 40% after a median follow-up of 61 months. After brachytherapy, the 5- and 10-year melanoma-specific mortality rates were 13.4 and 23.8%, respectively. The corresponding mortality rates for patients treated with primary enucleation were 49.5 and 49.5%.
Conclusion
After brachytherapy, many patients lost useful vision due to radiation-induced complications. The probability of retaining the eye was high and only two patients experienced recurrent tumour growth. The mortality rates compare well with published series, and the differences in tumour size explain the difference in mortality between the two treatment groups.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Singh AD, Topham A . Survival rates with uveal melanoma in the United States: 1973–1997. Ophthalmology 2003; 110: 962–965.
Collaborative Ocular Melanoma Study Group. The COMS randomized trial of iodine 125 brachytherapy for choroidal melanoma, III: initial mortality findings. COMS Report No. 18. Arch Ophthalmol 2001; 119: 969–982.
Shields JA, Shields CL . Atlas of intraocular tumors. Lippincott Williams & Wilkins: Philadelphia, 1999.
UICC, International Union against Cancer. TNM Atlas. Illustrated guide to the TNM/pTNM classification of malignant tumours, 3rd ed., 2nd revision. Springer-Verlag: Berlin, 1992.
Collaborative Ocular Melanoma Study Group. Comparison of clinical, echographic, and histopathological measurements from eyes with medium-sized choroidal melanoma in the collaborative ocular melanoma study. COMS Report No.21. Arch Ophthalmol 2003; 121: 1163–1171.
Astrahan MA, Luxton G, Pu Q, Petrovich Z . Conformal episcleral plaque therapy. Int J Radiat Oncol Biol Phys 1997; 39: 505–519.
Knutsen S, Hafslund R, Monge OR, Valen H, Muren LP, Rekstad BL et al. Dosimetric verification of a dedicated 3D treatment planning system for episcleral plaque therapy. Int J Radiat Oncol Biol Phys 2001; 51: 1159–1166.
Krohn J, Seland JH, Monge OR, Rekstad BL . Transillumination for accurate placement of radioactive plaques in brachytherapy of choroidal melanoma. Am J Ophthalmol 2001; 132: 418–419.
Gündüz K, Shields CL, Shields JA, Cater J, Freire JE, Brady LW . Radiation complications and tumor control after plaque radiotherapy of choroidal melanoma with macular involvement. Am J Ophthalmol 1999; 127: 579–589.
Callender GR . Malignant melanotic tumors of the eye: a study of histologic types in 111 cases. Trans Am Acad Ophthalmol Otolaryngol 1931; 36: 131–142.
Puusaari I, Heikkonen J, Kivelä T . Ocular complications after iodine brachytherapy for large uveal melanomas. Ophthalmology 2004; 111: 1768–1777.
Shields CL, Naseripour M, Cater J, Shields JA, Demirci H, Youseff A et al. Plaque radiotherapy for large posterior uveal melanomas (⩾8-mm thick) in 354 consecutive patients. Ophthalmology 2002; 109: 1838–1849.
Nag S, Quivey JM, Earle JD, Followill D, Fontanesi J, Finger PT . The American Brachytherapy Society recommendations for brachytherapy of uveal melanomas. Int J Radiat Oncol Biol Phys 2003; 56: 544–555.
Lommatzsch PK, Alberti W, Lommatzsch R, Rohrwacher F . Radiation effects on the optic nerve observed after brachytherapy of choroidal melanomas with 106Ru/106Rh plaques. Graefes Arch Clin Exp Ophthalmol 1994; 232: 482–487.
Gragoudas ES, Li W, Lane AM, Munzenrider J, Egan KM . Risk factors for radiation maculopathy and papillopathy after intraocular irradiation. Ophthalmology 1999; 106: 1571–1577.
Shields CL, Shields JA, Karlsson U, Markoe AM, Brady LW . Reasons for enucleation after plaque radiotherapy for posterior uveal melanoma. Ophthalmology 1989; 96: 919–924.
Char DH, Quivey JM, Castro JR, Kroll S, Phillips T . Helium ions versus iodine 125 brachytherapy in the management of uveal melanoma. A prospective, randomized, dynamically balanced trial. Ophthalmology 1993; 100: 1547–1554.
Packer S, Stoller S, Lesser ML, Mandel FS, Finger PT . Long-term results of iodine 125 irradiation of uveal melanoma. Ophthalmology 1992; 99: 767–773.
Wilson MW, Hungerford JL . Comparison of episcleral plaque and proton beam radiation therapy for the treatment of choroidal melanoma. Ophthalmology 1999; 106: 1579–1587.
Karlsson UL, Augsburger JJ, Shields JA, Markoe AM, Brady LW, Woodleigh R . Recurrence of posterior uveal melanoma after 60Co episcleral plaque therapy. Ophthalmology 1989; 96: 382–388.
Vrabec TR, Augsburger JJ, Gamel JW, Brady LW, Hernandez C, Woodleigh R . Impact of local tumor relapse on patient survival after cobalt 60 plaque radiotherapy. Ophthalmology 1991; 98: 984–988.
Seregard S, Kock E . Prognostic indicators following enucleation for posterior uveal melanoma. A multivariate analysis of long-term survival with minimized loss to follow-up. Acta Ophthalmol Scand 1995; 73: 340–344.
Cohen VML, Carter MJ, Kemeny A, Radatz M, Rennie IG . Metastasis-free survival following treatment for uveal melanoma with either stereotactic radiosurgery or enucleation. Acta Ophthalmol Scand 2003; 81: 383–388.
Isager P, Ehlers N, Overgaard J . Prognostic factors for survival after enucleation for choroidal and ciliary body melanomas. Acta Ophthalmol Scand 2004; 82: 517–525.
Shammas HF, Blodi FC . Prognostic factors in choroidal and ciliary body melanomas. Arch Ophthalmol 1977; 95: 63–69.
Bergman L, Seregard S, Nilsson B, Ringborg U, Lundell G, Ragnarsson-Olding B . Incidence of uveal melanoma in Sweden from 1960 to 1998. Invest Ophthalmol Vis Sci 2002; 43: 2579–2583.
Isager P, Østerlind A, Engholm G, Heegaard S, Lindegaard J, Overgaard J et al. Uveal and conjunctival malignant melanoma in Denmark, 1943–97: Incidence and validation study. Ophthalmic Epidemiol 2005; 12: 223–232.
Singh AD, Topham A . Incidence of uveal melanoma in the United States: 1973–1997. Ophthalmology 2003; 110: 956–961.
Mork T . Malignant neoplasms of the eye in Norway. Incidence, treatment and prognosis. Acta Ophthalmol (Copenh) 1961; 39: 824–831.
Acknowledgements
The study was supported by grants from The Norwegian Cancer Society.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Krohn, J., Monge, O., Skorpen, T. et al. Posterior uveal melanoma treated with I-125 brachytherapy or primary enucleation. Eye 22, 1398–1403 (2008). https://doi.org/10.1038/sj.eye.6702911
Received:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/sj.eye.6702911
Keywords
This article is cited by
-
Topography and clinical features of iris melanoma
BMC Ophthalmology (2022)
-
Ocular complications following I-125 brachytherapy for choroidal melanoma
Eye (2009)
-
Initial results of fractionated CyberKnife radiosurgery for uveal melanoma
Journal of Neuro-Oncology (2009)