Abstract
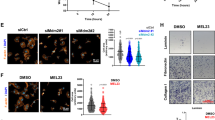
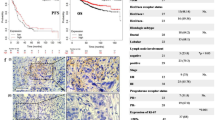
The p53 pathway is pivotal in tumor suppression. Cellular p53 activity is subject to tight regulation, in which the two related proteins Mdm2 and Mdm4 have major roles. The delicate interplay between the levels of Mdm2, Mdm4 and p53 is crucial for maintaining proper cellular homeostasis. microRNAs (miRNAs) are short non-coding RNAs that downregulate the level and translatability of specific target mRNAs. We report that miR-661, a primate-specific miRNA, can target both Mdm2 and Mdm4 mRNA in a cell type-dependent manner. miR-661 interacts with Mdm2 and Mdm4 RNA within living cells. The inhibitory effect of miR-661 is more prevalent on Mdm2 than on Mdm4. Interestingly, the predicted miR-661 targets in both mRNAs reside mainly within Alu elements, suggesting a primate-specific mechanism for regulatory diversification during evolution. Downregulation of Mdm2 and Mdm4 by miR-661 augments p53 activity and inhibits cell cycle progression in p53-proficient cells. Correspondingly, low miR-661 expression correlates with bad outcome in breast cancers that typically express wild-type p53. In contrast, the miR-661 locus tends to be amplified in tumors harboring p53 mutations, and miR-661 promotes migration of cells derived from such tumors. Thus, miR-661 may either suppress or promote cancer aggressiveness, depending on p53 status.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Abbreviations
- miRNA:
-
microRNA
- WTp53:
-
wild-type p53
- DBD:
-
DNA-binding domain
- ER+:
-
estrogen receptor positive
- GOF:
-
gain of function
- FBS:
-
fetal bovine serum
- miR-661:
-
miR-661 mimic
- miR-C:
-
miR-control mimic
- si-miR-661:
-
miR-661 inhibitor
- si-miR-C:
-
miR-control inhibitor
- sip53:
-
p53 siRNA
References
Levine AJ, Oren M . The first 30 years of p53: growing ever more complex. Nat Rev Cancer 2009; 9: 749–758.
Wade M, Li YC, Wahl GM . MDM2, MDMX and p53 in oncogenesis and cancer therapy. Nat Rev Cancer 2013; 13: 83–96.
Hu W, Feng Z, Levine AJ . The regulation of multiple p53 stress responses is mediated through MDM2. Genes Cancer 2012; 3: 199–208.
Li Q, Lozano G . Molecular pathways: targeting Mdm2 and Mdm4 in cancer therapy. Clin Cancer Res 2013; 19: 34–41.
Barak Y, Juven T, Haffner R, Oren M . Mdm2 expression is induced by wild type p53 activity. EMBO J 1993; 12: 461–468.
Wu X, Bayle JH, Olson D, Levine AJ . The p53-mdm-2 autoregulatory feedback loop. Genes Dev 1993; 7: 1126–1132.
Linares LK, Hengstermann A, Ciechanover A, Muller S, Scheffner M . HdmX stimulates Hdm2-mediated ubiquitination and degradation of p53. Proc Natl Acad Sci USA 2003; 100: 12009–12014.
Bartel DP . MicroRNAs: target recognition and regulatory functions. Cell 2009; 136: 215–233.
Hoffman Y, Dahary D, Bublik DR, Oren M, Pilpel Y . The majority of endogenous microRNA targets within Alu elements avoid the microRNA machinery. Bioinformatics 2013; 29: 894–902.
Hermeking H . MicroRNAs in the p53 network: micromanagement of tumour suppression. Nat Rev Cancer 2012; 12: 613–626.
Lujambio A, Lowe SW . The microcosmos of cancer. Nature 2012; 482: 347–355.
Wynendaele J, Bohnke A, Leucci E, Nielsen SJ, Lambertz I, Hammer S et al. An illegitimate microRNA target site within the 3' UTR of MDM4 affects ovarian cancer progression and chemosensitivity. Cancer Res 2010; 70: 9641–9649.
Orom UA, Lund AH . Isolation of microRNA targets using biotinylated synthetic microRNAs. Methods 2007; 43: 162–165.
Lal A, Thomas MP, Altschuler G, Navarro F, O'Day E, Li XL et al. Capture of microRNA-bound mRNAs identifies the tumor suppressor miR-34a as a regulator of growth factor signaling. PLoS Genet 2011; 7: e1002363.
O'Connor PM, Jackman J, Bae I, Myers TG, Fan S, Mutoh M et al. Characterization of the p53 tumor suppressor pathway in cell lines of the National Cancer Institute anticancer drug screen and correlations with the growth-inhibitory potency of 123 anticancer agents. Cancer Res 1997; 57: 4285–4300.
Antonov AV, Knight RA, Melino G, Barlev NA, Tsvetkov PO . MIRUMIR: an online tool to test microRNAs as biomarkers to predict survival in cancer using multiple clinical data sets. Cell Death Differ 2012; 20: 367.
Lyng MB, Laenkholm AV, Sokilde R, Gravgaard KH, Litman T, Ditzel HJ . Global microRNA expression profiling of high-risk ER+ breast cancers from patients receiving adjuvant tamoxifen mono-therapy: a DBCG study. PLoS One 2012; 7: e36170.
Sorlie T, Perou CM, Tibshirani R, Aas T, Geisler S, Johnsen H et al. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc Natl Acad Sci USA 2001; 98: 10869–10874.
Cerami E, Gao J, Dogrusoz U, Gross BE, Sumer SO, Aksoy BA et al. The cBio cancer genomics portal: an open platform for exploring multidimensional cancer genomics data. Cancer Discov 2012; 2: 401–404.
Ahmed AA, Etemadmoghadam D, Temple J, Lynch AG, Riad M, Sharma R et al. Driver mutations in TP53 are ubiquitous in high grade serous carcinoma of the ovary. J Pathol 2010; 221: 49–56.
Bernardini MQ, Baba T, Lee PS, Barnett JC, Sfakianos GP, Secord AA et al. Expression signatures of TP53 mutations in serous ovarian cancers. BMC Cancer 2010; 10: 237.
Oren M, Rotter V . Mutant p53 gain-of-function in cancer. Cold Spring Harb Perspect Biol 2010; 2: a001107.
Walerych D, Napoli M, Collavin L, Del Sal G . The rebel angel: mutant p53 as the driving oncogene in breast cancer. Carcinogenesis 2012; 33: 2007–2017.
Muller PA, Vousden KH, Norman JC . p53 and its mutants in tumor cell migration and invasion. J Cell Biol 2011; 192: 209–218.
Vetter G, Saumet A, Moes M, Vallar L, Le Bechec A, Laurini C et al. miR-661 expression in SNAI1-induced epithelial to mesenchymal transition contributes to breast cancer cell invasion by targeting Nectin-1 and StarD10 messengers. Oncogene 2010; 29: 4436–4448.
Reddy SD, Pakala SB, Ohshiro K, Rayala SK, Kumar R . MicroRNA-661, a c/EBPalpha target, inhibits metastatic tumor antigen 1 and regulates its functions. Cancer Res 2009; 69: 5639–5642.
Langerod A, Zhao H, Borgan O, Nesland JM, Bukholm IR, Ikdahl T et al. TP53 mutation status and gene expression profiles are powerful prognostic markers of breast cancer. Breast Cancer Res 2007; 9: R30.
Bausch D, Thomas S, Mino-Kenudson M, Fernandez-del CC, Bauer TW, Williams M et al. Plectin-1 as a novel biomarker for pancreatic cancer. Clin Cancer Res 2011; 17: 302–309.
Katada K, Tomonaga T, Satoh M, Matsushita K, Tonoike Y, Kodera Y et al. Plectin promotes migration and invasion of cancer cells and is a novel prognostic marker for head and neck squamous cell carcinoma. J Proteomics 2012; 75: 1803–1815.
Aylon Y, Ofir-Rosenfeld Y, Yabuta N, Lapi E, Nojima H, Lu X et al. The Lats2 tumor suppressor augments p53-mediated apoptosis by promoting the nuclear proapoptotic function of ASPP1. Genes Dev 2010; 24: 2420–2429.
Golomb L, Bublik DR, Wilder S, Nevo R, Kiss V, Grabusic K et al. Importin 7 and exportin 1 link c-Myc and p53 to regulation of ribosomal biogenesis. Mol Cell 2012; 45: 222–232.
Acknowledgements
We thank Judy Lieberman for generous help with pull-down expertise, Debora Marks for bioinformatics ideas and Gilad Fuchs for the fruitful discussions. This work was supported in part by an FP7 grant 201102 (ONCOMIRS) from the European Research Council (to MO and YP), the ‘Ideas’ program of the European Research Council (to YP), grant R37 CA40099 from the National Cancer Institute (to MO), the Dr. Miriam and Sheldon G Adelson Medical Research Foundation (to MO), the Ben May Charitable Trust (to YP) and the Weizmann Institute Kahn Center (to MO and YP). MO is an incumbent of the Andre Lwoff Chair in Molecular Biology. YP is an incumbent of the Ben-May Professorial Chair.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Edited by K Vousden
Supplementary Information accompanies this paper on Cell Death and Differentiation website
Supplementary information
Rights and permissions
About this article
Cite this article
Hoffman, Y., Bublik, D., Pilpel, Y. et al. miR-661 downregulates both Mdm2 and Mdm4 to activate p53. Cell Death Differ 21, 302–309 (2014). https://doi.org/10.1038/cdd.2013.146
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/cdd.2013.146
Keywords
This article is cited by
-
CircPDE7B/miR-661 axis accelerates the progression of human keloid fibroblasts by upregulating fibroblast growth factor 2 (FGF2)
Molecular and Cellular Biochemistry (2022)
-
MiR-661 promotes tumor invasion and metastasis by directly inhibiting RB1 in non small cell lung cancer
Molecular Cancer (2017)
-
miR-15a/miR-16 down-regulates BMI1, impacting Ub-H2A mediated DNA repair and breast cancer cell sensitivity to doxorubicin
Scientific Reports (2017)
-
MicroRNA-519a-3p mediates apoptosis resistance in breast cancer cells and their escape from recognition by natural killer cells
Cell Death & Disease (2017)
-
The p53 tetramer shows an induced-fit interaction of the C-terminal domain with the DNA-binding domain
Oncogene (2016)