Abstract
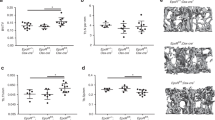
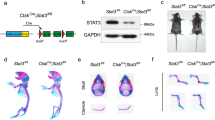
Human mutations and in vitro studies indicate that DLX3 has a crucial function in bone development, however, the in vivo role of DLX3 in endochondral ossification has not been established. Here, we identify DLX3 as a central attenuator of adult bone mass in the appendicular skeleton. Dynamic bone formation, histologic and micro-computed tomography analyses demonstrate that in vivo DLX3 conditional loss of function in mesenchymal cells (Prx1-Cre) and osteoblasts (OCN-Cre) results in increased bone mass accrual observed as early as 2 weeks that remains elevated throughout the lifespan owing to increased osteoblast activity and increased expression of bone matrix genes. Dlx3OCN-conditional knockout mice have more trabeculae that extend deeper in the medullary cavity and thicker cortical bone with an increased mineral apposition rate, decreased bone mineral density and increased cortical porosity. Trabecular TRAP staining and site-specific Q-PCR demonstrated that osteoclastic resorption remained normal on trabecular bone, whereas cortical bone exhibited altered osteoclast patterning on the periosteal surface associated with high Opg/Rankl ratios. Using RNA sequencing and chromatin immunoprecipitation-Seq analyses, we demonstrate that DLX3 regulates transcription factors crucial for bone formation such as Dlx5, Dlx6, Runx2 and Sp7 as well as genes important to mineral deposition (Ibsp, Enpp1, Mepe) and bone turnover (Opg). Furthermore, with the removal of DLX3, we observe increased occupancy of DLX5, as well as increased and earlier occupancy of RUNX2 on the bone-specific osteocalcin promoter. Together, these findings provide novel insight into mechanisms by which DLX3 attenuates bone mass accrual to support bone homeostasis by osteogenic gene pathway regulation.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Abbreviations
- Adv:
-
adenovirus
- ALPL:
-
alkaline phosphatase
- BMD:
-
bone mineral density
- BMSC:
-
bone marrow stromal cell
- ChIP:
-
chromatin immunoprecipitation
- cKO:
-
conditional knockout
- CTX-1:
-
carboxy-terminal type I collagen crosslink
- DLX3:
-
distal-less homeobox 3
- EBF:
-
endochondral bone formation
- ECM:
-
extracellular matrix
- H&E:
-
hematoxylin and eosin
- MAR:
-
mineral apposition rate
- M-BMM:
-
M-CSF-dependent bone marrow macrophages
- μCT:
-
micro-computed tomography
- OCN:
-
osteocalcin
- OPG:
-
osteoprotegerin
- RANKL:
-
receptor activator of nuclear factor kappa-B ligand
- TDO:
-
tricho-dento-osseous
- TF:
-
transcription factor
- TRAP:
-
tartrate-resistant acid phosphatase
- WT:
-
wild type
References
Cohen MM Jr . Perspectives on RUNX genes: an update. Am J Med Genet A 2009; 149A: 2629–2646.
Jensen ED, Gopalakrishnan R, Westendorf JJ . Regulation of gene expression in osteoblasts. Biofactors 2010; 36: 25–32.
Long F . Building strong bones: molecular regulation of the osteoblast lineage. Nat Rev Mol Cell Biol 2012; 13: 27–38.
Depew MJ, Simpson CA, Morasso M, Rubenstein JL . Reassessing the Dlx code: the genetic regulation of branchial arch skeletal pattern and development. J Anat 2005; 207: 501–561.
Panganiban G, Rubenstein JL . Developmental functions of the Distal-less/Dlx homeobox genes. Development 2002; 129: 4371–4386.
Samee N, de Vernejoul MC, Levi G . Role of DLX regulatory proteins in osteogenesis and chondrogenesis. Crit Rev Eukaryot Gene Expr 2007; 17: 173–186.
Shamseldin HE, Faden MA, Alashram W, Alkuraya FS . Identification of a novel DLX5 mutation in a family with autosomal recessive split hand and foot malformation. J Med Genet 2012; 49: 16–20.
Haldeman RJ, Cooper LF, Hart TC, Phillips C, Boyd C, Lester GE et al. Increased bone density associated with DLX3 mutation in the tricho-dento-osseous syndrome. Bone 2004; 35: 988–997.
Hassan MQ, Javed A, Morasso MI, Karlin J, Montecino M, van Wijnen AJ et al. Dlx3 transcriptional regulation of osteoblast differentiation: temporal recruitment of Msx2, Dlx3, and Dlx5 homeodomain proteins to chromatin of the osteocalcin gene. Mol Cell Biol 2004; 24: 9248–9261.
Hassan MQ, Tare RS, Lee SH, Mandeville M, Morasso MI, Javed A et al. BMP2 commitment to the osteogenic lineage involves activation of Runx2 by DLX3 and a homeodomain transcriptional network. J Biol Chem 2006; 281: 40515–40526.
Singh M, Del Carpio-Cano FE, Monroy MA, Popoff SN, Safadi FF . Homeodomain transcription factors regulate BMP-2-induced osteoactivin transcription in osteoblasts. J Cell Physiol 2012; 227: 390–399.
Duverger O, Isaac J, Zah A, Hwang J, Berdal A, Lian JB et al. In vivo impact of Dlx3 conditional inactivation in neural crest-derived craniofacial bones. J Cell Physiol 2013; 228: 654–664.
Choi SJ, Roodman GD, Feng JQ, Song IS, Amin K, Hart PS et al. In vivo impact of a 4 bp deletion mutation in the DLX3 gene on bone development. Dev Biol 2009; 325: 129–137.
Lezot F, Thomas BL, Blin-Wakkach C, Castaneda B, Bolanos A, Hotton D et al. Dlx homeobox gene family expression in osteoclasts. J Cell Physiol 2010; 223: 779–787.
Doi M, Nagano A, Nakamura Y . Molecular cloning and characterization of a novel gene, EMILIN-5, and its possible involvement in skeletal development. Biochem Biophys Res Commun 2004; 313: 888–893.
Li YC, Pirro AE, Amling M, Delling G, Baron R, Bronson R et al. Targeted ablation of the vitamin D receptor: an animal model of vitamin D-dependent rickets type II with alopecia. Proc Natl Acad Sci USA 1997; 94: 9831–9835.
Mackenzie NC, Zhu D, Milne EM, van 't Hof R, Martin A, Darryl Quarles L et al. Altered bone development and an increase in FGF-23 expression in Enpp1(−/−) mice. PLoS One 2012; 7: e32177.
Narisawa S, Frohlander N, Millan JL . Inactivation of two mouse alkaline phosphatase genes and establishment of a model of infantile hypophosphatasia. Dev Dyn 1997; 208: 432–446.
Staines KA, MacRae VE, Farquharson C . The importance of the SIBLING family of proteins on skeletal mineralisation and bone remodelling. J Endocrinol 2012; 214: 241–255.
Wellik DM . Hox genes and vertebrate axial pattern. Curr Top Dev Biol 2009; 88: 257–278.
Petersen DN, Tkalcevic GT, Mansolf AL, Rivera-Gonzalez R, Brown TA . Identification of osteoblast/osteocyte factor 45 (OF45), a bone-specific cDNA encoding an RGD-containing protein that is highly expressed in osteoblasts and osteocytes. J Biol Chem 2000; 275: 36172–36180.
Chen G, Deng C, Li YP . TGF-beta and BMP signaling in osteoblast differentiation and bone formation. Int J Biol Sci 2012; 8: 272–288.
Gazzerro E, Canalis E . Bone morphogenetic proteins and their antagonists. Rev Endocr Metab Disord 2006; 7: 51–65.
Monroe DG, McGee-Lawrence ME, Oursler MJ, Westendorf JJ . Update on Wnt signaling in bone cell biology and bone disease. Gene 2012; 492: 1–18.
Zanotti S, Canalis E . Notch signaling in skeletal health and disease. Eur J Endocrinol 2013; 168: R95–103.
Rawlinson SC, McKay IJ, Ghuman M, Wellmann C, Ryan P, Prajaneh S et al. Adult rat bones maintain distinct regionalized expression of markers associated with their development. PLoS One 2009; 4: e8358.
Leucht P, Kim JB, Amasha R, James AW, Girod S, Helms JA . Embryonic origin and Hox status determine progenitor cell fate during adult bone regeneration. Development 2008; 135: 2845–2854.
Hassan MQ, Saini S, Gordon JA, van Wijnen AJ, Montecino M, Stein JL et al. Molecular switches involving homeodomain proteins, HOXA10 and RUNX2 regulate osteoblastogenesis. Cells Tissues Organs 2009; 189: 122–125.
Otto F, Thornell AP, Crompton T, Denzel A, Gilmour KC, Rosewell IR et al. Cbfa1, a candidate gene for cleidocranial dysplasia syndrome, is essential for osteoblast differentiation and bone development. Cell 1997; 89: 765–771.
Nakashima K, Zhou X, Kunkel G, Zhang ZP, Deng JM, Behringer RR et al. The novel zinc finger-containing transcription factor Osterix is required for osteoblast differentiation and bone formation. Cell 2002; 108: 17–29.
Ferrari D, Sumoy L, Gannon J, Sun HL, Brown AMC, Upholt WB et al. The expression pattern of the distal-less homeobox-containing gene Dlx-5 in the developing chick limb bud suggests its involvement in apical ectodermal ridge activity, pattern-formation, and cartilage differentiation. Mech Dev 1995; 52: 257–264.
Lee MH, Kim YJ, Yoon WJ, Kim JI, Kim BG, Hwang YS et al. Dlx5 specifically regulates Runx2 type II expression by binding to homeodomain-response elements in the Runx2 distal promoter. J Biol Chem 2005; 280: 35579–35587.
Samee N, Geoffroy V, Marty C, Schiltz C, Vieux-Rochas M, Levi G et al. Dlx5, a positive regulator of osteoblastogenesis, is essential for osteoblast-osteoclast coupling. Am J Pathol 2008; 173: 773–780.
Tadic T, Dodig M, Erceg I, Marijanovic I, Mina M, Kalajzic Z et al. Overexpression of Dlx5 in chicken calvarial cells accelerates osteoblastic differentiation. J Bone Miner Res 2002; 17: 1008–1014.
Depew MJ, Lufkin T, Rubenstein JL . Specification of jaw subdivisions by Dlx genes. Science 2002; 298: 381–385.
Nam HK, Liu J, Li Y, Kragor A, Hatch NE . Ectonucleotide pyrophosphatase/phosphodiesterase-1 (ENPP1) protein regulates osteoblast differentiation. J Biol Chem 2011; 286: 39059–39071.
Malaval L, Wade-Gueye NM, Boudiffa M, Fei J, Zirngibl R, Chen F et al. Bone sialoprotein plays a functional role in bone formation and osteoclastogenesis. J Exp Med 2008; 205: 1145–1153.
Ulsamer A, Ortuno MJ, Ruiz S, Susperregui AR, Osses N, Rosa JL et al. BMP-2 induces Osterix expression through up-regulation of Dlx5 and its phosphorylation by p38. J Biol Chem 2008; 283: 3816–3826.
Kim Y, Lee M, Wozney JM, Cho J, Ryoo H . BMP2-induced alkaline phosphatase expression is regulated by Dlx5, which is interrupted by Msx2 as a binding competition to the common response element. J Bone Miner Res 2004; 19: S264–S264.
Benson MD, Bargeon JL, Xiao G, Thomas PE, Kim A, Cui Y et al. Identification of a homeodomain binding element in the bone sialoprotein gene promoter that is required for its osteoblast-selective expression. J Biol Chem 2000; 275: 13907–13917.
Nakayama Y, Nakajima Y, Kato N, Takai H, Kim DS, Arai M et al. Insulin-like growth factor-1 increases bone sialoprotein (BSP) expression through fibroblast growth factor-2 response element and homeodomain protein-binding site in the proximal promoter of the BSP gene. J Cell Physiol 2006; 208: 326–335.
Hayashibara T, Hiraga T, Sugita A, Wang L, Hata K, Ooshima T et al. Regulation of osteoclast differentiation and function by phosphate: potential role of osteoclasts in the skeletal abnormalities in hypophosphatemic conditions. J Bone Miner Res 2007; 22: 1743–1751.
Kramer I, Halleux C, Keller H, Pegurri M, Gooi JH, Weber PB et al. Osteocyte Wnt/beta-catenin signaling is required for normal bone homeostasis. Mol Cell Biol 2010; 30: 3071–3085.
Kulkarni RN, Bakker AD, Everts V, Klein-Nulend J . Inhibition of osteoclastogenesis by mechanically loaded osteocytes: involvement of MEPE. Calcif Tissue Int 2010; 87: 461–468.
Liu JC, Lengner CJ, Gaur T, Lou Y, Hussain S, Jones MD et al. Runx2 protein expression utilizes the Runx2 P1 promoter to establish osteoprogenitor cell number for normal bone formation. J Biol Chem 2011; 286: 30057–30070.
Aioub M, Lezot F, Molla M, Castaneda B, Robert B, Goubin G et al. Msx2 −/− transgenic mice develop compound amelogenesis imperfecta, dentinogenesis imperfecta and periodental osteopetrosis. Bone 2007; 41: 851–859.
Grcevic D, Pejda S, Matthews BG, Repic D, Wang L, Li H et al. In vivo fate mapping identifies mesenchymal progenitor cells. Stem Cells 2012; 30: 187–196.
Hwang J, Kita R, Kwon HS, Choi EH, Lee SH, Udey MC et al. Epidermal ablation of Dlx3 is linked to IL-17-associated skin inflammation. Proc Natl Acad Sci USA 2011; 108: 11566–11571.
Acknowledgements
We thank Dr. T Clemens for the OCN-cre mice; S Russell, of the University of Massachusetts MusculoSkeletal Imaging Facility for performing μCT analyses; Dr. K Zaal of the NIAMS Light Imaging Core Facility; G Gutierrez-Cruz of the Genome Analyzer Core Facility; Drs. A Maeda and M Young of the NIDCR, NIH; Dr. B Foster of NIAMS, NIH for assistance with ALPL staining, Audrey Asselin of the INSERM UMRS 1138 (Team Berdal), for assistance with cell culture, and Christophe Klein of the INSERM UMRS 1138 (CICC) and Laura Solomon (UVM graduate student) for osteoclast quantification. The Morasso and Lian laboratories and their funding contributed equally to this project. This research was supported by the Intramural Research Program of the National Institute of Arthritis and Musculoskeletal and Skin Diseases of the NIH. JBL is supported by NIH grant R37 DE012528.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Edited by G Melino
Supplementary Information accompanies this paper on Cell Death and Differentiation website
Supplementary information
Rights and permissions
About this article
Cite this article
Isaac, J., Erthal, J., Gordon, J. et al. DLX3 regulates bone mass by targeting genes supporting osteoblast differentiation and mineral homeostasis in vivo. Cell Death Differ 21, 1365–1376 (2014). https://doi.org/10.1038/cdd.2014.82
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/cdd.2014.82
This article is cited by
-
Temporal Modulation of DNA Methylation and Gene Expression in Monolayer and 3D Spheroids of Dental Pulp Stem Cells during Osteogenic Differentiation: A Comparative Study
Tissue Engineering and Regenerative Medicine (2022)
-
DLX5 and HOXC8 enhance the chondrogenic differentiation potential of stem cells from apical papilla via LINC01013
Stem Cell Research & Therapy (2020)
-
DLX3 interacts with GCM1 and inhibits its transactivation-stimulating activity in a homeodomain-dependent manner in human trophoblast-derived cells
Scientific Reports (2017)
-
Senescence: novel insight into DLX3 mutations leading to enhanced bone formation in Tricho-Dento-Osseous syndrome
Scientific Reports (2016)
-
Dlx genes and the maintenance of bone homeostasis and skeletal integrity
Cell Death & Differentiation (2014)