Abstract
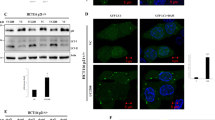
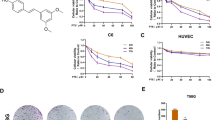
Loss of the tumor suppressor phosphatase and tensin homolog (PTEN) has frequently been observed in human gliomas, conferring AKT activation and resistance to ionizing radiation (IR) and drug treatments. Recent reports have shown that PTEN loss or AKT activation induces premature senescence, but many details regarding this effect remain obscure. In this study, we tested whether the status of PTEN determined fate of the cell by examining PTEN-deficient U87, U251, and U373, and PTEN-proficient LN18 and LN428 glioma cells after exposure to IR. These cells exhibited different cellular responses, senescence or apoptosis, depending on the PTEN status. We further observed that PTEN-deficient U87 cells with high levels of both AKT activation and intracellular reactive oxygen species (ROS) underwent senescence, whereas PTEN-proficient LN18 cells entered apoptosis. ROS were indispensable for inducing senescence in PTEN-deficient cells, but not for apoptosis in PTEN-proficient cells. Furthermore, transfection with wild-type (wt) PTEN or AKT small interfering RNA induced a change from premature senescence to apoptosis and depletion of p53 or p21 prevented IR-induced premature senescence in U87 cells. Our data indicate that PTEN acts as a pivotal determinant of cell fate, regarding senescence and apoptosis in IR-exposed glioma cells. We conclude that premature senescence could have a compensatory role for apoptosis in the absence of the tumor suppressor PTEN through the AKT/ROS/p53/p21 signaling pathway.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Abbreviations
- BrdU:
-
bromodeoxyuridine
- H2O2:
-
hydrogen peroxide
- NAC:
-
N-acetyl-l-cysteine
- ROS:
-
reactive oxygen species
- RT:
-
radiotherapy
- SA-β-Gal:
-
senescence-associated β-galactosidase
- siRNA:
-
small interfering RNA
- wt:
-
wild-type
References
Hirose Y, Berger MS, Pieper RO . Abrogation of the Chk1-mediated G(2) checkpoint pathway potentiates temozolomide-induced toxicity in a p53-independent manner in human glioblastoma cells. Cancer Res 2001; 61: 5843–5849.
Ohgaki H, Dessen P, Jourde B, Horstmann S, Nishikawa T, Di Patre PL et al. Genetic pathways to glioblastoma: a population-based study. Cancer Res 2004; 64: 6892–6899.
Bleeker FE, Lamba S, Zanon C, van Tilborg AA, Leenstra S, Troost D et al. Absence of AKT1 mutations in glioblastoma. PLoS One 2009; 4: e5638.
Keniry M, Parsons R . The role of PTEN signaling perturbations in cancer and in targeted therapy. Oncogene 2008; 27: 5477–5485.
Nogueira V, Park Y, Chen CC, Xu PZ, Chen ML, Tonic I et al. Akt determines replicative senescence and oxidative or oncogenic premature senescence and sensitizes cells to oxidative apoptosis. Cancer Cell 2008; 14: 458–470.
Miyauchi H, Minamino T, Tateno K, Kunieda T, Toko H, Komuro I . Akt negatively regulates the in vitro lifespan of human endothelial cells via a p53/p21-dependent pathway. EMBO J 2004; 23: 212–220.
Braig M, Schmitt CA . Oncogene-induced senescence: putting the brakes on tumor development. Cancer Res 2006; 66: 2881–2884.
Collado M, Serrano M . Senescence in tumours: evidence from mice and humans. Nat Rev Cancer 2010; 10: 51–57.
Roninson IB . Tumor cell senescence in cancer treatment. Cancer Res 2003; 63: 2705–2715.
Hornsby PJ . Senescence as an anticancer mechanism. J Clin Oncol. 2007; 25: 1852–1857.
Wu CH, van Riggelen J, Yetil A, Fan AC, Bachireddy P, Felsher DW . Cellular senescence is an important mechanism of tumor regression upon c-Myc inactivation. Proc Natl Acad Sci USA 2007; 104: 13028–13033.
Ishii N, Maier D, Merlo A, Tada M, Sawamura Y, Diserens AC et al. Frequent co-alterations of TP53, p16/CDKN2A, p14ARF, PTEN tumor suppressor genes in human glioma cell lines. Brain Pathol 1999; 9: 469–479.
Dalton WB, Yu B, Yang VW . p53 suppresses structural chromosome instability after mitotic arrest in human cells. Oncogene 2010; 29: 1929–1940.
Tao Y, Leteur C, Yang C, Zhang P, Castedo M, Pierré A et al. Radiosensitization by Chir-124, a selective CHK1 inhibitor. Cell Cycle 2009; 8: 1196–1205.
Cai XM, Tao BB, Wang LY, Liang YL, Jin JW, Yang Y et al. Protein phosphatase activity of PTEN inhibited the invasion of glioma cells with epidermal growth factor receptor mutation type III expression. Int J Cancer 2005; 117: 905–912.
Schmitt CA . Cellular senescence and cancer treatment. Biochim Biophys Acta 2007; 1775: 5–20.
Xue W, Zender L, Miething C, Dickins RA, Hernando E, Krizhanovsky V et al. Senescence and tumour clearance is triggered by p53 restoration in murine liver carcinomas. Nature 2007; 445: 656–660.
Wu CH, van Riggelen J, Yetil A, Fan AC, Bachireddy P, Felsher DW . Cellular senescence is an important mechanism of tumor regression upon c-Myc inactivation. Proc Natl Acad Sci USA 2007; 104: 13028–13033.
Ventura A, Kirsch DG, McLaughlin ME, Tuveson DA, Grimm J, Lintault L et al. Restoration of p53 function leads to tumour regression in vivo. Nature 2007; 445: 661–665.
Byun HO, Han NK, Lee HJ, Kim KB, Ko YG, Yoon G et al. Cathepsin D and eukaryotic translation elongation factor 1 as promising markers of cellular senescence. Cancer Res 2009; 69: 4638–4647.
Mischel PS, Cloughesy TF . Targeted molecular therapy of GBM. Brain Pathol 2003; 13: 52–61.
Wang Y, Zhu S, Cloughesy TF, Liau LM, Mischel PS . p53 disruption profoundly alters the response of human glioblastoma cells to DNA topoisomerase I inhibition. Oncogene 2004; 23: 1283–1290.
Solomon DA, Kim JS, Jenkins S, Ressom H, Huang M, Coppa N et al. Identification of p18 INK4c as a tumor suppressor gene in glioblastoma multiforme. Cancer Res 2008; 68: 2564–2569.
Mischel PS, Shai R, Shi T, Horvath S, Lu KV, Choe G et al. Identification of molecular subtypes of glioblastoma by gene expression profiling. Oncogene 2003; 22: 2361–2373.
Minamino T, Miyauchi H, Tateno K, Kunieda T, Komuro I . Akt-induced cellular senescence: implication for human disease. Cell Cycle 2004; 3: 449–451.
Shiohara M, Gombart AF, Berman JD, Koike K, Komiyama A, Koeffler HP . Cytostatic effect of TNFα on cancer cells is independent of p21WAF1. Oncogene 1997; 15: 1605–1609.
Sagulenko V, Muth D, Sagulenko E, Paffhausen T, Schwab M, Westermann F . Cathepsin D protects human neuroblastoma cells from doxorubicin-induced cell death. Carcinogenesis 2008; 29: 1869–1877.
Chen Z, Trotman LC, Shaffer D, Lin HK, Dotan ZA, Niki M et al. Crucial role of p53-dependent cellular senescence in suppression of Pten-deficient tumorigenesis. Nature 2005; 436: 725–730.
Das M, Mukherjee SB, Shaha C . Hydrogen peroxide induces apoptosis-like death in Leishmania donovani promastigotes. J Cell Sci 2001; 114: 2461–2469.
Quillet-Mary A, Jaffrézou JP, Mansat V, Bordier C, Naval J, Laurent G . Implication of mitochondrial hydrogen peroxide generation in ceramide-induced apoptosis. J Biol Chem 1997; 272: 21388–21395.
Song YS, Lee BY, Hwang ES . Distinct ROS and biochemical profiles in cells undergoing DNA damage-induced senescence and apoptosis. Mech Ageing Dev 2005; 126: 580–590.
Derouet-Hümbert E, Drãgan CA, Hakki T, Bureik M . ROS production by adrenodoxin does not cause apoptosis in fission yeast. Apoptosis 2007; 12: 2135–2142.
Sun Y, St Clair DK, Xu Y, Crooks PA, St Clair WH . A NADPH oxidase-dependent redox signaling pathway mediates the selective radiosensitization effect of parthenolide in prostate cancer cells. Cancer Res 2010; 70: 2880–2890.
Suzuki M, Boothman DA . Stress-induced premature senescence (SIPS): influence of SIPS on radiotherapy. J Radiat Res 2008; 49: 105–112.
Dewey WC, Ling CC, Meyn RE . Radiation-induced apoptosis: relevance to radiotherapy. Int J Radiat Oncol Biol Phys 1995; 33: 781–796.
Ross GM . Induction of cell death by radiotherapy. Endocr Relat Cancer 1999; 6: 41–44.
Hendry JH, Potten CS, Merritt A . Apoptosis induced by high- and low-LET radiations. Radiat Environ Biophys 1995; 34: 59–62.
Nomura T, Kinuta M, Hongyo T, Nakajima H, Hatanaka T . Programmed cell death in whole body and organ systems by low dose radiation. J Radiat Res 1992; 33: 109–123.
Lee JJ, Lee JH, Ko YG, Hong SI, Lee JS . Prevention of premature senescence requires JNK regulation of Bcl-2 and reactive oxygen species. Oncogene 2010; 29: 561–575.
Dimri GP, Lee X, Basile G, Acosta M, Scott G, Roskelley C et al. A biomarker that identifies senescent human cells in culture and in aging skin in vivo. Proc Natl Acad Sci USA 1995; 92: 9363–9367.
Acknowledgements
This work was supported by the Nuclear Research & Development Program and Radiological Translational Research Program (RTR10-03) of the Korea Science and Engineering Foundation grant funded by the Korean government (MEST).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Edited by JA Cidlowski
Supplementary Information accompanies the paper on Cell Death and Differentiation website
Rights and permissions
About this article
Cite this article
Lee, JJ., Kim, B., Park, MJ. et al. PTEN status switches cell fate between premature senescence and apoptosis in glioma exposed to ionizing radiation. Cell Death Differ 18, 666–677 (2011). https://doi.org/10.1038/cdd.2010.139
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/cdd.2010.139
Keywords
This article is cited by
-
NLRP6 potentiates PI3K/AKT signalling by promoting autophagic degradation of p85α to drive tumorigenesis
Nature Communications (2023)
-
Comparison of the effects of oxidative and inflammatory stresses on rat chondrocyte senescence
Scientific Reports (2023)
-
Gene Expression Profile of 3D Spheroids in Comparison with 2D Cell Cultures and Tissue Strains of Diffuse High-Grade Gliomas
Bulletin of Experimental Biology and Medicine (2023)
-
An untapped window of opportunity for glioma: targeting therapy-induced senescence prior to recurrence
npj Precision Oncology (2023)
-
Exploiting senescence for the treatment of cancer
Nature Reviews Cancer (2022)