Abstract
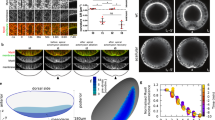
By laser scanning fluorescence microscopy for quantitative measurement of fluorescence intensity changes on egg surface stained with fluorescein isothiocvanate during cleavage furrow extending forward, it was found that in area of presumptive cleavage furrow the scanning curve became ∨ shape, indicating dark stripe appeared in that place. Then the fluorescence intensity increased at the place where the bottom of ∨ shape had located, and the scanning curve turned to ∧ shape, indicating single stripe was formed. While enhanced fluorescence appeared on the borders of ∧ shape, an M shape curve was found, showing double stripe occurred. During the distance between two borders of M shape increasing from 50 μm to 100 μm, a fluorescence peak came to sight in the middle of the M shape, which being the cleavge furrow bottom. The two lateral sides of furrow bottom with decreasing fluorescence were nascent membrane. At that time the curve became W shape. By the sides of cleavage furrow the the stress folds became conspicous after double stripe stage, showing the stretching of the egg surface being increased. With our31, 33 and others32 reports that polylysine could induce the appearance of nascent membrane and phytohemagglutinins could decrease or prevent the appearance of nascent membrane, we believed the idea of Schroeder25 that increasing mechanical stress could initiate nascent membrane formation and thought that the stress lay to the outsides of cleavage furrow.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Dan K, Kojima MK . A study on the mechanism of cleavage in the amphibian egg. J Exp Biol; 1963; 40:7–14.
Sawai T. On propagation of cortical factor and cytoplasmic factor participating in cleavage furrow formation of the newt's egg. Dev Growth Differ 1980; 22:437–44.
Mabuchi I, Tsukita Sh, Tsukita Sa, Sawai T . Cleavage furrow isolated from newt eggs: Contraction, organization of the actin filaments, and protein components of the furrow. Proc Natl Acad Sci USA 1988; 85:5966–70.
Sato N, Yonemurea S, Obinata T, Tsukita Sa, Tsukita Sh . Radixin, a barbed end-capping actin-modulating protein, is concentrated at the cleavage furrow during cytokinesis. J Cell Biol 1991; 113:321–30.
Yonemura S, Nagafuchi A, Sato N, Tsukita S . Concentration of an integral membrane protein, CD43 (Leukosialin, Sialophorin), in the cleavage furrow through the interaction of its cytoplasmic domain with actin-based cytoskeletons. J Cell Biol 1993; 120:437–49.
Ku KY, Xu CT, Zhang KH . Cleavage furrow formation of Rana Amurensis eggs observed with fluorescence pattern photobleaching. Cell Biol Inter Rep 1988; 12:175–87.
Bluemink JK . Cytokinesis and cytochalasin-induced furrow regression in the first cleavage zygote of Xenopus laevis. Z Zellforsch Mikrosk Anat 1971; 121:102–26.
Singal PK, Sanders EJ . An ultrastructural study of the first cleavage of Xenopus embryos. J Ultrastruct Res 1974; 47: 433–51.
Akkas N . Editor of “Biomechanics of Cell Division”. Plenum Press. New York and London 1987.
White JG, Borisy GG . On the mechanisms of cytokinesis in animal cell. J Theor Biol 1983; 101: 289–316.
Gao QR, Liu XH, Zhang KH, et a1. Surface contraction wave and cleavage initiation contraction on the fertilized egg of Rana Amurensis. Acta Biol Exp Sinica 1993; 26: 449–61.
Sawai T . Cytoplasmic and cortical factors participating in cleavage furrow formation in eggs of three amphibian genera: Ambystoma, Xenopus and Cynops. J Embryol Exp Morph 1983; 77: 243–54.
Jiang S, Li MF, Ku KY . Bioassay and isolation of contraction factor(s) from Rana Amurensis embryo. Acta Biol Exp Sinica 1988; 21:383–91.
Schroeder TE . The contractile ring and furrowing in dividing cells. Annals New York Academy of Sciences 1990; 582: 78–87.
Hong LS, Gao KG, Cheng PR, Ku KY, A scanning electron microscopic study of the formation and regression of the first cleavage furrow of Rana Amurensis. Acta Biol Exp Sinica 1981; 14:161–71.
Ku KY, Hong LS, Jiang S . 1982 Morphological changes of the surface of the eggs of Rana Amurensis during first cleavage by fluorescence microscopy. Acta Biol Exp Sinica 15: 375–85.
Satterwhite LL, Pollard TD . Cytokinesis. Current Opinion in Cell Biology. 1992; 4: 43–52.
Gao QR, Zhang KH, Hong LS, Xu CT, Gu Guo-yan (formerly Ku KY). The development of the region between the preexisting and nascent membranes during the first cleavage of Rana Amurensis eggs. Cell Reseach 1992; 2: 35–43.
Selman GG, Waddington CH . The mechanism of cell division in the cleavage of the newt's egg. J Exp Biol 1955; 32: 700–35.
Byers TJ, Armstron PB . Membrane protein redistribution during Xenopus first cleavage. J Cell Biol 1986; 102: 2176–84.
Sawai T . Movement of the cell surface and change in surface area during cleavage in the newt's egg. J Cell Sci 1976; 21: 537–51.
Bluemink JK, De Laat SW . New membrane fclrmation during cytokinesis in normal and cytochalasin B-treated eggs of Xenopus laevis. I. Electron microscope observations. J Cell Biol 1973; 59: 89–108.
Gao QR, Hong LS, Jiang S, Ku KY . Detergent, Brij, increasing the area of new surface membrane during the early cleavage of eggs of Rana Amurensis. Cell Biol Inter Rep 1986; 10:969–77.
Bieliavsky N, Geuskeas M . Interblastomeric plasma membrane formation during cleavage of Xenopus laevis embryos. J Submicrosc Cytol Pathol, 1990; 22:445–57.
Schreder TE . Interrelations between the cell surface and the cytoskeleton in cleaving sea uvclin eggs. In: cytoskeletal elements and plasms membrane organization (poste G. & Nicolzon, G. L. eds). 1981:pp 169–215.
Ku KY, Xu CT, Zhang KH, Gao QR, Jiang S . The Movement of egg surface of Rana Amurensis before and during cytokinesis. First Congress of Asian-pacific Organization for Cell Biology. Abstracts. 1990: s5.5, pp. 67–8.
Kobayakawa Y, Kubota HY . Temporal pattern of cleavage and the onset of gastrulation in amphibian embryos developed from with the reduced cytoplasm. J Embryol exp Morph 1981; 62: 83–94.
Li MF, Hong LS, Ku KY . A rapid method for the removal of jelly coat and vitelline membrane of fertilized eggs of Rana Amurensis. Acta Biol Exp Sinica 1980; 13: 105–11.
Sawai T . Cychc changes in the cortical layer of non-nucleated framents of the newt's egg. J Embryol Exp Morph 1979; 51: 183–93.
Kubota HY, Itoh K, Kubota MA . Cytological and maternal-effect mutant embryos with abnormal cleavage furrow formation in Xenopus laevis. Dev Biol 1991; 144:145–51.
Wu YL, Jiang S, Ku KY . The effects of polylysine on the aggregation of surface and on the cleavage furrow of Rana Amurensis eggs. Acta Biol Exp Sinica 1993; 26:51–64.
Tencer R . Transmembrane effects of lectins. I. Effects of wheat germ agglutinin and soybean agglutinin on furrow formation and cortical wound healing in Xenopus laevis eggs. Exp Cell Res 1978; 116: 253–60.
Ku KY, Hong LS . The inhibitory effects of lectins on the exposure of new membrane during the first cleavage of Rana Amurensis eggs. Acta Biol Exp Sinica 1984;17: 329–35.
Author information
Authors and Affiliations
Additional information
*Dedicated to Prefessor Zhen YAO on the occasion of his 80th birthday.
†This work was supported by the director's foundation of Shanghai Institute of Cell Biology
Rights and permissions
About this article
Cite this article
Gu, G., Xu, C., Zhang, K. et al. Laser scanning fluorescence microscopic measurement of the movement of cleaving egg surface of Rana Amurensis . Cell Res 5, 9–24 (1995). https://doi.org/10.1038/cr.1995.2
Received:
Revised:
Accepted:
Issue date:
DOI: https://doi.org/10.1038/cr.1995.2