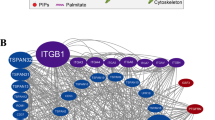
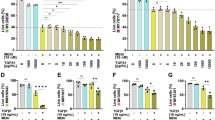
Abstract
In normal melanocytes, TGF-β signaling has a cytostatic effect. However, in primary melanoma cells, TGF-β-induced cytostasis is diminished, thus allowing melanoma growth. Later, a second phase of TGF-β signaling supports melanoma EMT-like changes, invasion and metastasis. In parallel with these “present-absent-present” TGF-β signaling phases, cell surface protein EWI motif-containing protein 2 (EWI-2 or IgSF8) is “absent-present-absent” in melanocytes, primary melanoma, and metastatic melanoma, respectively, suggesting that EWI-2 may serve as a negative regulator of TGF-β signaling. Using melanoma cell lines and melanoma short-term cultures, we performed RNAi and overexpression experiments and found that EWI-2 negatively regulates TGF-β signaling and its downstream events including cytostasis (in vitro and in vivo), EMT-like changes, cell migration, CD271-dependent invasion, and lung metastasis (in vivo). When EWI-2 is present, it associates with cell surface tetraspanin proteins CD9 and CD81 — molecules not previously linked to TGF-β signaling. Indeed, when associated with EWI-2, CD9 and CD81 are sequestered and have no impact on TβR2-TβR1 association or TGF-β signaling. However, when EWI-2 is knocked down, CD9 and CD81 become available to provide critical support for TβR2-TβR1 association, thus markedly elevating TGF-β signaling. Consequently, all of those TGF-β-dependent functions specifically arising due to EWI-2 depletion are reversed by blocking or depleting cell surface tetraspanin proteins CD9 or CD81. These results provide new insights into regulation of TGF-β signaling in melanoma, uncover new roles for tetraspanins CD9 and CD81, and strongly suggest that EWI-2 could serve as a favorable prognosis indicator for melanoma patients.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Flaherty KT . Targeting metastatic melanoma. Annu Rev Med 2012; 63:171–183.
Tsao H, Chin L, Garraway LA, Fisher DE . Melanoma: from mutations to medicine. Genes Dev 2012; 26:1131–1155.
Tuong W, Cheng LS, Armstrong AW . Melanoma: epidemiology, diagnosis, treatment, and outcomes. Dermatol Clin 2012; 30:113–124, ix.
Javelaud D, Alexaki VI, Mauviel A . Transforming growth factor-beta in cutaneous melanoma. Pigment Cell Melanoma Res 2008; 21:123–132.
Lasfar A, Cohen-Solal KA . Resistance to transforming growth factor beta-mediated tumor suppression in melanoma: are multiple mechanisms in place? Carcinogenesis 2010; 31:1710–1717.
Perrot CY, Javelaud D, Mauviel A . Insights into the transforming growth factor-beta signaling pathway in cutaneous melanoma. Ann Dermatol 2013; 25:135–144.
Stipp CS, Kolesnikova TV, Hemler ME . EWI-2 is a major CD9 and CD81 partner, and member of a novel Ig protein subfamily. J Biol Chem 2001; 276:40545–40554.
Charrin S, Le Naour F, Labas V, et al. EWI-2 is a new component of the tetraspanin web in hepatocytes and lymphoid cells. Biochem J 2003; 373(Part 2):409–421.
Clark KL, Zeng Z, Langford AL, Bowen SM, Todd SC . Pgrl is a major CD81-associated protein on lymphocytes and distinguishes a new family of cell surface proteins. J Immunol 2001; 167:5115–5121.
Kolesnikova TV, Stipp CS, Rao RM, Lane WS, Luscinskas FW, Hemler ME . EWI-2 modulates lymphocyte integrin alpha4beta1 functions. Blood 2004; 103:3013–3019.
Yang XH, Kovalenko OV, Kolesnikova TV, et al. Contrasting Effects of EWI proteins, integrins, and protein palmitoylation on cell surface CD9 organization. J Biol Chem 2006; 281:12976–12985.
Fan J, Zhu GZ, Niles RM . Expression and function of CD9 in melanoma cells. Mol Carcinog 2010; 49:85–93.
Longo N, Yanez-Mo M, Mittelbrunn M, et al. Regulatory role of tetraspanin CD9 in tumor-endothelial cell interaction during transendothelial invasion of melanoma cells. Blood 2001; 98:3717–3726.
Hong IK, Byun HJ, Lee J, et al. The tetraspanin CD81 protein increases melanoma cell motility by up-regulating metalloproteinase MT1-MMP expression through the pro-oncogenic Akt-dependent Sp1 activation signaling pathways. J Biol Chem 2014; 289:15691–15704.
Mischiati C, Natali PG, Sereni A, et al. cDNA-array profiling of melanomas and paired melanocyte cultures. J Cell Physiol 2006; 207:697–705.
Si Z, Hersey P . Expression of the neuroglandular antigen and analogues in melanoma. CD9 expression appears inversely related to metastatic potential of melanoma. Int J Cancer 1993; 54:37–43.
Stipp CS, Kolesnikova TV, Hemler ME . EWI-2 regulates α3β1 integrin-dependent cell functions on laminin-5. J Cell Biol 2003; 163:1167–1177.
Kolesnikova TV, Kazarov AR, Lemieux ME, et al. Glioblastoma inhibition by cell surface immunoglobulin protein EWI-2, in vitro and in vivo. Neoplasia 2009; 11:77–86.
Rocha-Perugini V, Montpellier C, Delgrange D, et al. The CD81 partner EWI-2wint inhibits hepatitis C virus entry. PLoS One 2008; 3:e1866.
Glazar AI, Evans JP . Immunoglobulin superfamily member IgSF8 (EWI-2) and CD9 in fertilisation: evidence of distinct functions for CD9 and a CD9-associated protein in mammalian sperm-egg interaction. Reprod Fertil Dev 2009; 21:293–303.
Lin WM, Baker AC, Beroukhim R, et al. Modeling genomic diversity and tumor dependency in malignant melanoma. Cancer Res 2008; 68:664–673.
Jakowlew SB . Transforming growth factor-beta in cancer and metastasis. Cancer Metastasis Rev 2006; 25:435–457.
Scherf U, Ross DT, Waltham M, et al. A gene expression database for the molecular pharmacology of cancer. Nat Genet 2000; 24:236–244.
Stipp CS, Orlicky D, Hemler ME . FPRP: A major, highly stoichiometric, highly specific CD81 and CD9-associated protein. J Biol Chem 2001; 276:4853–4862.
Gaggioli C, Sahai E . Melanoma invasion — current knowledge and future directions. Pigment Cell Res 2007; 20:161–172.
Wrighton KH, Lin X, Feng XH . Phospho-control of TGF-beta superfamily signaling. Cell Res 2009; 19:8–20.
Lonn P, Moren A, Raja E, Dahl M, Moustakas A . Regulating the stability of TGFbeta receptors and Smads. Cell Res 2009; 19:21–35.
Akhurst RJ, Hata A . Targeting the TGFbeta signalling pathway in disease. Nat Rev Drug Discov 2012; 11:790–811.
Massague J . TGFbeta signalling in context. Nat Rev Mol Cell Biol 2012; 13:616–630.
Laping NJ, Grygielko E, Mathur A, et al. Inhibition of transforming growth factor (TGF)-beta1-induced extracellular matrix with a novel inhibitor of the TGF-beta type I receptor kinase activity: SB-431542. Mol Pharmacol 2002; 62:58–64.
Huber MA, Kraut N, Beug H . Molecular requirements for epithelial-mesenchymal transition during tumor progression. Curr Opin Cell Biol 2005; 17:548–558.
Qiao B, Johnson NW, Gao J . Epithelial-mesenchymal transition in oral squamous cell carcinoma triggered by transforming growth factor-beta1 is Snail family-dependent and correlates with matrix metalloproteinase-2 and -9 expressions. Int J Oncol 2010; 37:663–668.
Civenni G, Walter A, Kobert N, et al. Human CD271-positive melanoma stem cells associated with metastasis establish tumor heterogeneity and long-term growth. Cancer Res 2011; 71:3098–3109.
Boiko AD, Razorenova OV, van de Rijn M, et al. Human melanoma-initiating cells express neural crest nerve growth factor receptor CD271. Nature 2010; 466:133–137.
Bakin AV, Tomlinson AK, Bhowmick NA, Moses HL, Arteaga CL . Phosphatidylinositol 3-kinase function is required for transforming growth factor beta-mediated epithelial to mesenchymal transition and cell migration. J Biol Chem 2000; 275:36803–36810.
Thannickal VJ, Lee DY, White ES, et al. Myofibroblast differentiation by transforming growth factor-beta1 is dependent on cell adhesion and integrin signaling via focal adhesion kinase. J Biol Chem 2003; 278:12384–12389.
Nguyen DX, Bos PD, Massague J . Metastasis: from dissemination to organ-specific colonization. Nat Rev Cancer 2009; 9:274–284.
Schmalhofer O, Brabletz S, Brabletz T . E-cadherin, beta-catenin, and ZEB1 in malignant progression of cancer. Cancer Metastasis Rev 2009; 28:151–166.
Giampieri S, Manning C, Hooper S, Jones L, Hill CS, Sahai E . Localized and reversible TGFbeta signalling switches breast cancer cells from cohesive to single cell motility. Nat Cell Biol 2009; 11:1287–1296.
Orgaz JL, Sanz-Moreno V . Emerging molecular targets in melanoma invasion and metastasis. Pigment Cell Melanoma Res 2013; 26:39–57.
Arozarena I, Bischof H, Gilby D, Belloni B, Dummer R, Wellbrock C . In melanoma, beta-catenin is a suppressor of invasion. Oncogene 2011; 30:4531–4543.
Kovalenko OV, Yang XH, Hemler ME . A novel cysteine cross-linking method reveals a direct association between claudin-1 and tetraspanin CD9. Mol Cell Proteomics 2007; 6:1855–1867.
Le Naour F, Andre M, Greco C, et al. Profiling of the tetraspanin web of human colon cancer cells. Mol Cell Proteomics 2006; 5:845–857.
Miyake M, Inufusa H, Adachi M, et al. Suppression of pulmonary metastasis using adenovirally motility related protein-1 (MRP-1/CD9) gene delivery. Oncogene 2000; 19:5221–5226.
Oren R, Takahashi S, Doss C, Levy R, Levy S . TAPA-1, the target of an antiproliferative antibody, defines a new family of transmembrane proteins. Mol Cell Biol 1990; 10:4007–4015.
Inui S, Higashiyama S, Hashimoto K, Higashiyama M, Yoshikawa K, Taniguchi N . Possible role of coexpression of CD9 with membrane-anchored heparin-binding EGF-like growth factor and amphiregulin in cultured human keratinocyte growth. J Cell Physiol 1997; 171:291–298.
Romanska HM, Berditchevski F . Tetraspanins in human epithelial malignancies. J Pathol 2011; 223:4–14.
Zoller M . Tetraspanins: push and pull in suppressing and promoting metastasis. Nat Rev Cancer 2009; 9:40–55.
Rubinstein E, Ziyyat A, Wolf JP, Le Naour F, Boucheix C . The molecular players of sperm-egg fusion in mammals. Semin Cell Dev Biol 2006; 17:254–263.
Reich M, Liefeld T, Gould J, Lerner J, Tamayo P, Mesirov JP . GenePattern 2.0. Nat Genet 2006; 38:500–501.
Acknowledgements
This work was supported by the National Institutes of Health (CA42368 to MEH).
Author information
Authors and Affiliations
Corresponding author
Additional information
( Supplementary information is linked to the online version of the paper on the Cell Research website.)
Supplementary information
Supplementary information, Figure S1
EWI-2 expression in melanoma patient tissue and melanoma short term cultures (MSTC's). (PDF 422 kb)
Supplementary information, Figure S2
EWI-2 knockdown is associated with irregular tumor xenograft borders. (PDF 271 kb)
Supplementary information, Figure S3
Demonstration of RNAi knockdown efficiencies. (PDF 119 kb)
Supplementary information, Figure S4
Changes in cell morphology associated with EWI-2 knockdown. (PDF 121 kb)
Supplementary information, Figure S5
Additional evidence that EWI-2 supports melanoma cell growth. (PDF 92 kb)
Supplementary information, Figure S6
Effects of EWI-2 ablation on melanoma cell gene expression, TGF-β receptor expression, and TGF-β1 sensitivity. (PDF 300 kb)
Supplementary information, Figure S7
Smad2 knockdown reverses effects of EWI-2 ablation. (PDF 290 kb)
Supplementary information, Figure S8
Test of SB-431542 specificity. (PDF 159 kb)
Supplementary information, Figure S9
Supporting evidence for CD9 and CD81 interactions with EWI-2. (PDF 101 kb)
Supplementary information, Table S1
Genes in SK-Mel-28 cells most affected by EWI-2 (PDF 58 kb)
Supplementary information, Table S2
A partial list of genes, regulated through Smad2/3, that are upregulated in EWI-2 knockdown cells (PDF 10 kb)
Rights and permissions
About this article
Cite this article
Wang, HX., Sharma, C., Knoblich, K. et al. EWI-2 negatively regulates TGF-β signaling leading to altered melanoma growth and metastasis. Cell Res 25, 370–385 (2015). https://doi.org/10.1038/cr.2015.17
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/cr.2015.17
Keywords
This article is cited by
-
SET8 is a novel negative regulator of TGF-β signaling in a methylation-independent manner
Scientific Reports (2023)
-
EWI2 and its relatives in Tetraspanin-enriched membrane domains regulate malignancy
Oncogene (2023)
-
Immunoglobulin superfamily member 8 maintains myeloid leukemia stem cells through inhibition of β-catenin degradation
Leukemia (2022)
-
The Hippo pathway links adipocyte plasticity to adipose tissue fibrosis
Nature Communications (2022)
-
EWI2 prevents EGFR from clustering and endocytosis to reduce tumor cell movement and proliferation
Cellular and Molecular Life Sciences (2022)