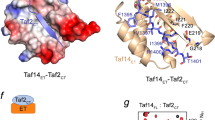
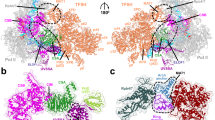
Abstract
The general transcription factor IID (TFIID) initiates RNA polymerase II-mediated eukaryotic transcription by nucleating pre-initiation complex formation at the core promoter of protein-encoding genes. TAF1, the largest integral subunit of TFIID, contains an evolutionarily conserved yet poorly characterized central core domain, whose specific mutation disrupts cell proliferation in the temperature-sensitive mutant hamster cell line ts13. Although the impaired TAF1 function in the ts13 mutant has been associated with defective transcriptional regulation of cell cycle genes, the mechanism by which TAF1 mediates transcription as part of TFIID remains unclear. Here, we present the crystal structure of the human TAF1 central core domain in complex with another conserved TFIID subunit, TAF7, which biochemically solubilizes TAF1. The TAF1-TAF7 complex displays an inter-digitated compact architecture, featuring an unexpected TAF1 winged helix (WH) domain mounted on top of a heterodimeric triple barrel. The single TAF1 residue altered in the ts13 mutant is buried at the junction of these two structural domains. We show that the TAF1 WH domain has intrinsic DNA-binding activity, which depends on characteristic residues that are commonly used by WH fold proteins for interacting with DNA. Importantly, mutations of these residues not only compromise DNA binding by TAF1, but also abrogate its ability to rescue the ts13 mutant phenotype. Together, our results resolve the structural organization of the TAF1-TAF7 module in TFIID and unveil a critical promoter-binding function of TAF1 in transcription regulation.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Accession codes
References
Thomas MC, Chiang CM . The general transcription machinery and general cofactors. Crit Rev Biochem Mol Biol 2006; 41:105–178.
Grünberg S, Hahn S . Structural insights into transcription initiation by RNA polymerase II. Trends Biochem Sci 2013; 38:603–611.
Buratowski S, Hahn S, Guarente L, Sharp PA . Five intermediate complexes in transcription initiation by RNA polymerase II. Cell 1989; 56:549–561.
Van Dyke MW, Roeder RG, Sawadogo M . Physical analysis of transcription preinitiation complex assembly on a class II gene promoter. Science 1988; 241:1335–1338.
Papai G, Weil PA, Schultz P . New insights into the function of transcription factor TFIID from recent structural studies. Curr Opin Genet Dev 2011; 21:219–224.
Pugh BF, Tjian R . Transcription from a TATA-less promoter requires a multisubunit TFIID complex. Genes Dev 1991; 5:1935–1945.
Tanese N, Pugh BF, Tjian R . Coactivators for a proline-rich activator purified from the multisubunit human TFIID complex. Genes Dev 1991; 5:2212–2224.
Wassarman DA, Sauer F . TAF(II)250: a transcription toolbox. J Cell Sci 2001; 114:2895–2902.
Mizzen CA, Yang XJ, Kokubo T, et al. The TAF(II)250 subunit of TFIID has histone acetyltransferase activity. Cell 1996; 87:1261–1270.
Dikstein R, Ruppert S, Tjian R . TAFII250 is a bipartite protein kinase that phosphorylates the base transcription factor RAP74. Cell 1996; 84:781–790.
Ruppert S, Tjian R . Human TAFII250 interacts with RAP74: implications for RNA polymerase II initiation. Genes Dev 1995; 9:2747–2755.
Jacobson RH, Ladurner AG, King DS, Tjian R . Structure and function of a human TAFII250 double bromodomain module. Science 2000; 288:1422–1425.
Cianfrocco MA, Kassavetis GA, Grob P, et al. Human TFIID binds to core promoter DNA in a reorganized structural state. Cell 2013; 152:120–131.
Hisatake K, Hasegawa S, Takada R, Nakatani Y, Horikoshi M, Roeder RG . The p250 subunit of native TATA box-binding factor TFIID is the cell-cycle regulatory protein CCG1. Nature 1993; 362:179–181.
Ruppert S, Wang EH, Tjian R . Cloning and expression of human TAFII250: a TBP-associated factor implicated in cell-cycle regulation. Nature 1993; 362:175–179.
Hayashida T, Sekiguchi T, Noguchi E, Sunamoto H, Ohba T, Nishimoto T . The CCG1/TAFII250 gene is mutated in thermosensitive G1 mutants of the BHK21 cell line derived from golden hamster. Gene 1994; 141:267–270.
Hilton TL, Li Y, Dunphy EL, Wang EH . TAF1 histone acetyltransferase activity in Sp1 activation of the cyclin D1 promoter. Mol Cell Biol 2005; 25:4321–4332.
Chiang CM, Roeder RG . Cloning of an intrinsic human TFIID subunit that interacts with multiple transcriptional activators. Science 1995; 267:531–536.
Lavigne AC, Mengus G, May M, et al. Multiple interactions between hTAFII55 and other TFIID subunits. Requirements for the formation of stable ternary complexes between hTAFII55 and the TATA-binding protein. J Biol Chem 1996; 271:19774–19780.
Bhattacharya S, Lou X, Hwang P, et al. Structural and functional insight into TAF1-TAF7, a subcomplex of transcription factor II D. Proc Natl Acad Sci USA 2014; 111:9103–9108.
Weissman JD, Brown JA, Howcroft TK, et al. HIV-1 tat binds TAFII250 and represses TAFII250-dependent transcription of major histocompatibility class I genes. Proc Natl Acad Sci USA 1998; 95:11601–11606.
Dunphy EL, Johnson T, Auerbach SS, Wang EH . Requirement for TAF(II)250 acetyltransferase activity in cell cycle progression. Mol Cell Biol 2000; 20:1134–1139.
Gaiser F, Tan S, Richmond TJ . Novel dimerization fold of RAP30/RAP74 in human TFIIF at 1.7 Å resolution. J Mol Biol 2000; 302:1119–1127.
Shaban NM, Harvey S, Perrino FW, Hollis T . The structure of the mammalian RNase H2 complex provides insight into RNA.NA hybrid processing to prevent immune dysfunction. J Biol Chem 2010; 285:3617–3624.
Geiger SR, Lorenzen K, Schreieck A, et al. RNA polymerase I contains a TFIIF-related DNA-binding subcomplex. Mol Cell 2010; 39:583–594.
Taylor NM, Baudin F, von Scheven G, Müller CW . RNA polymerase III-specific general transcription factor IIIC contains a heterodimer resembling TFIIF Rap30/Rap74. Nucleic Acids Res 2013; 41:9183–9196.
Teichmann M, Dumay-Odelot H, Fribourg S . Structural and functional aspects of winged-helix domains at the core of transcription initiation complexes. Transcription 2012; 3:2–7.
Gajiwala KS, Burley SK . Winged helix proteins. Curr Opin Struct Biol 2000; 10:110–116.
Zheng N, Fraenkel E, Pabo CO, Pavletich NP . Structural basis of DNA recognition by the heterodimeric cell cycle transcription factor E2F-DP. Genes Dev 1999; 13:666–674.
Lee DH, Gershenzon N, Gupta M, Ioshikhes IP, Reinberg D, Lewis BA . Functional characterization of core promoter elements: the downstream core element is recognized by TAF1. Mol Cell Biol 2005; 25:9674–9686.
Chalkley GE, Verrijzer CP . DNA binding site selection by RNA polymerase II TAFs: a TAF(II)250-TAF(II)150 complex recognizes the initiator. EMBO J 1999; 18:4835–4845.
Juven-Gershon T, Cheng S, Kadonaga JT . Rational design of a super core promoter that enhances gene expression. Nat Methods 2006; 3:917–922.
Zhao S, Choi M, Overton JD, et al. Landscape of somatic single-nucleotide and copy-number mutations in uterine serous carcinoma. Proc Natl Acad Sci USA 2013; 110:2916–2921.
Kloet SL, Whiting JL, Gafken P, Ranish J, Wang EH . Phosphorylation-dependent regulation of cyclin D1 and cyclin A gene transcription by TFIID subunits TAF1 and TAF7. Mol Cell Biol 2012; 32:3358–3369.
Gegonne A, Weissman JD, Singer DS . TAFII55 binding to TAFII250 inhibits its acetyltransferase activity. Proc Natl Acad Sci USA 2001; 98:12432–12437.
Devaiah BN, Lu H, Gegonne A, et al. Novel functions for TAF7, a regulator of TAF1-independent transcription. J Biol Chem 2010; 285:38772–38780.
Marmorstein R, Trievel RC . Histone modifying enzymes: structures, mechanisms, and specificities. Biochim Biophys Acta 2009; 1789:58–68.
Otwinowski Z, Minor W, eds. Processing of X-ray Diffraction Data Collected in Oscillation Mode. New York: Academic Press 1997.
Adams PD, Grosse-Kunstleve RW, Hung LW, et al. PHENIX: building new software for automated crystallographic structure determination. Acta Crystallogr D Biol Crystallogr 2002; 58:1948–1954.
CCP4. The CCP4 Suite: programs for protein crystallography. Acta Crystallogr D Biol Crystallogr 1994; D50:760–763.
Acknowledgements
We thank the beam line staff of the Advanced Light Source at the University of California at Berkeley for help with data collection, undergraduates E Larson and M Kufeld for assistance with the in vivo complementation experiments, and members of the Zheng and Wang laboratories for discussion. This work is supported by the Howard Hughes Medical Institute (NZ) and the University of Washington Royalty Research Fund (EHW).
Author information
Authors and Affiliations
Corresponding authors
Additional information
( Supplementary information is linked to the online version of the paper on the Cell Research website.)
Supplementary information
Supplementary information, Figure S1
Sequence alignment of representative eukaryotic TAF7 orthologs annotated with secondary structures. (PDF 160 kb)
Supplementary information, Figure S2
Sequence alignment of DUF3591 domains of representative eukaryotic TAF1 orthologs annotated with secondary structure labeled. (PDF 346 kb)
Supplementary information, Figure S3
Two salt bridges formed among three TAF7 β-hairpin residues are buried in the TAF1 barrel. (PDF 634 kb)
Supplementary information, Figure S4
A close-up view of the C-terminal residues in the TAF1 DUF3591 fragment together with positive Fo-Fc electron density contoured at 3.0 σ (red) and 2Fo-Fc electron density contoured at 1.0 σ (blue) calculated before the phosphate groups were built. (PDF 1170 kb)
Supplementary information, Figure S5
Structure comparison between the yeast HAT enzyme ESA1 and TAF1 in complex with TAF7. (PDF 1719 kb)
Supplementary information, Table S1
Summary of crystallographic information (PDF 114 kb)
Supplementary information, Table S2
Nucleic acid sequences of the probes used in EMSA analysis and the primers for the site mutagenesis (PDF 44 kb)
Rights and permissions
About this article
Cite this article
Wang, H., Curran, E., Hinds, T. et al. Crystal structure of a TAF1-TAF7 complex in human transcription factor IID reveals a promoter binding module. Cell Res 24, 1433–1444 (2014). https://doi.org/10.1038/cr.2014.148
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/cr.2014.148
Keywords
This article is cited by
-
Nonlinear feature selection using sparsity-promoted centroid-encoder
Neural Computing and Applications (2023)
-
TAF1 plays a critical role in AML1-ETO driven leukemogenesis
Nature Communications (2019)
-
TAF1, associated with intellectual disability in humans, is essential for embryogenesis and regulates neurodevelopmental processes in zebrafish
Scientific Reports (2019)
-
Identification of the gene encoding the TATA box-binding protein-associated factor 1 (TAF1) and its putative role in the heat shock response in the protozoan parasite Entamoeba histolytica
Parasitology Research (2019)
-
Zinc knuckle of TAF1 is a DNA binding module critical for TFIID promoter occupancy
Scientific Reports (2018)