Abstract
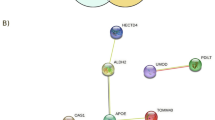
We performed gene–environment interaction genome-wide association analysis (G × E GWAS) to identify SNPs whose effects on metabolic traits are modified by chronic psychosocial stress in the Multi-Ethnic Study of Atherosclerosis (MESA). In Whites, the G × E GWAS for hip circumference identified five SNPs within the Early B-cell Factor 1 (EBF1) gene, all of which were in strong linkage disequilibrium. The gene-by-stress interaction (SNP × STRESS) term P-values were genome-wide significant (Ps=7.14E−09 to 2.33E−08, uncorrected; Ps=1.99E−07 to 5.18E−07, corrected for genomic control). The SNP-only (without interaction) model P-values (Ps=0.011–0.022) were not significant at the conventional genome-wide significance level. Further analysis of related phenotypes identified gene-by-stress interaction effects for waist circumference, body mass index (BMI), fasting glucose, type II diabetes status, and common carotid intimal–medial thickness (CCIMT), supporting a proposed model of gene-by-stress interaction that connects cardiovascular disease (CVD) risk factor endophenotypes such as central obesity and increased blood glucose or diabetes to CVD itself. Structural equation path analysis suggested that the path from chronic psychosocial stress to CCIMT via hip circumference and fasting glucose was larger (estimate=0.26, P=0.033, 95% CI=0.02–0.49) in the EBF1 rs4704963 CT/CC genotypes group than the same path in the TT group (estimate=0.004, P=0.34, 95% CI=−0.004–0.012). We replicated the association of the EBF1 SNPs and hip circumference in the Framingham Offspring Cohort (gene-by-stress term P-values=0.007–0.012) as well as identified similar path relationships. This observed and replicated interaction between psychosocial stress and variation in the EBF1 gene may provide a biological hypothesis for the complex relationship between psychosocial stress, central obesity, diabetes, and cardiovascular disease.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Go AS, Mozaffarian D, Roger VL et al. Heart disease and stroke statistics-2013 update: a report from the American Heart Association. Circulation 2013; 127: e6–e245.
World Health Organization: Global health risk: mortality and burden of disease attributable to selected major risks. Tech. Rep. ISBN 978 92 4 156387 1. Geneva, Switzerland: WHO Press, 2009.
Elder SJ, Lichtenstein AH, Pittas AG et al. Genetic and environmental influences on factors associated with cardiovascular disease and the metabolic syndrome. J Lipid Res 2009; 50: 1917–1926.
Rosengren A, Hawken S, Ôunpuu S et al. Association of psychosocial risk factors with risk of acute myocardial infarction in 11,119 cases and 13,648 controls from 52 countries (the INTERHEART study): case-control study. Lancet 2004; 364: 953–962.
Williams RB : Psychosocial and biobehavioral factors and their interplay in coronary heart disease. Annu Rev Clin Psychol 2008; 4: 349–365.
Williams RB, Barefoot JC, Blumenthal JA et al. Psychosocial correlates of job strain in a sample of working women. Arch Gen Psychiatry 1997; 54: 543–548.
Scherwitz LW, Perkins LL, Chesrtey MA, Hughes GH, Sidney S, Manolio TA : Hostility and health behaviors in young adults: the CARDIA Study. Coronary Artery Risk Development in Young Adults Study. Am J Epidemiol 1992; 136: 136–145.
Siegler IC, Peterson BL, Barfoot JC, Williams RB : Hostility during late adolescence predicts coronary risk factors at mid-life. Am J Epidemiol 1992; 136: 146–154.
Speaker KJ, Fleshner M : Interleukin-1 beta: a potential link between stress and the development of visceral obesity. BMC Physiol 2012; 12: 1–15.
Labonte B, Yerko V, Gross J et al. Differential glucocorticoid receptor exon 1(B), 1(C), and 1(H) expression and methylation in suicide completers with a history of childhood abuse. Biol Psychiatry 2012; 72: 41–48.
Hunter DJ : Gene-environment interactions in human diseases. Nat Rev Genet 2005; 6: 287–298.
Thomas D : Gene-environment-wide association studies: emerging approaches. Nat Rev Genet 2010; 11: 259–272.
Bornstein SR, Schuppenies A, Wong ML, Licinio J : Approaching the shared biology of obesity and depression: the stress axis as the locus of gene-environment interactions. Mol Psychiatry 2006; 10: 892–902.
Bild DE, Bluemke DA, Burke GL et al. Multi-ethnic study of atherosclerosis: objectives and design. Am J Epidemiol 2002; 156: 871–881.
Lorenz MW, Markus HS, Bots ML, Rosvall M, Sitzer M : Prediction of clinical cardiovascular events with carotid intima-media thickness: a systematic review and meta-analysis. Circulation 2007; 115: 459–467.
Purcell S, Neale B, Todd-Brown K et al. PLINK: a toolset for whole-genome association and population-based linkage analysis. Am J Hum Genet 2007; 81: 560–575.
Price AL, Patterson NJ, Plenge RM et al. Principal components analysis corrects for stratification in genome-wide association studies. Nat Genet 2006; 38: 904–909.
Devlin B, Bacanu SA, Roeder K : Genomic control to the extreme. Nat Genet 2004; 36: 1129–1130.
Muthen LK, Muthen BO : Mplus User’s Guide Sixth Edition Muthen & Muthen: Los Angeles, CA, 1998–2010.
Streiner DL : Finding our way: an introduction to path analysis. Can J Psychiatry 2005; 50: 115–122.
Feinleib M, Kannel WB, Garrison RJ, McNamara PM, Castelli WP : The Framingham Offspring Study. Design and preliminary data. Prev Med 1975; 4: 518–525.
Lukin K, Fields S, Hartley J, Hagman J : Early B cell factor: regulator of B lineage specification and commitment. Semin Immunol 2008; 20: 221–227.
Nolan DK, Sutton B, Haynes C et al. Fine mapping of a linkage peak with integration of lipid traits identifies novel coronary artery disease genes on chromosome 5. BMC Genet 2012; 13: 12.
Hauser ER, Crossman DC, Granger CB et al. A genomewide scan for early-onset coronary artery disease in 438 families: the GENECARD study. Am J Hum Genet 2004; 75: 436–447.
Shah SH, Kraus WE, Crossman DC et al. Serum lipids in the GENECARD study of coronary artery disease identify quantitative trait loci and phenotypic subsets on chromosomes 3q and 5q. Ann Hum Genet 2006; 70: 738–748.
International Consortium for Blood Pressure Genome-Wide Association Studies: Genetic variants in novel pathways influence blood pressure and cardiovascular disease risk. Nature 2011; 478: 103–109.
Wain LV, Verwoert GC, O’Reilly PF et al. Genome-wide association study identifies six new loci influencing pulse pressure and mean arterial pressure. Nat Genet 2011; 43: 1005–1011.
Huan T, Zhang B, Wang Z et al. A systems biology framework identifies molecular underpinnings of coronary heart disease. Arterioscler Thromb Vasc Biol 2013; 33: 1427–1434.
Speliotes EK, Willer CJ, Berndt SI et al. Association analyses of 249,796 individuals reveal 18 new loci associated with body mass index. Nat Genet 2010; 42: 937–948.
Manning AK, Hivert MF, Scott RA et al. A genome-wide approach accounting for body mass index identifies genetic variants influencing fasting glycemic traits and insulin resistance. Nat Genet 2012; 44: 659–669.
Morris AP, Voight BF, Teslovich TM et al. Large-scale association analysis provides insights into the genetic architecture and pathophysiology of type 2 diabetes. Nat Genet 2012; 44: 981–990.
Schunkert H, König IR, Kathiresan S et al. Large-scale association analysis identifies 13 new susceptibility loci for coronary artery disease. Nat Genet 2011; 43: 333–338.
Fretz JA, Nelson T, Xi Y et al. Altered metabolism and lipodystrophy in the early B-cell factor 1-deficient mouse. Endocrinology 2010; 151: 1611–1621.
Jimenez MA, Akerblad P, Sigvardsson M, Rosen ED : Critical role for Ebf1 and Ebf2 in the adipogenic transcriptional cascade. Mol Cell Biol 2007; 27: 743–757.
Williams RB, Marchuk DA, Gadde KM et al. Serotonin-related gene polymorphisms and central nervous system serotonin function. Neuropsychopharmacology 2003; 28: 533–541.
Gong M, Hubner N : Molecular genetics of human hypertension. Clin Sci 2006; 110: 315–326.
Acknowledgements
This work was supported by NIH/NHLBI grants HL095987 (to Shah), HL073389-01 (to Hauser), and HL036587 (to Williams). The MESA and Framingham data sets were obtained from NIH dbGaP.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Redford B Williams is a founder of and stockholder in Williams LifeSkills, Inc. The other authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on European Journal of Human Genetics website
Supplementary information
Rights and permissions
About this article
Cite this article
Singh, A., Babyak, M., Nolan, D. et al. Gene by stress genome-wide interaction analysis and path analysis identify EBF1 as a cardiovascular and metabolic risk gene. Eur J Hum Genet 23, 854–862 (2015). https://doi.org/10.1038/ejhg.2014.189
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/ejhg.2014.189
This article is cited by
-
Genetic Contributions to Risk of Adverse Pregnancy Outcomes
Current Cardiovascular Risk Reports (2023)
-
Pleiotropic genetic influence on birth weight and childhood obesity
Scientific Reports (2021)
-
The -514C>T polymorphism in the LIPC gene modifies type 2 diabetes risk through modulation of HDL-cholesterol levels in Mexicans
Journal of Endocrinological Investigation (2021)
-
Evaluating the precision of EBF1 SNP x stress interaction association: sex, race, and age differences in a big harmonized data set of 28,026 participants
Translational Psychiatry (2020)
-
Developing a synthetic psychosocial stress measure and harmonizing CVD-risk data: a way forward to GxE meta- and mega-analyses
BMC Research Notes (2018)