Abstract
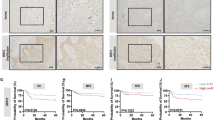
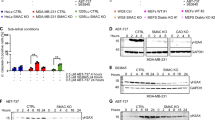
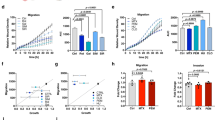
Ca2+-mediated mitochondrial permeability transition (mPT) is the final common pathway of stress-induced cell death in many major pathologies, but its regulation in intact cells is poorly understood. Here we report that the mitochondrial carrier SCaMC-1/SLC25A24 mediates ATP-Mg2−/Pi2− and/or HADP2−/Pi2− uptake into the mitochondria after an increase in cytosolic [Ca2+]. ATP and ADP contribute to Ca2+ buffering in the mitochondrial matrix, resulting in desensitization of the mPT. Comprehensive gene expression analysis showed that SCaMC-1 overexpression is a general feature of transformed and cancer cells. Knockdown of the transporter led to vast reduction of mitochondrial Ca2+ buffering capacity and sensitized cells to mPT-mediated necrotic death triggered by oxidative stress and Ca2+ overload. These findings revealed that SCaMC-1 exerts a negative feedback control between cellular Ca2+ overload and mPT-dependent cell death, suggesting that the carrier might represent a novel target for cancer therapy.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Abbreviations
- [Ca2+]cyt:
-
cytosolic [Ca2+]
- [Ca2+]m:
-
mitochondrial [Ca2+]
- ANT:
-
adenine nucleotide translocator
- BKA:
-
bongkrekic acid
- CAT:
-
carboxyatractyloside
- CRC:
-
Ca2+ retention capacity
- CsA:
-
cyclosporin-A
- CyP-D:
-
cyclophilin-D
- cytAEQ:
-
cytosolic aequorin
- ER:
-
endoplasmic reticulum
- IC:
-
intracellular
- IMM:
-
inner mitochondrial membrane
- IP3:
-
inositol-1,4,5-trisphosphate
- MCU:
-
mitochondrial Ca2+ uniporter
- mPT:
-
mitochondrial permeability transition
- mPTP:
-
mitochondrial permeability transition pore
- mtAEQmut:
-
low-affinity mitochondrial aequorin
- mtAEQwt:
-
mitochondrial aequorin
- RR:
-
ruthenium red
- ROS:
-
reactive oxygen species
- VDAC:
-
voltage-dependent anion channel
- ΔΨ:
-
mitochondrial membrane potential
References
Hanahan D, Weinberg RA . Hallmarks of cancer: the next generation. Cell 2011; 144: 646–674.
Kroemer G, Pouyssegur J . Tumor cell metabolism: cancer's Achilles’ heel. Cancer Cell 2008; 13: 472–482.
Kroemer G, Galluzzi L, Brenner C . Mitochondrial membrane permeabilization in cell death. Physiol Rev 2007; 87: 99–163.
Di Lisa F, Bernardi P . Mitochondria and ischemia-reperfusion injury of the heart: fixing a hole. Cardiovasc Res 2006; 70: 191–199.
Baines CP, Kaiser RA, Purcell NH, Blair NS, Osinska H, Hambleton MA et al. Loss of cyclophilin D reveals a critical role for mitochondrial permeability transition in cell death. Nature 2005; 434: 658–662.
Scorrano L, Oakes SA, Opferman JT, Cheng EH, Sorcinelli MD, Pozzan T et al. BAX and BAK regulation of endoplasmic reticulum Ca2+: a control point for apoptosis. Science 2003; 300: 135–139.
Chiara F, Castellaro D, Marin O, Petronilli V, Brusilow WS, Juhaszova M et al. Hexokinase II detachment from mitochondria triggers apoptosis through the permeability transition pore independent of voltage-dependent anion channels. PLoS One 2008; 3: e1852.
Rasola A, Sciacovelli M, Chiara F, Pantica B, Brusilow WS, Bernardi P . Activation of mitochondrial ERK protects cancer cells from death through inhibition of the permeability transition. Proc Natl Acad Sci USA 2010; 107: 726–731.
Jones AW, Szabadkai G . Ca2+ transfer from the ER to mitochondria: channeling cell death by a tumor suppressor. Dev Cell 2011; 19: 789–790.
Zoratti M, Szabò I . The mitochondrial permeability transition. Biochim Biophys Acta 1995; 1241: 139–176.
Aprille JR . Mechanism and regulation of the mitochondrial ATP-Mg/P(i) carrier. J Bioenerg Biomembr 1993; 25: 473–481.
Satrústegui J, Pardo B, del Arco A . Mitochondrial transporters as novel targets for intracellular calcium signaling. Physiol Rev 2007; 87: 29–67.
Traba J, Satrústegui J, del Arco A . Characterization of SCaMC-3-like/slc25a41, a novel calcium-independent mitochondrial ATP-Mg/Pi carrier. Biochem J 2009; 418: 125–133.
del Arco A, Satrústegui J . New mitochondrial carriers: an overview. Cell Mol Life Sci 2005; 62: 2204–2227.
del Arco A, Satrústegui J . Identification of a novel human subfamily of mitochondrial carriers with calcium-binding domains. J Biol Chem 2004; 279: 24701–24713.
Traba J, Froschauer EM, Wiesenberger G, Satrústegui J, del Arco A . Yeast mitochondria import ATP through the calcium-dependent ATP-Mg/Pi carrier Sal1p, and are ATP consumers during aerobic growth in glucose. Mol Microbiol 2008; 69: 570–585.
Poncet D, Pauleau AL, Szabadkai G, Vozza A, Scholz SR, Le Bras M et al. Cytopathic effects of the cytomegalovirus-encoded apoptosis inhibitory protein vMIA. J Cell Biol 2006; 174: 985–996.
Fiermonte G, De Leonardis F, Todisco S, Palmieri L, Lasorsa FM, Palmieri F . Identification of the mitochondrial ATP-Mg/Pi transporter. Bacterial expression, reconstitution, functional characterization, and tissue distribution. J Biol Chem 2004; 279: 30722–30730.
Duchen MR . Mitochondria and Ca(2+)in cell physiology and pathophysiology. Cell Calcium 2000; 28: 339–348.
Bernardi P, Krauskopf A, Basso E, Petronilli V, Blachly-Dyson E, Di Lisa F et al. The mitochondrial permeability transition from in vitro artifact to disease target. FEBS J 2006; 273: 2077–2099.
Szabadkai G, Simoni AM, Chami M, Wieckowski MR, Youle RJ, Rizzuto R . Drp-1-dependent division of the mitochondrial network blocks intraorganellar Ca2+ waves and protects against Ca2+-mediated apoptosis. Mol Cell 2004; 16: 59–68.
Chiesa A, Rapizzi E, Tosello V, Pinton P, de Virgilio M, Fogarty KE et al. Recombinant aequorin and green fluorescent protein as valuable tools in the study of cell signalling. Biochem J 2001; 355: 1–12.
Nicholls DG, Chalmers S . The integration of mitochondrial calcium transport and storage. J Bioenerg Biomembr 2004; 36: 277–281.
Carafoli E, Rossi CS, Lehninger AL . Uptake of adenine nucleotides by respiring mitochondria during active accumulation of Ca++ and phosphate. J Biol Chem 1965; 240: 2254–2261.
Kristián T, Pivovarova NB, Fiskum G, Andrews SB . Calcium-induced precipitate formation in brain mitochondria: composition, calcium capacity, and retention. J Neurochem 2007; 102: 1346–1356.
Kokoszka JE, Waymire KG, Levy SE, Sligh JE, Cai J, Jones DP et al. The ADP/ATP translocator is not essential for the mitochondrial permeability transition pore. Nature 2004; 427: 461–465.
Gizatullina ZZ, Chen Y, Zierz S, Gellerich FN . Effects of extramitochondrial ADP on permeability transition of mouse liver mitochondria. Biochim Biophys Acta 2005; 1706: 98–104.
Kristián T, Gertsch J, Bates TE, Siesjö BK . Characteristics of the calcium-triggered mitochondrial permeability transition in nonsynaptic brain mitochondria: effect of cyclosporin A and ubiquinone O. J Neurochem 2000; 74: 1999–2009.
Chen JS, Chen KT, Fan CW, Han CL, Chen YJ, Yu JS et al. Comparison of membrane fraction proteomic profiles of normal and cancerous human colorectal tissues with gel-assisted digestion and iTRAQ labeling mass spectrometry. FEBS J 2010; 277: 3028–3038.
Chen YW, Chou HC, Lyu PC, Yin HS, Huang FL, Chang WS et al. Mitochondrial proteomics analysis of tumorigenic and metastatic breast cancer markers. Funct Integr Genomics 2011; 11: 225–239.
Fountoulakis M, Schlaeger EJ . The mitochondrial proteins of the neuroblastoma cell line IMR-32. Electrophoresis 2003; 24: 260–275.
Pinton P, Ferrari D, Rapizzi E, Di Virgilio F, Pozzan T, Rizzuto R . The Ca2+ concentration of the endoplasmic reticulum is a key determinant of ceramide-induced apoptosis: significance for the molecular mechanism of Bcl-2 action. EMBO J 2001; 20: 2690–2701.
Roy SS, Madesh M, Davies E, Antonsson B, Danial N, Hajnóczky G . Bad targets the permeability transition pore independent of Bax or Bak to switch between Ca2+-dependent cell survival and death. Mol Cell 2009; 33: 377–388.
Hagen T, Lagace CJ, Modica-Napolitano JS, Aprille JR . Permeability transition in rat liver mitochondria is modulated by the ATP-Mg/Pi carrier. Am J Physiol Gastrointest Liver Physiol 2003; 285: G274–G281.
Chinopoulos C, Adam-Vizi V . Mitochondrial Ca(2+) sequestration and precipitation revisited. FEBS J 2010; 277: 3637–3651.
Weinbach EC, von Brand T . The isolation and composition of dense granules from Ca++-loaded mitochondria. Biochem Biophys Res Commun 1965; 19: 133–137.
Becker GL, Chen CH, Greenawalt JW, Lehninger AL . Calcium phosphate granules in the hepatopancreas of the blue crab Callinectes sapidus. J Cell Biol 1974; 61: 316–326.
Pivovarova NB, Andrews SB . Calcium-dependent mitochondrial function and dysfunction in neurons. FEBS J 2010; 277: 3622–3636.
Hosokawa T, Saito T, Asada A, Fukunaga K, Hisanaga S . Quantitative measurement of in vivo phosphorylation states of Cdk5 activator p35 by Phos-tag SDS-PAGE. Mol Cell Proteomics 2010; 9: 1133–1143.
Uhlén M, Björling E, Agaton C, Szigyarto CA, Amini B, Andersen E et al. A human protein atlas for normal and cancer tissues based on antibody proteomics. Mol Cell Proteomics 2005; 4: 1920–1932.
Acknowledgements
We thank Dr. María Sánchez-Aragó, Dr. Laura Formentini, Laura Sánchez-Cenizo and María Royo for help, reagents and comments; Dr. Claudia Castillo for help with data analysis; and Alejandro Arandilla for technical assistance. We also thank Dr. Paolo Bernardi and Dr. Andrea Rasola for the supply of plasmid pcDNA3-FLAG-CyP-D. This work was supported by grants from Ministerio de Educación y Ciencia (BFU2008-04084/BMC), Comunidad de Madrid (S-GEN-0269-2006 MITOLAB-CM), the European Union (LSHM-CT-2006-518153), and CIBERER (an initiative of the ISCIII) to JS; by grants from the ISCIII (PI080610) to AdelA; and by an institutional grant from the Fundación Ramón Areces to the Centro de Biología Molecular Severo Ochoa. GS was supported by Parkinson's UK (G-0905). JT is a recipient of a fellowship from Comunidad de Madrid.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Edited by R Gottlieb
Supplementary Information accompanies the paper on Cell Death and Differentiation website
Supplementary information
Rights and permissions
About this article
Cite this article
Traba, J., del Arco, A., Duchen, M. et al. SCaMC-1 promotes cancer cell survival by desensitizing mitochondrial permeability transition via ATP/ADP-mediated matrix Ca2+ buffering. Cell Death Differ 19, 650–660 (2012). https://doi.org/10.1038/cdd.2011.139
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/cdd.2011.139
Keywords
This article is cited by
-
Proteomic study of left ventricle and cortex in rats after myocardial infarction
Scientific Reports (2024)
-
Tumor bud-derived CCL5 recruits fibroblasts and promotes colorectal cancer progression via CCR5-SLC25A24 signaling
Journal of Experimental & Clinical Cancer Research (2022)
-
Lipodystrophy-associated progeroid syndromes
Hormones (2022)
-
A 9-year-old Korean girl with Fontaine progeroid syndrome: a case report with further phenotypical delineation and description of clinical course during long-term follow-up
BMC Medical Genetics (2019)
-
Second signals rescue B cells from activation-induced mitochondrial dysfunction and death
Nature Immunology (2018)