Abstract
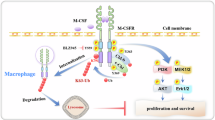
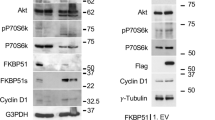
Macrophages play pivotal roles in development, homeostasis, tissue repair and immunity. Macrophage proliferation is promoted by macrophage colony-stimulating factor (M-CSF)-induced Akt signaling; yet, how this process is terminated remains unclear. Here, we identify casein kinase 2-interacting protein-1 (CKIP-1) as a novel inhibitor of macrophage proliferation. In resting macrophages, CKIP-1 was phosphorylated at Serine 342 by constitutively active GSK3β, the downstream target of Akt. This phosphorylation triggers the polyubiquitination and proteasomal degradation of CKIP-1. Upon M-CSF stimulation, Akt is activated by CSF-1R-PI3K and then inactivates GSK3β, leading to the stabilization of CKIP-1 and β-catenin proteins. β-catenin promotes the expression of proliferation genes including cyclin D and c-Myc. CKIP-1 interacts with TRAF6, a ubiquitin ligase required for K63-linked ubiquitination and plasma membrane recruitment of Akt, and terminates TRAF6-mediated Akt activation. By this means, CKIP-1 inhibits macrophage proliferation specifically at the late stage after M-CSF stimulation. Furthermore, CKIP-1 deficiency results in increased proliferation and decreased apoptosis of macrophages in vitro and CKIP-1−/− mice spontaneously develop a macrophage-dominated splenomegaly and myeloproliferation. Together, these data demonstrate that CKIP-1 plays a critical role in the regulation of macrophage homeostasis by inhibiting TRAF6-mediated Akt activation.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Martinez FO, Helming L, Gordon S . Alternative activation of macrophages: an immunologic functional perspective. Annu Rev Immunol 2009; 27:451–483.
Hamilton JA . Colony-stimulating factors in inflammation and autoimmunity. Nat Rev Immunol 2008; 8:533–544.
Chitu V, Stanley ER . Colony-stimulating factor-1 in immunity and inflammation. Curr Opin Immunol 2006; 18:39–48.
Stanley ER, Berg KL, Einstein DB, et al. Biology and action of colony-stimulating factor-1. Mol Reprod Dev 1997; 46:4–10.
Wiktor-Jedrzejczak W, Ratajczak MZ, Ptasznik A, Sell KW, Ahmed-Ansari A, Ostertag W . CSF-1 deficiency in the op/op mouse has differential effects on macrophage populations and differentiation stages. Exp Hematol 1992; 20:1004–1010.
Hamilton JA . CSF-1 signal transduction. J Leukoc Biol 1997; 62:145–155.
Pixley FJ, Stanley ER . CSF-1 regulation of the wandering macrophage: complexity in action. Trends Cell Biol 2004; 14:628–638.
Lee AW, States DJ . Colony-stimulating factor-1 requires PI3-kinase-mediated metabolism for proliferation and survival in myeloid cells. Cell Death Differ 2006; 13:1900–1914.
Smith JL, Schaffner AE, Hofmeister JK, et al. ets-2 is a target for an akt (protein kinase B)/jun N-terminal kinase signaling pathway in macrophages of motheaten-viable mutant mice. Mol Cell Biol 2000; 20:8026–8034.
Lee AW . Synergistic activation of mitogen-activated protein kinase by cyclic AMP and myeloid growth factors opposes cyclic AMP's growth-inhibitory effects. Blood 1999; 93:537–553.
Manning BD, Cantley LC . AKT/PKB signaling: navigating downstream. Cell 2007; 129:1261–1274.
Varticovski L, Druker B, Morrison D, Cantley L, Roberts T . The colony stimulating factor-1 receptor associates with and activates phosphatidylinositol-3 kinase. Nature 1989; 342:699–702.
Datta SR, Brunet A, Greenberg ME . Cellular survival: a play in three Akts. Genes Dev 1999; 13:2905–2927.
Sampaio NG, Yu W, Cox D, et al. Phosphorylation of CSF-1R Y721 mediates its association with PI3K to regulate macrophage motility and enhancement of tumor cell invasion. J Cell Sci 2011; 124:2021–2031.
Otero K, Turnbull IR, Poliani PL, et al. Macrophage colony-stimulating factor induces the proliferation and survival of macrophages via a pathway involving DAP12 and beta-catenin. Nat Immunol 2009; 10:734–743.
Zhang L, Xing G, Tie Y, et al. Role for the pleckstrin homology domain-containing protein CKIP-1 in AP-1 regulation and apoptosis. EMBO J 2005; 24:766–778.
Bosc DG, Graham KC, Saulnier RB, et al. Identification and characterization of CKIP-1, a novel pleckstrin homology domain-containing protein that interacts with protein kinase CK2. J Biol Chem 2000; 275:14295–14306.
Safi A, Vandromme M, Caussanel S, et al. Role for the pleckstrin homology domain-containing protein CKIP-1 in phosphatidylinositol 3-kinase-regulated muscle differentiation. Mol Cell Biol 2004; 24:1245–1255.
Canton DA, Olsten ME, Kim K, et al. The pleckstrin homology domain-containing protein CKIP-1 is involved in regulation of cell morphology and the actin cytoskeleton and interaction with actin capping protein. Mol Cell Biol 2005; 25:3519–3534.
Nie J, Liu L, He F, Fu X, Han W, Zhang L . CKIP-1: a scaffold protein and potential therapeutic target integrating multiple signaling pathways and physiological functions. Ageing Res Rev 2013; 12:276–281.
Lu K, Yin X, Weng T, et al. Targeting WW domains linker of HECT-type ubiquitin ligase Smurf1 for activation by CKIP-1. Nat Cell Biol 2008; 10:994–1002.
Ling S, Sun Q, Li Y, et al. CKIP-1 inhibits cardiac hypertrophy by regulating class II histone deacetylase phosphorylation through recruiting PP2A. Circulation 2012; 126:3028–3040.
Zhang L, Tang Y, Tie Y, et al. The PH domain containing protein CKIP-1 binds to IFP35 and Nmi and is involved in cytokine signaling. Cell Signal 2007; 19:932–944.
Juhasz K, Zvara A, Lipp AM, et al. Casein kinase 2-interacting protein-1, an inflammatory signaling molecule interferes with TNF reverse signaling in human model cells. Immunol Lett 2013; 152:55–64.
Klappacher GW, Lunyak VV, Sykes DB, et al. An induced Ets repressor complex regulates growth arrest during terminal macrophage differentiation. Cell 2002; 109:169–180.
Yang WL, Wang J, Chan CH, et al. The E3 ligase TRAF6 regulates Akt ubiquitination and activation. Science 2009; 325:1134–1138.
Lomaga MA, Yeh WC, Sarosi I, et al. TRAF6 deficiency results in osteopetrosis and defective interleukin-1, CD40, and LPS signaling. Genes Dev 1999; 13:1015–1024.
Kobayashi N, Kadono Y, Naito A, et al. Segregation of TRAF6-mediated signaling pathways clarifies its role in osteoclastogenesis. EMBO J 2001; 20:1271–1280.
Tokuda E, Fujita N, Oh-hara T, et al. Casein kinase 2-interacting protein-1, a novel Akt pleckstrin homology domain-interacting protein, down-regulates PI3K/Akt signaling and suppresses tumor growth in vivo. Cancer Res 2007; 67:9666–9676.
Wang Y, Tang Y, Teng L, Wu Y, Zhao X, Pei G . Association of beta-arrestin and TRAF6 negatively regulates toll-like receptor-interleukin 1 receptor signaling. Nat Immunol 2006; 7:139–147.
Vallabhapurapu S, Karin M . Regulation and function of NF-kappaB transcription factors in the immune system. Annu Rev Immunol 2009; 27:693–733.
Pagliari LJ, Perlman H, Liu H, Pope RM . Macrophages require constitutive NF-kappaB activation to maintain A1 expression and mitochondrial homeostasis. Mol Cell Biol 2000; 20:8855–8865.
Wang Y, Mo X, Piper MG, et al. M-CSF induces monocyte survival by activating NF-kappaB p65 phosphorylation at Ser276 via protein kinase C. PLoS One 2011; 6:e 28081.
Lee AW . The role of atypical protein kinase C in CSF-1-dependent Erk activation and proliferation in myeloid progenitors and macrophages. PLoS One 2011; 6:e 25580.
Frankenberger M, Pforte A, Sternsdorf T, Passlick B, Baeuerle PA, Ziegler-Heitbrock HW . Constitutive nuclear NF-kappa B in cells of the monocyte lineage. Biochem J 1994; 304 (Part 1):87–94.
Kuo AH, Stoica GE, Riegel AT, Wellstein A . Recruitment of insulin receptor substrate-1 and activation of NF-kappaB essential for midkine growth signaling through anaplastic lymphoma kinase. Oncogene 2007; 26:859–869.
Simoncic PD, Bourdeau A, Lee-Loy A, et al. T-cell protein tyrosine phosphatase (Tcptp) is a negative regulator of colony-stimulating factor 1 signaling and macrophage differentiation. Mol Cell Biol 2006; 26:4149–4160.
Wynn TA, Chawla A, Pollard JW . Macrophage biology in development, homeostasis and disease. Nature 2013; 496:445–455.
Teitelbaum SL, Ross FP . Genetic regulation of osteoclast development and function. Nat Rev Genet 2003; 4:638–649.
Cecchini MG, Dominguez MG, Mocci S, et al. Role of colony stimulating factor-1 in the establishment and regulation of tissue macrophages during postnatal development of the mouse. Development 1994; 120:1357–1372.
Ryan GR, Dai XM, Dominguez MG, et al. Rescue of the colony-stimulating factor 1 (CSF-1)-nullizygous mouse (Csf1(op)/Csf1(op)) phenotype with a CSF-1 transgene and identification of sites of local CSF-1 synthesis. Blood 2001; 98:74–84.
Yoshida H, Hayashi S, Kunisada T, et al. The murine mutation osteopetrosis is in the coding region of the macrophage colony stimulating factor gene. Nature 1990; 345:442–444.
Dai XM, Ryan GR, Hapel AJ, et al. Targeted disruption of the mouse colony-stimulating factor 1 receptor gene results in osteopetrosis, mononuclear phagocyte deficiency, increased primitive progenitor cell frequencies, and reproductive defects. Blood 2002; 99:111–120.
Naito A, Azuma S, Tanaka S, et al. Severe osteopetrosis, defective interleukin-1 signalling and lymph node organogenesis in TRAF6-deficient mice. Genes Cells 1999; 4:353–362.
Lamothe B, Webster WK, Gopinathan A, Besse A, Campos AD, Darnay BG . TRAF6 ubiquitin ligase is essential for RANKL signaling and osteoclast differentiation. Biochem Biophys Res Commun 2007; 359:1044–1049.
Armstrong AP, Tometsko ME, Glaccum M, Sutherland CL, Cosman D, Dougall WC . A RANK/TRAF6-dependent signal transduction pathway is essential for osteoclast cytoskeletal organization and resorptive function. J Biol Chem 2002; 277:44347–44356.
Bai S, Kitaura H, Zhao H, et al. FHL2 inhibits the activated osteoclast in a TRAF6-dependent manner. J Clin Invest 2005; 115:2742–2751.
Vremec D, Shortman K . Dendritic cell subtypes in mouse lymphoid organs: cross-correlation of surface markers, changes with incubation, and differences among thymus, spleen, and lymph nodes. J Immunol 1997; 159:565–573.
Shortman K, Caux C . Dendritic cell development: multiple pathways to nature's adjuvants. Stem Cells 1997; 15:409–419.
Vremec D, Pooley J, Hochrein H, Wu L, Shortman K . CD4 and CD8 expression by dendritic cell subtypes in mouse thymus and spleen. J Immunol 2000; 164:2978–2986.
Kobayashi T, Walsh PT, Walsh MC, et al. TRAF6 is a critical factor for dendritic cell maturation and development. Immunity 2003; 19:353–363.
MacDonald KP, Rowe V, Bofinger HM, et al. The colony-stimulating factor 1 receptor is expressed on dendritic cells during differentiation and regulates their expansion. J Immunol 2005; 175:1399–1405.
Chow A, Brown BD, Merad M . Studying the mononuclear phagocyte system in the molecular age. Nat Rev Immunol 2011; 11:788–798.
Cross DA, Alessi DR, Cohen P, Andjelkovich M, Hemmings BA . Inhibition of glycogen synthase kinase-3 by insulin mediated by protein kinase B. Nature 1995; 378:785–789.
Acknowledgements
We thank Drs Jiang Peng for RAW264.7 cell line, Changyan Li for lentiviral transduction, Hongbing Shu for TARF6 plasmid, Jiyan Zhang for BMDM cultures. This work was supported by Chinese National Basic Research Programs (2011CB910802, 2012CB910304), the National Key Technologies R&D Program for New Drugs (2014ZX09J14106-04C), Chinese National Natural Science Foundation Projects (31125010, 31270912 and 81221004) and a grant from State Key Laboratory of Proteomics (SKLP-K201102).
Author information
Authors and Affiliations
Corresponding authors
Additional information
( Supplementary information is linked to the online version of the paper on the Cell Research website.)
Supplementary information
Supplementary information, Figure S1
Normal differentiation of CKIP-1−/− BMDMs. (PDF 156 kb)
Supplementary information, Figure S2
CKIP-1 silencing augments M-CSF-induced Akt activation. (PDF 264 kb)
Supplementary information, Figure S3
TRAF6 is required for M-CSF-induced Akt activation. (PDF 307 kb)
Supplementary information, Figure S4
PI3K/Akt/GSK3β pathway controls CKIP-1 protein level in macrophage. (PDF 326 kb)
Rights and permissions
About this article
Cite this article
Zhang, L., Wang, Y., Xiao, F. et al. CKIP-1 regulates macrophage proliferation by inhibiting TRAF6-mediated Akt activation. Cell Res 24, 742–761 (2014). https://doi.org/10.1038/cr.2014.53
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/cr.2014.53
Keywords
This article is cited by
-
Regulation of Microglial Activation by Wnt/β-Catenin Signaling After Global Cerebral Ischemia in Mice
Molecular Neurobiology (2024)
-
IL-36α inhibits melanoma by inducing pro-inflammatory polarization of macrophages
Cancer Immunology, Immunotherapy (2023)
-
PLEKHO2 inhibits TNFα-induced cell death by suppressing RIPK1 activation
Cell Death & Disease (2021)
-
KDM4B facilitates colorectal cancer growth and glucose metabolism by stimulating TRAF6-mediated AKT activation
Journal of Experimental & Clinical Cancer Research (2020)
-
Upregulation of CKIP-1 inhibits high-glucose induced inflammation and oxidative stress in HRECs and attenuates diabetic retinopathy by modulating Nrf2/ARE signaling pathway: an in vitro study
Cell & Bioscience (2019)