Abstract
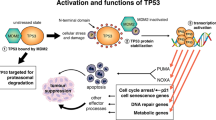
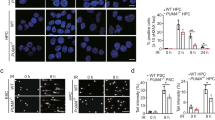
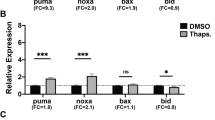
The ability of p53 to induce apoptosis in cells with damaged DNA is thought to contribute greatly to its tumour suppressor function. P53 has been proposed to induce apoptosis via numerous transcriptional targets or even by direct cytoplasmic action. Two transcriptional targets shown to mediate its apoptotic role in several cell types encode Noxa and Puma, BH3-only members of the Bcl-2 family. To test if their functions in p53-dependent apoptosis overlap, we generated mice lacking both. These mice develop normally and no tumours have yet arisen. In embryonic fibroblasts, the absence of both Noxa and Puma prevented induction of apoptosis by etoposide. Moreover, following whole body γ-irradiation, the loss of both proteins protected thymocytes better than loss of Puma alone. Indeed, their combined deficiency protected thymocytes as strongly as loss of p53 itself. These results indicate that, at least in fibroblasts and thymocytes, p53-induced apoptosis proceeds principally via Noxa and Puma, with Puma having the predominant role in diverse cell types. The absence of tumours in the mice suggests that tumour suppression by p53 requires functions in addition to induction of apoptosis.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Abbreviations
- BH:
-
Bcl-2-homology
- DKO:
-
double knockout
- DP:
-
double-positive
- MEF:
-
mouse embryonic fibroblasts
- PMA:
-
phorbol 12-myristate 13-acetate
- TUNEL:
-
terminal deoxynucleotidyl transferase-mediated dUTP nick end labelling
References
Vousden KH, Lu X . Live or let die: the cell's response to p53. Nat Rev Cancer 2002; 2: 594–604.
Donehower LA, Harvey M, Slagle BL, McArthur MJ, Montgomery CAJ, Butel JS et al. Mice deficient for p53 are developmentally normal but are susceptible to spontaneous tumours. Nature 1992; 356: 215–221.
Jacks T, Remington L, Williams BO, Schmitt EM, Halachmi S, Bronson RT et al. Tumor spectrum analysis in p53-mutant mice. Curr Biol 1994; 4: 1–7.
Wang Y, Szekely L, Okan I, Klein G, Wiman KG . Wild-type p53-triggered apoptosis is inhibited by bcl-2 in a v-myc-induced T-cell lymphoma line. Oncogene 1993; 8: 3427–3431.
Strasser A, Harris AW, Cory S . Bcl-2 transgene inhibits T cell death and perturbs thymic self-censorship. Cell 1991; 67: 889–899.
Strasser A, Harris AW, Jacks T, Cory S . DNA damage can induce apoptosis in proliferating lymphoid cells via p53-independent mechanisms inhibitable by Bcl-2. Cell 1994; 79: 329–339.
Mihara M, Erster S, Zaika A, Petrenko O, Chittenden T, Pancoska P et al. p53 has a direct apoptogenic role at the mitochondria. Mol Cell 2003; 11: 577–590.
Erster S, Mihara M, Kim RH, Petrenko O, Moll UM . In vivo mitochondrial p53 translocation triggers a rapid first wave of cell death in response to DNA damage that can precede p53 target gene activation. Mol Cell Biol 2004; 24: 6728–6741.
Leu JI, Dumont P, Hafey M, Murphy ME, George DL . Mitochondrial p53 activates Bak and causes disruption of a Bak-Mcl1 complex. Nat Cell Biol 2004; 6: 443–450.
Chipuk JE, Kuwana T, Bouchier-Hayes L, Droin NM, Newmeyer DD, Schuler M et al. Direct activation of Bax by p53 mediates mitochondrial membrane permeabilization and apoptosis. Science 2004; 303: 1010–1014.
Chipuk JE, Bouchier-Hayes L, Kuwana T, Newmeyer DD, Green DR . PUMA couples the nuclear and cytoplasmic proapoptotic function of p53. Science 2005; 309: 1732–1735.
Danial NN, Korsmeyer SJ . Cell death: critical control points. Cell 2004; 116: 205–219.
Wei MC, Zong WX, Cheng EH, Lindsten T, Panoutsakopoulou V, Ross AJ et al. Proapoptotic BAX and BAK: a requisite gateway to mitochondrial dysfunction and death. Science 2001; 292: 727–730.
Huang DCS, Strasser A . BH3-only proteins – essential initiators of apoptotic cell death. Cell 2000; 103: 839–842.
Oda E, Ohki R, Murasawa H, Nemoto J, Shibue T, Yamashita T et al. Noxa, a BH3-only member of the bcl-2 family and candidate mediator of p53-induced apoptosis. Science 2000; 288: 1053–1058.
Nakano K, Vousden KH . PUMA a novel proapoptotic gene is induced by p53. Molecular Cell 2001; 7: 683–694.
Yu J, Zhang L, Hwang PM, Kinzler KW, Vogelstein B . PUMA induces the rapid apoptosis of colorectal cancer cells. Molecular Cell 2001; 7: 673–682.
Han J, Flemington C, Houghton AB, Gu Z, Zambetti GP, Lutz RJ et al. Expression of bbc3, a pro-apoptotic BH3-only gene, is regulated by diverse cell death and survival signals. Proc Natl Acad Sci USA 2001; 98: 11318–11323.
Villunger A, Michalak EM, Coultas L, Müllauer F, Böck G, Ausserlechner MJ et al. p53- and drug-induced apoptotic responses mediated by BH3-only proteins Puma and Noxa. Science 2003; 302: 1036–1038.
Jeffers JR, Parganas E, Lee Y, Yang C, Wang J, Brennan J et al. Puma is an essential mediator of p53-dependent and -independent apoptotic pathways. Cancer Cell 2003; 4: 321–328.
Erlacher M, Michalak EM, Kelly PN, Labi V, Niederegger H, Coultas L et al. BH3-only proteins Puma and Bim are rate-limiting for {gamma} -radiation and glucocorticoid-induced apoptosis of lymphoid cells in vivo. Blood 2005; 106: 4131–4138.
Shibue T, Takeda K, Oda E, Tanaka H, Murasawa H, Takaoka A et al. Integral role of Noxa in p53-mediated apoptotic response. Genes Dev 2003; 17: 2233–2238.
Lowe SW, Schmitt EM, Smith SW, Osborne BA, Jacks T . p53 is required for radiation-induced apoptosis in mouse thymocytes. Nature 1993; 362: 847–849.
Clarke AR, Purdie CA, Harrison DJ, Morris RG, Bird CC, Hooper ML et al. Thymocyte apoptosis induced by p53-dependent and independent pathways. Nature 1993; 362: 849–852.
Bouillet P, Metcalf D, Huang DCS, Tarlinton DM, Kay TWH, Köntgen F et al. Proapoptotic Bcl-2 relative Bim required for certain apoptotic responses, leukocyte homeostasis, and to preclude autoimmunity. Science 1999; 286: 1735–1738.
Lowe SW, Ruley HE . Stabilization of the p53 tumor suppressor is induced by adenovirus 5 E1A and accompanies apoptosis. Genes and Development 1993; 7: 535–545.
Sellins KS, Cohen JJ . Gene induction by g-irradiation leads to DNA fragmentation in lymphocytes. J Immunol 1987; 139: 3199–3206.
Chen L, Willis SN, Wei A, Smith BJ, Fletcher JI, Hinds MG et al. Differential targeting of pro-survival Bcl-2 proteins by their BH3-only ligands allows complementary apoptotic function. Mol Cell 2005; 17: 393–403.
Kuwana T, Bouchier-Hayes L, Chipuk JE, Bonzon C, Sullivan BA, Green DR et al. BH3 Domains of BH3-Only Proteins Differentially Regulate Bax-Mediated Mitochondrial Membrane Permeabilization Both Directly and Indirectly. Mol Cell 2005; 17: 525–535.
MacFarlane M, Jones NA, Dive C, Cohen GM . DNA-damaging agents induce both p53-dependent and p53-independent apoptosis in immature thymocytes. Mol Pharmacol 1996; 50: 900–911.
Erlacher M, Laabi V, Manzl C, Bock G, Tzankov A, Haecker G et al. Puma cooperates with Bim, the rate-limiting BH3-only protein in cell death during lymphocyte development, in apoptosis induction. J Exp Med 2006; 203: 2939–2951.
Wang P, Yu J, Zhang L . The nuclear function of p53 is required for PUMA-mediated apoptosis induced by DNA damage. Proc Natl Acad Sci USA 2007; 104: 4054–4059.
Callus BA, Ekert PG, Heraud JE, Jabbour AM, Kotevski A, Vince JE et al. Cytoplasmic p53 is not required for PUMA-induced apoptosis. Cell Death Differ 2008; 15: 213–221.
Uren RT, Dewson G, Chen L, Coyne SC, Huang DCS, Adams JM et al. Mitochondrial permeabilization relies on BH3 ligands engaging multiple pro-survival Bcl-2 relatives, not Bak. J Cell Biol 2007; 177: 277–287.
Willis SN, Fletcher JI, Kaufmann T, van Delft MF, Chen L, Czabotar PE et al. Apoptosis initiated when BH3 ligands engage multiple Bcl-2 homologs, not Bax or Bak. Science 2007; 315: 856–859.
Ming L, Wang P, Bank A, Yu J, Zhang L . PUMA dissociates Bax and Bcl-XL to induce apoptosis in colon cancer cells. J Biol Chem 2006; 281: 16034–16042.
Martin-Caballero J, Flores JM, Garcia-Palencia P, Serrano M . Tumor susceptibility of p21(Waf1/Cip1)-deficient mice. Cancer Res 2001; 61: 6234–6238.
Metz T, Harris AW, Adams JM . Absence of p53 allows direct immortalization of hematopoietic cells by the myc and raf oncogenes. Cell 1995; 82: 29–36.
Matoba S, Kang JG, Patino WD, Wragg A, Boehm M, Gavrilova O et al. p53 regulates mitochondrial respiration. Science 2006; 312: 1650–1653.
Mestre-Escorihuela C, Rubio-Moscardo F, Richter JA, Siebert R, Climent J, Fresquet V et al. Homozygous deletions localize novel tumor suppressor genes in B-cell lymphomas. Blood 2007; 109: 271–280.
Hemann MT, Zilfou JT, Zhao Z, Burgess DJ, Hannon GJ, Lowe SW . Suppression of tumorigenesis by the p53 target PUMA. Proc Natl Acad Sci USA 2004; 101: 9333–9338.
Egle A, Harris AW, Bouillet P, Cory S . Bim is a suppressor of Myc-induced mouse B cell leukemia. Proc Natl Acad Sci USA 2004; 101: 6164–6169.
Christophorou MA, Ringshausen I, Finch AJ, Swigart LB, Evan GI . The pathological response to DNA damage does not contribute to p53-mediated tumour suppression. Nature 2006; 443: 214–217.
Acknowledgements
We thank N Iannarella, G Siciliano and A Naughton for animal care; Dr. F Battye, C Tarlinton, V Milovac, J Garbe and C Young for cell sorting; T Nikolaou and G Thomas for γ-irradiation; J Corbin for haematological analysis; Dr. S Mihajlovic, A Hasanein, K Weston for histological sections; Drs. M Schuler, S Lowe and G Hannon for expression vectors; and Professor S Cory and Drs. A Harris, D Huang, H Puthalakath, P Bouillet, L O’Reilly, M Pellegrini, L Coultas and C Scott for their input and interesting discussions. This work was supported by fellowships and grants from the NHMRC (Canberra; program #257502), the Leukemia and Lymphoma Society (SCOR grant #7015), the NIH (CA043540-18 and CA80188-6), the JDRF/NHMRC, the Leukemia Research Foundation (LRF), the Cancer Council Victoria Postdoctoral Cancer Research Fellowship and the Austrian Science Fund (FWF).
Author information
Authors and Affiliations
Corresponding author
Additional information
Edited by KH Vousden
Supplementary Information accompanies the paper on Cell Death and Differentiation website (http://www.nature.com/cdd)
Rights and permissions
About this article
Cite this article
Michalak, E., Villunger, A., Adams, J. et al. In several cell types tumour suppressor p53 induces apoptosis largely via Puma but Noxa can contribute. Cell Death Differ 15, 1019–1029 (2008). https://doi.org/10.1038/cdd.2008.16
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/cdd.2008.16
Keywords
This article is cited by
-
Combined absence of TRP53 target genes ZMAT3, PUMA and p21 cause a high incidence of cancer in mice
Cell Death & Differentiation (2024)
-
Tissue specificity and spatio-temporal dynamics of the p53 transcriptional program
Cell Death & Differentiation (2023)
-
Phototoxicity-free blue light for enhancing therapeutic angiogenic efficacy of stem cells
Cell Biology and Toxicology (2023)
-
BCL-2 protein family: attractive targets for cancer therapy
Apoptosis (2023)
-
Of the many cellular responses activated by TP53, which ones are critical for tumour suppression?
Cell Death & Differentiation (2022)