Abstract
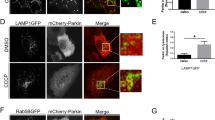
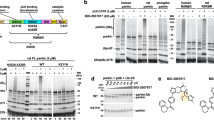
Pink1, a mitochondrial kinase, and Parkin, an E3 ubiquitin ligase, function in mitochondrial maintenance. Pink1 accumulates on depolarized mitochondria, where it recruits Parkin to mainly induce K63-linked chain ubiquitination of outer membrane proteins and eventually mitophagy. Parkin belongs to the RBR E3 ligase family. Recently, it has been proposed that the RBR domain transfers ubiquitin to targets via a cysteine∼ubiquitin enzyme intermediate, in a manner similar to HECT domain E3 ligases. However, direct evidence for a ubiquitin transfer mechanism and its importance for Parkin's in vivo function is still missing. Here, we report that Parkin E3 activity relies on cysteine-mediated ubiquitin transfer during mitophagy. Mutating the putative catalytic cysteine to serine (Parkin C431S) traps ubiquitin, and surprisingly, also abrogates Parkin mitochondrial translocation, indicating that E3 activity is essential for Parkin translocation. We found that Parkin can bind to K63-linked ubiquitin chains, and that targeting K63-mimicking ubiquitin chains to mitochondria restores Parkin C431S localization. We propose that Parkin translocation is achieved through a novel catalytic activity coupled mechanism.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Rugarli EI, Langer T . Mitochondrial quality control: a matter of life and death for neurons. EMBO J 2012; 31:1336–1349.
Youle RJ, Narendra DP . Mechanisms of mitophagy. Nat Rev Mol Cell Biol 2011; 12:9–14.
Greene JC, Whitworth AJ, Kuo I, Andrews LA, Feany MB, Pallanck LJ . Mitochondrial pathology and apoptotic muscle degeneration in Drosophila parkin mutants. Proc Natl Acad Sci USA 2003; 100:4078–4083.
Pesah Y, Pham T, Burgess H, et al. Drosophila parkin mutants have decreased mass and cell size and increased sensitivity to oxygen radical stress. Development 2004; 131:2183–2194.
Clark IE, Dodson MW, Jiang C, et al. Drosophila pink1 is required for mitochondrial function and interacts genetically with parkin. Nature 2006; 441:1162–1166.
Park J, Lee SB, Lee S, et al. Mitochondrial dysfunction in Drosophila PINK1 mutants is complemented by parkin. Nature 2006; 441:1157–1161.
Narendra D, Tanaka A, Suen DF, Youle RJ . Parkin is recruited selectively to impaired mitochondria and promotes their autophagy. J Cell Biol 2008; 183:795–803.
Deng H, Dodson MW, Huang H, Guo M . The Parkinson's disease genes pink1 and parkin promote mitochondrial fission and/or inhibit fusion in Drosophila. Proc Natl Acad Sci USA 2008; 105:14503–14508.
Poole AC, Thomas RE, Andrews LA, McBride HM, Whitworth AJ, Pallanck LJ . The PINK1/Parkin pathway regulates mitochondrial morphology. Proc Natl Acad Sci USA. 2008; 105:1638–1643.
Ziviani E, Tao RN, Whitworth AJ . Drosophila parkin requires PINK1 for mitochondrial translocation and ubiquitinates mitofusin. Proc Natl Acad Sci USA 2010; 107:5018–5023.
Wang X, Winter D, Ashrafi G, et al. PINK1 and Parkin target Miro for phosphorylation and degradation to arrest mitochondrial motility. Cell 2011; 147:893–906.
Dawson TM, Dawson VL . The role of parkin in familial and sporadic Parkinson's disease. Mov Disord 2010; 25 Suppl 1:S32–9.
Kahle PJ, Haass C . How does parkin ligate ubiquitin to Parkinson's disease? EMBO Rep 2004; 5:681–685.
Lim KL, Chew KC, Tan JM, et al. Parkin mediates nonclassical, proteasomal-independent ubiquitination of synphilin-1: implications for Lewy body formation. J Neurosci 2005; 25:2002–2009.
Geisler S, Holmström KM, Skujat D, et al. PINK1/Parkin-mediated mitophagy is dependent on VDAC1 and p62/SQSTM1. Nat Cell Biol 2010; 12:119–131.
Matsuda N, Sato S, Shiba K, et al. PINK1 stabilized by mitochondrial depolarization recruits Parkin to damaged mitochondria and activates latent Parkin for mitophagy. J Cell Biol 2010; 189:211–221.
Narendra D, Jin SM, Tanaka A, et al. PINK1 is selectively stabilized on impaired mitochondria to activate Parkin. PLoS Biol 2010; 8:e1000298
Rakovic A, Grünewald A, Seibler P, et al. Effect of endogenous mutant and wild-type PINK1 on Parkin in fibroblasts from Parkinson disease patients. Hum Mol Genet 2010; 19:3124–3137.
Vives-Bauza C, Zhou C, Huang Y, et al. PINK1-dependent recruitment of Parkin to mitochondria in mitophagy. Proc Natl Acad Sci USA 2010; 107:378–383.
Jin SM, Lazarou M, Wang C, Kane LA, Narendra DP, Youle RJ . Mitochondrial membrane potential regulates PINK1 import and proteolytic destabilization by PARL. J Cell Biol 2010; 191:933–942.
Lazarou M, Jin SM, Kane LA, Youle RJ . Role of PINK1 binding to the TOM complex and alternate intracellular membranes in recruitment and activation of the E3 ligase Parkin. Dev Cell 2012; 22:320–333.
Xiong H, Wang D, Chen L, et al. Parkin, PINK1, and DJ-1 form a ubiquitin E3 ligase complex promoting unfolded protein degradation. J Clin Invest 2009; 119:650–660.
Okatsu K, Oka T, Iguchi M, et al. PINK1 autophosphorylation upon membrane potential dissipation is essential for Parkin recruitment to damaged mitochondria. Nat Commun 2012; 3:1016.
Kondapalli C, Kazlauskaite A, Zhang N, et al. PINK1 is activated by mitochondrial membrane potential depolarization and stimulates Parkin E3 ligase activity by phosphorylating Serine 65. Open Biol 2012; 2:120080.
Shiba-Fukushima K, Imai Y, Yoshida S, et al. PINK1-mediated phosphorylation of the Parkin ubiquitin-like domain primes mitochondrial translocation of Parkin and regulates mitophagy. Sci Rep 2012; 2:1002.
Deshaies RJ, Joazeiro CA . RING domain E3 ubiquitin ligases. Annu Rev Biochem 2009; 78:399–434.
Wenzel DM, Lissounov A, Brzovic PS, Klevit RE . UBCH7 reactivity profile reveals parkin and HHARI to be RING/HECT hybrids. Nature 2011; 474:105–108.
Smit JJ, Monteferrario D, Noordermeer SM, van Dijk WJ, van der Reijden BA, Sixma TK . The E3 ligase HOIP specifies linear ubiquitin chain assembly through its RING-IBR-RING domain and the unique LDD extension. EMBO J 2012; 31:3833–3844.
Stieglitz B, Morris-Davies AC, Koliopoulos MG, Christodoulou E, Rittinger K . LUBAC synthesizes linear ubiquitin chains via a thioester intermediate. EMBO Rep 2012; 13:840–846.
Hristova VA, Beasley SA, Rylett RJ, Shaw GS . Identification of a novel Zn2+-binding domain in the autosomal recessive juvenile Parkinson-related E3 ligase parkin. J Biol Chem 2009; 284:14978–14986.
Wenzel DM, Klevit RE . Following Ariadne's thread: a new perspective on RBR ubiquitin ligases. BMC Biol 2012; 10:24.
Chaugule VK, Burchell L, Barber KR, et al. Autoregulation of Parkin activity through its ubiquitin-like domain. EMBO J 2011; 30:2853–2867.
Matsuda N, Kitami T, Suzuki T, Mizuno Y, Hattori N, Tanaka K . Diverse effects of pathogenic mutations of Parkin that catalyze multiple monoubiquitylation in vitro. J Biol Chem 2006; 281:3204–3209.
Maruyama M, Ikeuchi T, Saito M, et al. Novel mutations, pseudo-dominant inheritance, and possible familial affects in patients with autosomal recessive juvenile parkinsonism. Ann Neurol 2000; 48:245–250.
Ye Y, Rape M . Building ubiquitin chains: E2 enzymes at work. Nat Rev Mol Cell Biol. 2009; 10:755–764.
Chan NC, Salazar AM, Pham AH, et al. Broad activation of the ubiquitin-proteasome system by Parkin is critical for mitophagy. Hum Mol Genet 2011; 20:1726–1737.
Husnjak K, Dikic I . Ubiquitin-binding proteins: decoders of ubiquitin-mediated cellular functions. Annu Rev Biochem 2012; 81:291–322.
Wang X, Herr RA, Chua WJ, Lybarger L, Wiertz EJ, Hansen TH . Ubiquitination of serine, threonine, or lysine residues on the cytoplasmic tail can induce ERAD of MHC-I by viral E3 ligase mK3. J Cell Biol 2007; 177:613–624.
Kim HC, Steffen AM, Oldham ML, Chen J, Huibregtse JM . Structure and function of a HECT domain ubiquitin-binding site. EMBO Rep 2011; 12:334–341.
French ME, Kretzmann BR, Hicke L . Regulation of the RSP5 ubiquitin ligase by an intrinsic ubiquitin-binding site. J Biol Chem 2009; 284:12071–12079.
Komander D, Reyes-Turcu F, Licchesi JD, Odenwaelder P, Wilkinson KD, Barford D . Molecular discrimination of structurally equivalent Lys 63-linked and linear polyubiquitin chains. EMBO Rep 2009; 10:466–473.
Schlossmacher MG, Frosch MP, Gai WP, et al. Parkin localizes to the Lewy bodies of Parkinson disease and dementia with Lewy bodies. Am J Pathol 2002; 160:1655–1667.
Lazarou M, Narendra DP, Jin SM, Tekle E, Banerjee S, Youle RJ . PINK1 drives Parkin self-association and HECT-like E3 activity upstream of mitochondrial binding. J Cell Biol 2013; 200:163–172.
Sarraf SA, Raman M, Guarani-Pereira V, et al. Landscape of the PARKIN-dependent ubiquitylome in response to mitochondrial depolarization. Nature 2013; 496:372–376.
Müller-Rischart AK, Pilsl A, Beaudette P, et al. The E3 ligase Parkin maintains mitochondrial integrity by increasing linear ubiquitination of NEMO. Mol Cell 2013; 49:908–921.
Acknowledgements
We thank Dr Zhuohua Zhang (Sanford-Burnham Medical Research Institute, USA) for Pink1-null and WT MEFs, and Dr Richard Youle (NINDS, NIH, USA) for helpful discussions. We thank other members of the Hunter lab for helpful discussions, Suzy Simon for assistance in picking MGC cDNA clones, and Jill Meisenhelder and Justin Zimmerman for laboratory support. This study was supported by NIH grants (CA14195, CA80100 and CA82683) to TH. TH is a Frank and Else Schilling American Cancer Society Professor, and the Renato Dulbecco Chair in Cancer Biology. XZ is supported by fellowship from California Institute of Regenerative Medicine.
Author information
Authors and Affiliations
Corresponding authors
Additional information
( Supplementary information is linked to the online version of the paper on the Cell Research website.)
Supplementary information
Supplementary information, Figure S1
(A) Mutation of the putative catalytic cysteine 431 to alanine in the Parkin RBR domain abrogated mitochondrial translocation and clearance. (PDF 158 kb)
Rights and permissions
About this article
Cite this article
Zheng, X., Hunter, T. Parkin mitochondrial translocation is achieved through a novel catalytic activity coupled mechanism. Cell Res 23, 886–897 (2013). https://doi.org/10.1038/cr.2013.66
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/cr.2013.66
Keywords
This article is cited by
-
Mitochondrial network remodeling: an important feature of myogenesis and skeletal muscle regeneration
Cellular and Molecular Life Sciences (2021)
-
Inflammatory injury and mitophagy of the brain in chicken exposed to Cr(VI)
Environmental Science and Pollution Research (2021)
-
Mechanisms of PINK1, ubiquitin and Parkin interactions in mitochondrial quality control and beyond
Cellular and Molecular Life Sciences (2019)
-
Building and decoding ubiquitin chains for mitophagy
Nature Reviews Molecular Cell Biology (2018)
-
Parkin–phosphoubiquitin complex reveals cryptic ubiquitin-binding site required for RBR ligase activity
Nature Structural & Molecular Biology (2017)